The robotic surgical team in the operating room plays an important role in determining the outcome of a robotic approach. This study aimed to compare the outcomes of 2 hospitals with different levels of expertise in robot-assisted rectal cancer surgery.
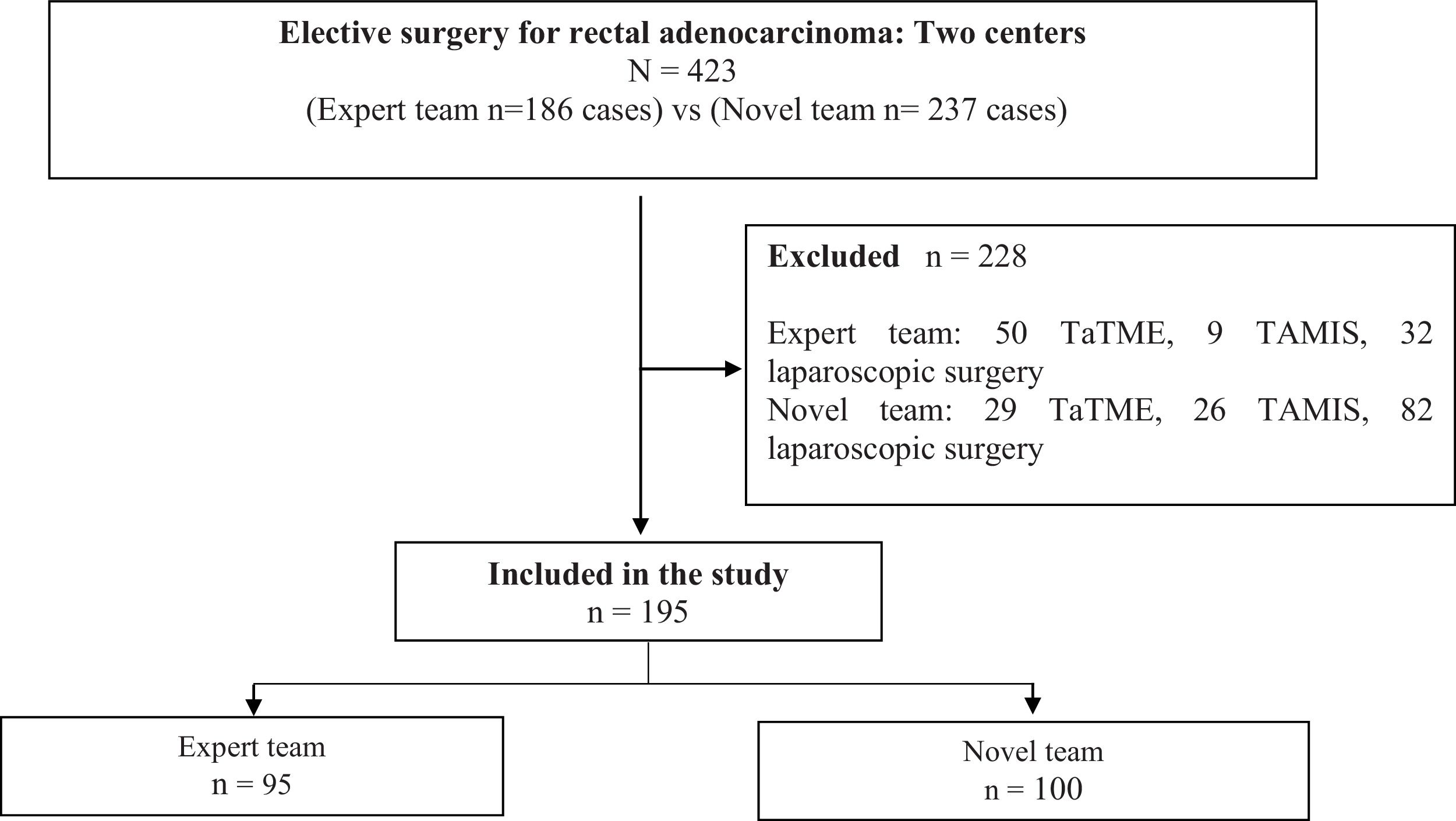
MethodsThis retrospective study analyzed 195 patients who underwent robot-assisted rectal resection at 2 referral centers for the treatment of rectal cancer between March 2018 and December 2021.
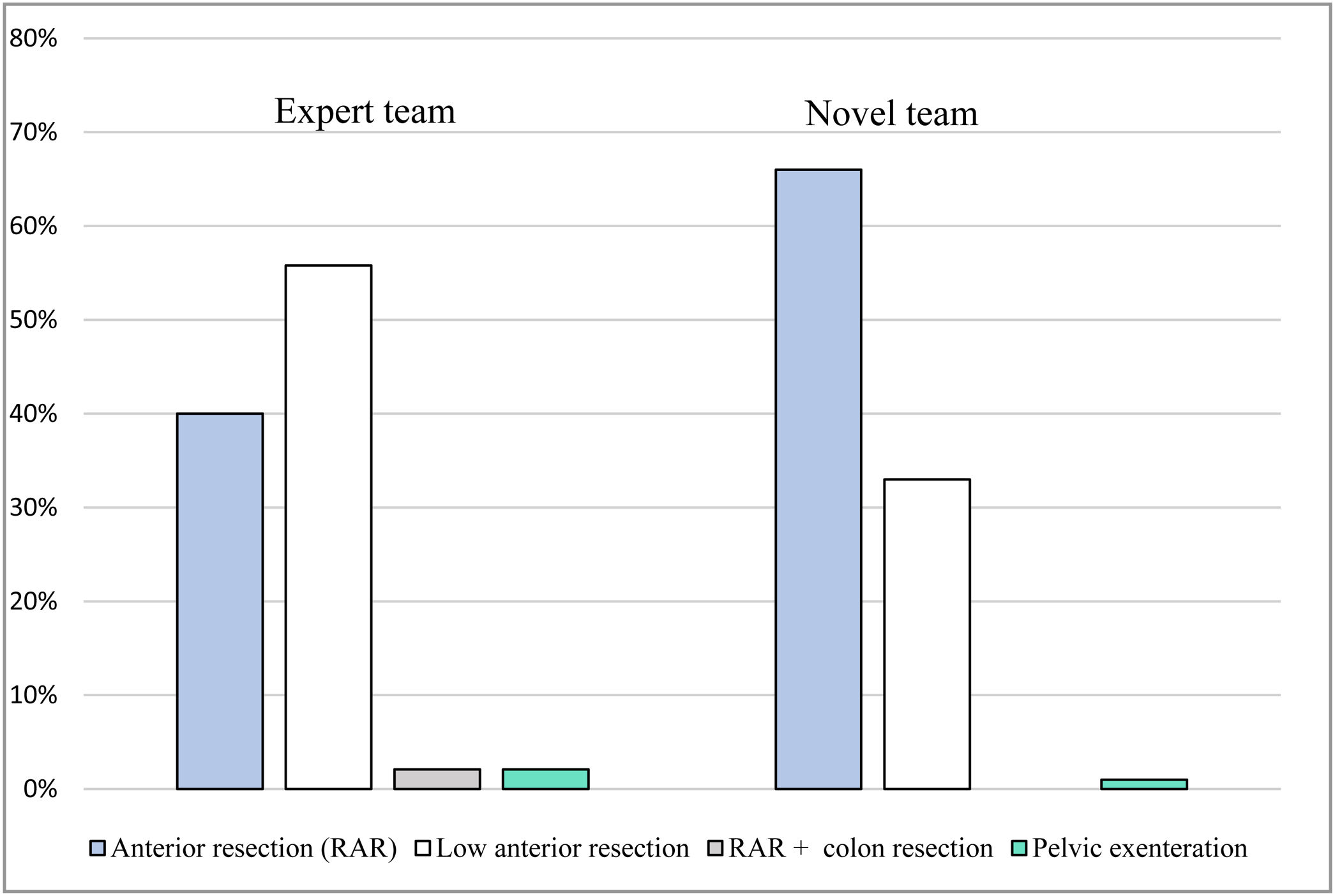
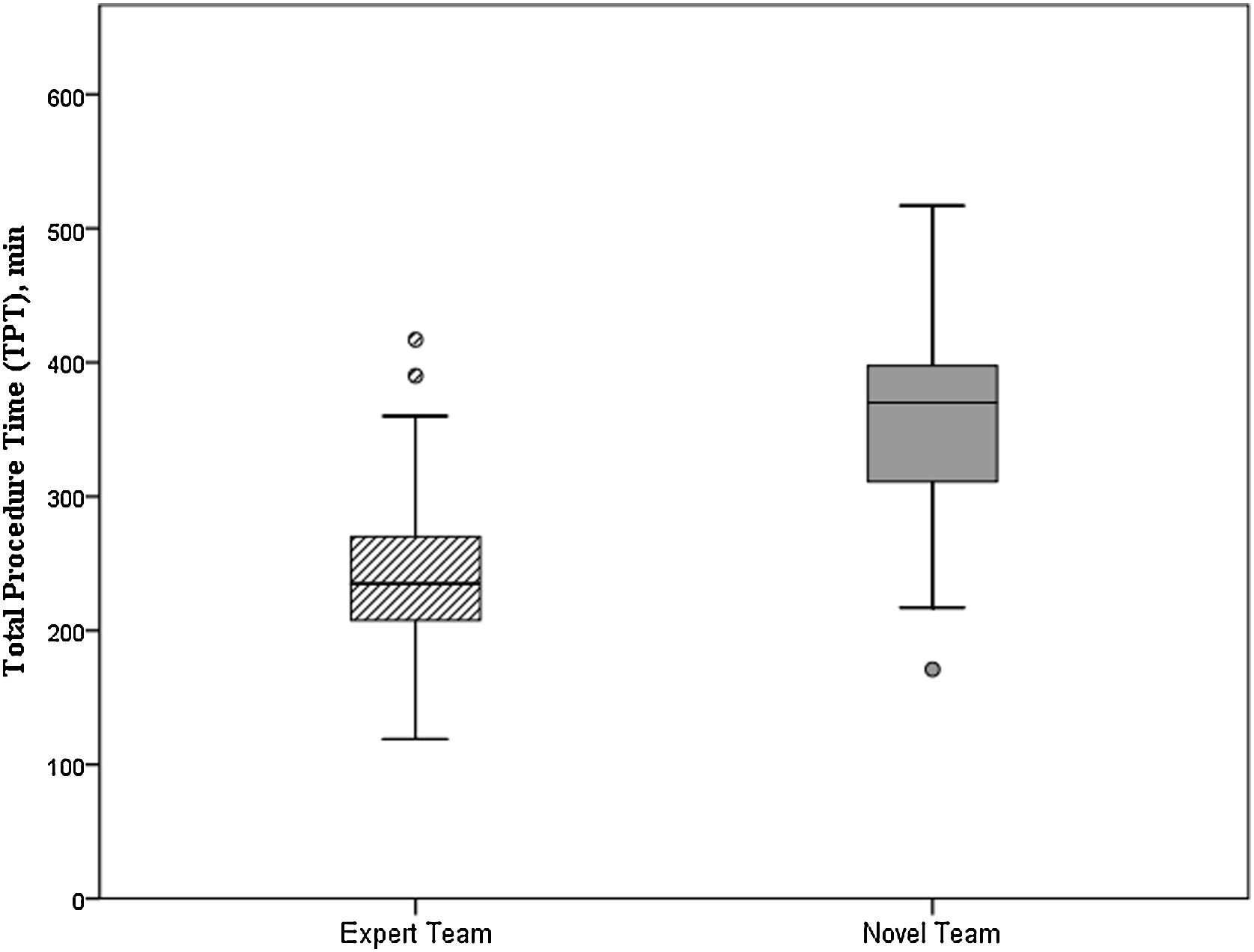
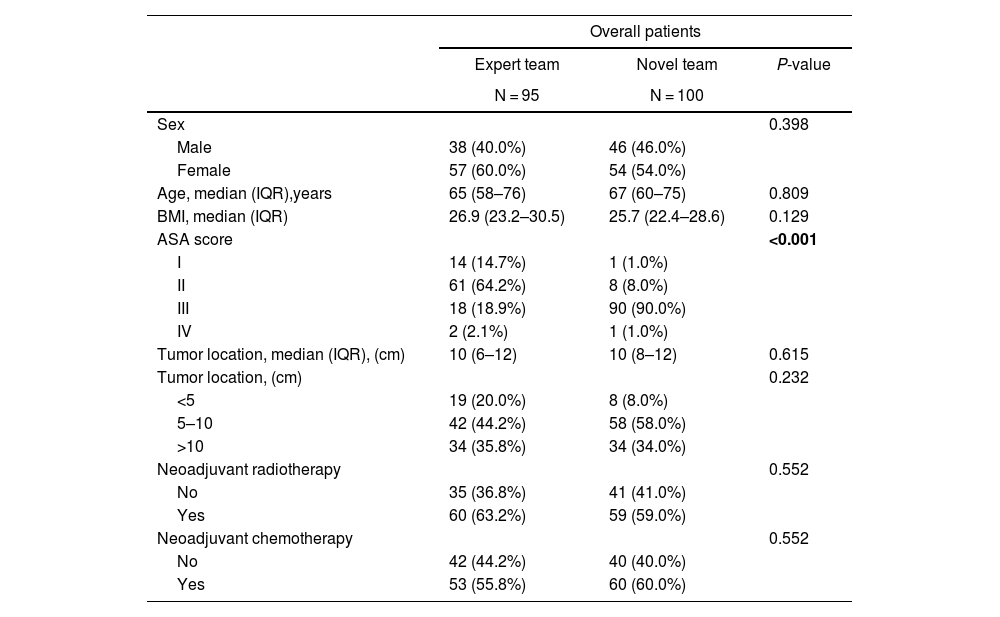
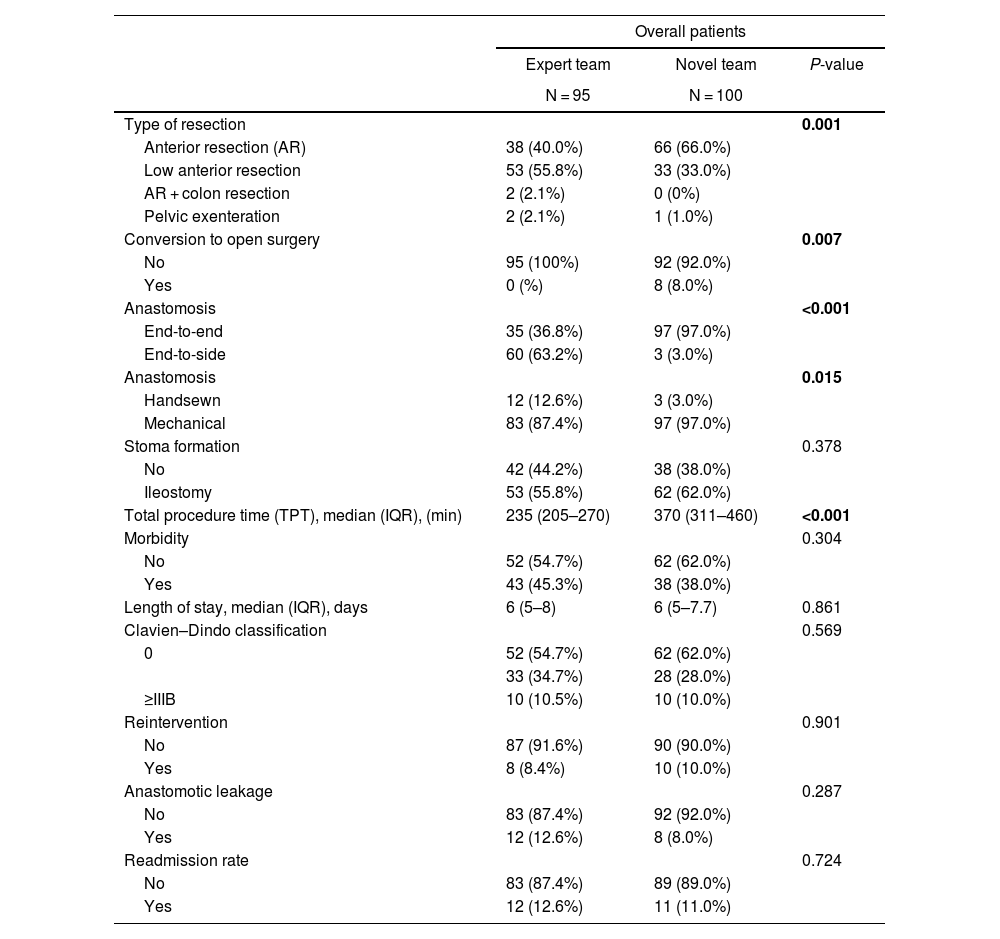
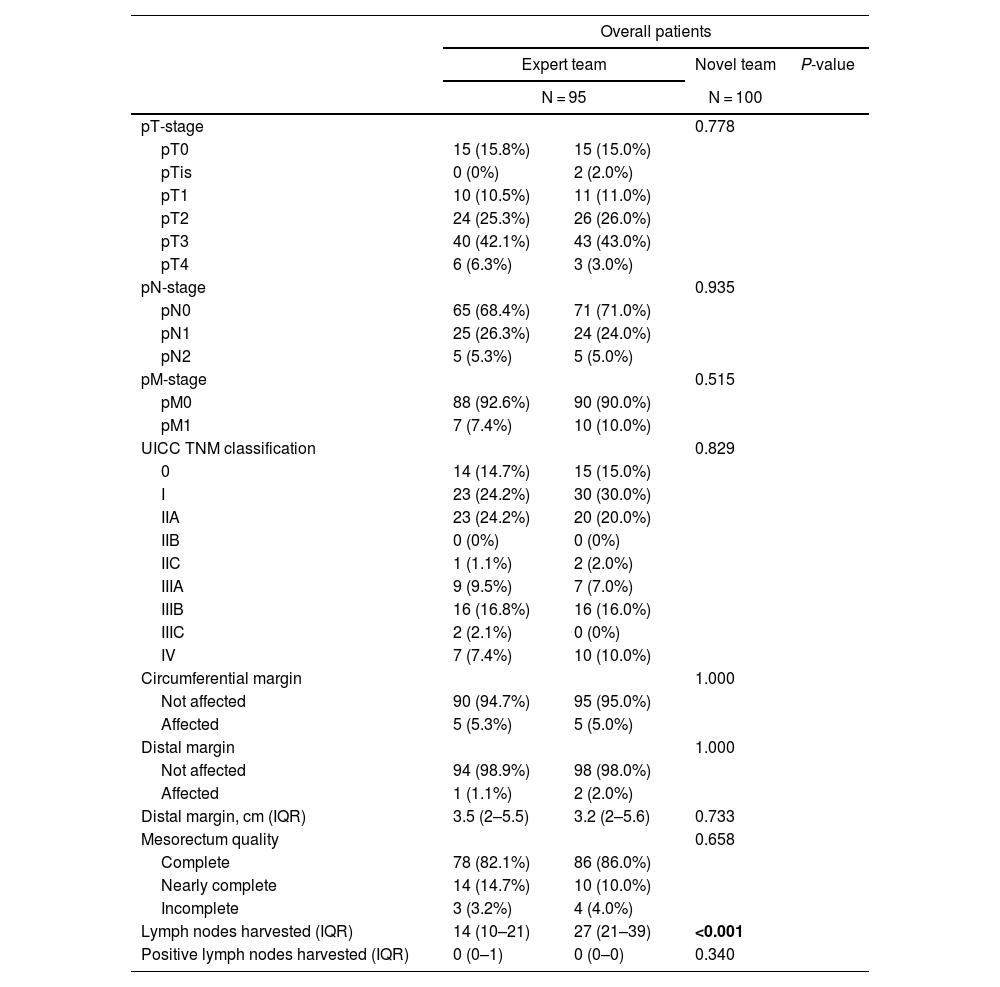
ResultsIn total, 195 patients had undergone robotic rectal cancer surgery: 95 performed by an expert team, and 100 by a novel team. The expert team performed more low anterior resections (55.8%) than the novel team (33%) (P = 0.001), and the total operative time varied significantly between the groups (P < 0.001). The novel team’s operative time was 135 min longer than the expert team’s. The expert team had no conversions to open surgery, while the novel team had an 8% conversion rate (P = 0.007). In this study, overall morbidity was 45.3% among patients treated by the expert team versus 38% among those treated by the novice team (P = 0.304). Severe complications (Clavien–Dindo grade >IIIB) occurred at a rate of 10% in both groups. Incomplete mesorectal excision was observed in 3.2% of the expert team’s patients versus 4.2% of the novice team’s (P = 0.65).
ConclusionThe expert team achieved a shorter operative time and less conversion to open surgery. However, the morbidity and pathological outcomes were comparable between the teams. The introduction of robotic surgery in a team with early-stage surgical experience was safe.
El equipo quirúrgico robótico en el quirófano desempeña un papel importante en la determinación de resultados. Este estudio tuvo como objetivo comparar los resultados de dos centros con diferentes niveles de experiencia en la cirugía robótica para el cáncer de recto.
MétodosEste estudio retrospectivo analizó a 195 pacientes que se sometieron a resección rectal asistida por robot en dos centros de referencia para el tratamiento del cáncer de recto entre marzo de 2018 y diciembre de 2021.
ResultadosUn total de 195 pacientes se sometieron a cirugía robótica para el cáncer de recto: 95 de un equipo experto y 100 de un equipo novel. El equipo experto realizó más resecciones anteriores bajas (55.8%) en comparación con el equipo novel (33%) (p = 0.001), y el tiempo operatorio total varió significativamente entre los centros (p < 0.001). El tiempo operatorio del equipo novel fue 135 minutos más largo que el del equipo experto. El equipo experto no tuvo conversiones a cirugía abierta, mientras que el equipo novel tuvo una tasa de conversión del 8% (p = 0.007). En este estudio, la morbilidad general fue del 45.3% en el equipo experto y del 38% en el equipo novel (p = 0.304). Las complicaciones graves (clasificación Clavien-Dindo > IIIB) ocurrieron a una tasa del 10% en ambos grupos. Se observó una escisión mesorrectal incompleta en el 3.2% de los pacientes del equipo experto y en el 4.2% del equipo novel (p = 0.65).
ConclusiónEl equipo experto logró un menor tiempo operatorio y menos conversiones a cirugía abierta. Sin embargo, la morbilidad y los resultados patológicos fueron comparables entre los equipos. La introducción de la cirugía robótica en un equipo con experiencia quirúrgica temprana fue segura.