There is uncertainty about the role of triple inhaled therapy with LAMA/LABA/ICS (long-acting muscarinic antagonist/long-acting β2-agonist/inhaled glucocorticoids) in chronic obstructive pulmonary disease (COPD) on cardiovascular mortality. We estimated the effect of triple inhaled therapy (TT) compared with dual inhaled therapy (DT, including either LAMA/LABA or LABA/ICS) on all-cause and cardiovascular mortality in an evidence synthesis,
MethodsFollowing prospective registration (https://osf.io/gtfvm), a comprehensive search strategy of PubMed, Scopus, and Embase was performed, without language or time restrictions until September 30, 2024. All randomized clinical trials (RCTs) evaluating TT vs. DT and reporting cardiovascular or all-cause mortality were included. We assessed risk of bias and conducted a random effect meta-analysis estimating summary relative risk (RR) with 95% confidence intervals (CI), evaluating heterogeneity using I2. A network meta-analysis (NMA) was undertaken to hierarchically rank the therapies using P-score.
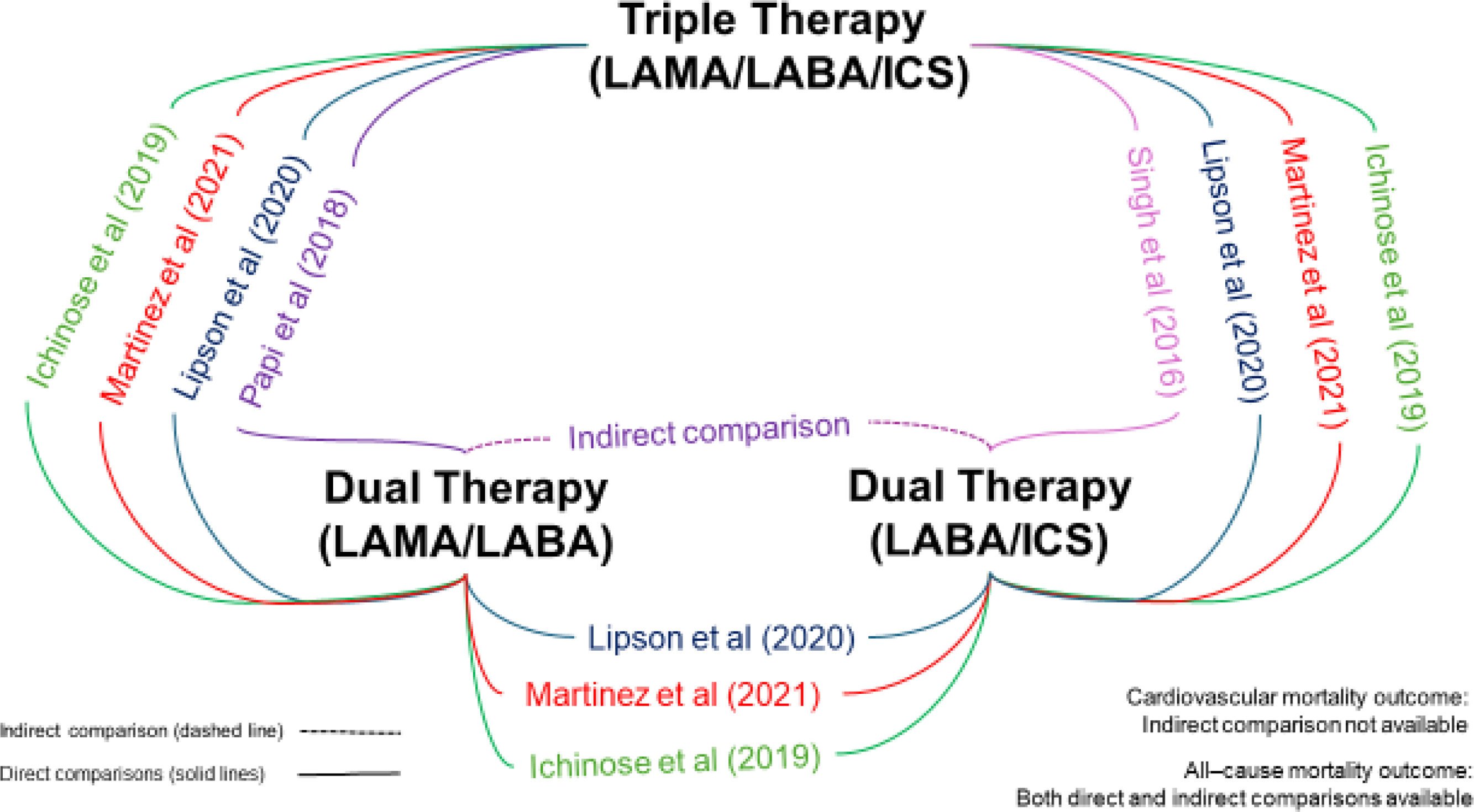
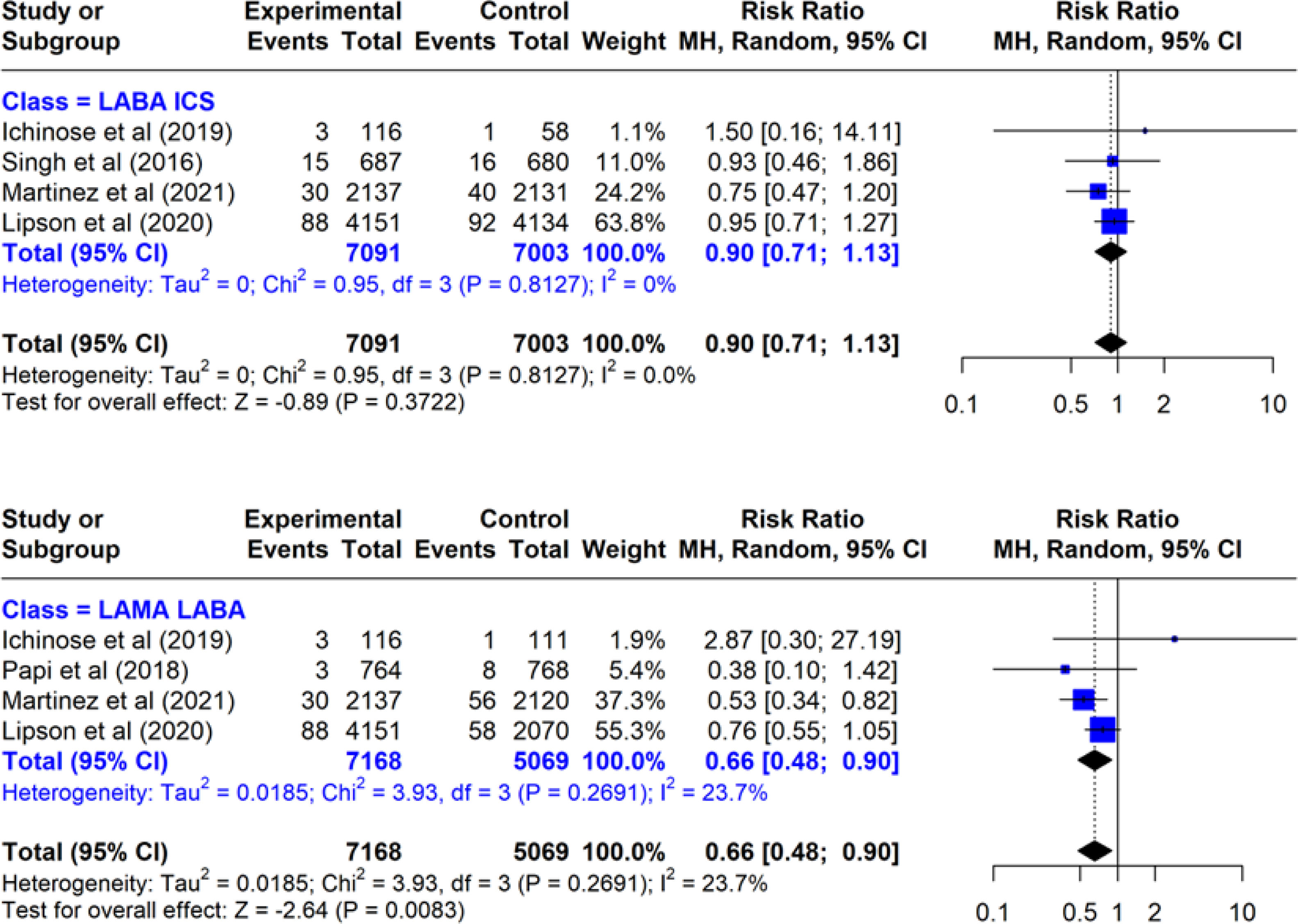
ResultsFrom 781 citations, 5 RCTs were selected. There were 3 three-arm RCTs comparing TT vs. LABA/ICS vs. LAMA/LABA, 1 two-arm RCT comparing TT vs. LABA/ICS, and 1 two-arm RCT comparing TT vs. LAMA/LABA (total of 7855 patients receiving TT, 7003 LABA/ICS and 5059 LAMA/LABA). The risk of bias was moderate in 2 (40%), and low in 3 (60%) RCTs. TT reduced cardiovascular mortality by 48% vs. LAMA/LABA (RR 0.52, 95% CI 0.32–0.86, 3 RCTs, I2=0%) and by a non-significant 11% vs. LABA/ICS (RR 0.89, 95% CI 0.57–1.37, 3 RCTs, I2=0%). TT reduced all-cause mortality by 34% vs. LAMA/LABA (RR 0.66, 95% CI 0.48–0.90, 4 RCTs, I2=23.7%) and by 10% vs. LABA/ICS (RR 0.90, 95% CI 0.71–1.13, 4 RCTs, I2=0%). For both cardiovascular and all-cause mortality, NMA P-score showed that TT ranked first (81%/91%), LABA/ICS ranked second (58%/57%) and LAMA/LABA ranked last (11%/<1%) in effectiveness.
ConclusionsIn patients with moderate to very severe COPD and previous exacerbations, TT inhaled significantly reduces cardiovascular and all-cause mortality compared to LAMA/LABA dual therapy, but not when compared to LABA/ICS.
No está claramente definido el efecto sobre las complicaciones cardiovasculares de la triple terapia inhalada (TT) con LAMA/LABA/ICS (antagonista muscarínico de acción prolongada/agonista β2 de acción prolongada/glucocorticoides inhalados) en la enfermedad pulmonar obstructiva crónica (EPOC). Para ello realizamos una revisión sistemática y metaanálisis para estimar el efecto de la TT en comparación con la doble terapia inhalada (DT) sobre la mortalidad cardiovascular y la mortalidad por todas las causas.
MétodosTras el registro prospectivo (https://osf.io/gtfvm), se realizó una estrategia de búsqueda exhaustiva en PubMed, Scopus y Embase, sin restricciones de idioma o tiempo hasta el 30 de septiembre de 2024. Se incluyeron todos los ensayos clínicos aleatorizados (ECA) que evaluaron TT frente a DT simultáneamente y que disponían de datos de mortalidad cardiovascular y por cualquier causa. Se evaluó el riesgo de sesgo y se realizó un metaanálisis de efectos aleatorios estimando el riesgo relativo (RR) con intervalos de confianza (IC) del 95%, evaluando la heterogeneidad mediante I2. Se realizó un metaanálisis de red (NMA) para clasificar jerárquicamente los tratamientos.
ResultadosDe 781 citas, se seleccionaron 5 ECA. Tres ECA de 3 brazos que compararon TT frente a LABA/ICS y LAMA/LABA, un ECA de 2 brazos que comparó TT frente a LABA/ICS y un ECA de 2 brazos que comparó TT frente a LAMA/LABA (un total de 7855 pacientes recibieron TT, 7003 LABA/ICS y 5059 LAMA/LABA). El riesgo de sesgo de los ECA fue moderado en 2 (40%) y bajo en 3 (60%) ECA. La TT redujo la mortalidad cardiovascular en un 48% frente a LAMA/LABA (RR 0,52; IC 95% 0,32-0,86; 3 ECA; I2=0%) y en un 11% no estadísticamente significativo frente a LABA/ICS (RR 0,89; IC 95% 0,57-1,37; 3 ECA; I2=0%). La TT redujo la mortalidad por todas las causas en un 34% frente a LAMA/LABA (RR 0,66; IC 95% 0,48-0,90; 4 ECA; I2=23,7%) y en un 10% frente a LABA/ICS (RR 0,90; IC 95% 0,71-1,13; 4 ECA; I2=0%). Tanto para la mortalidad cardiovascular como para la mortalidad por todas las causas, la TT fue jerárquicamente más eficiente (81%/91%, respectivamente), LABA/ICS la segunda (58%/57%) y LAMA/LABA la tercera (11%/<1%).
ConclusionesEn pacientes con EPOC de moderada a muy severa y con exacerbaciones previas, la TT reduce significativamente la mortalidad cardiovascular y por cualquier causa en comparación con la terapia dual LAMA/LABA, pero no cuando se compara con LABA/ICS.
Artículo
Diríjase al área privada de socios de la web de la SEMERGEN, (https://www.semergen.es/index.php?seccion=biblioteca&subSeccion=revistaSEMERGEN ) y autentifíquese.