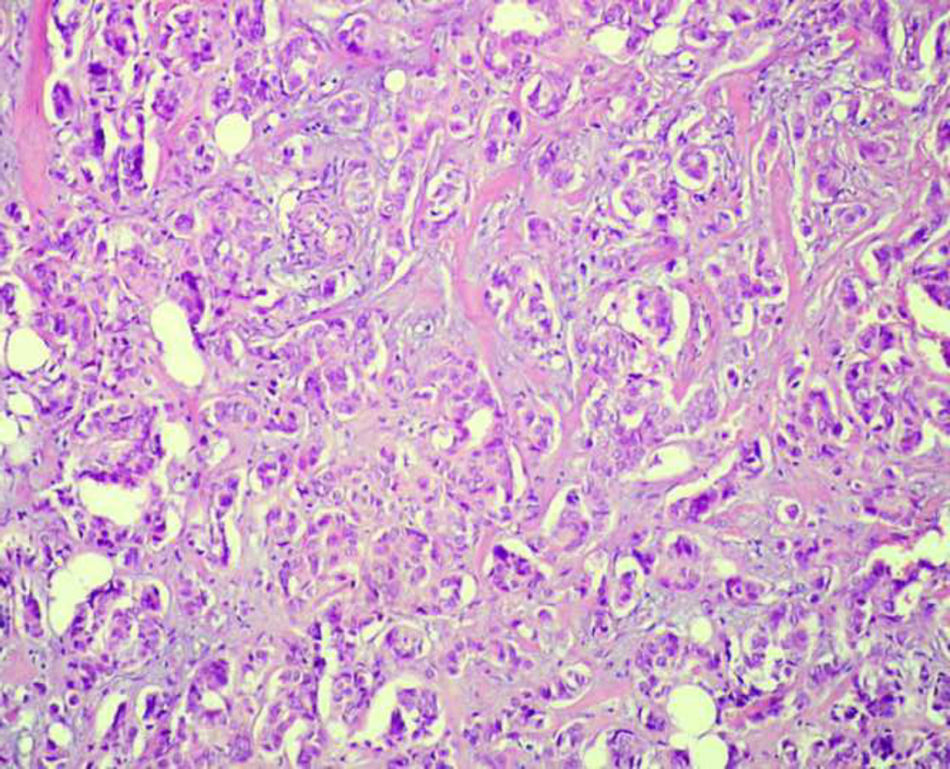
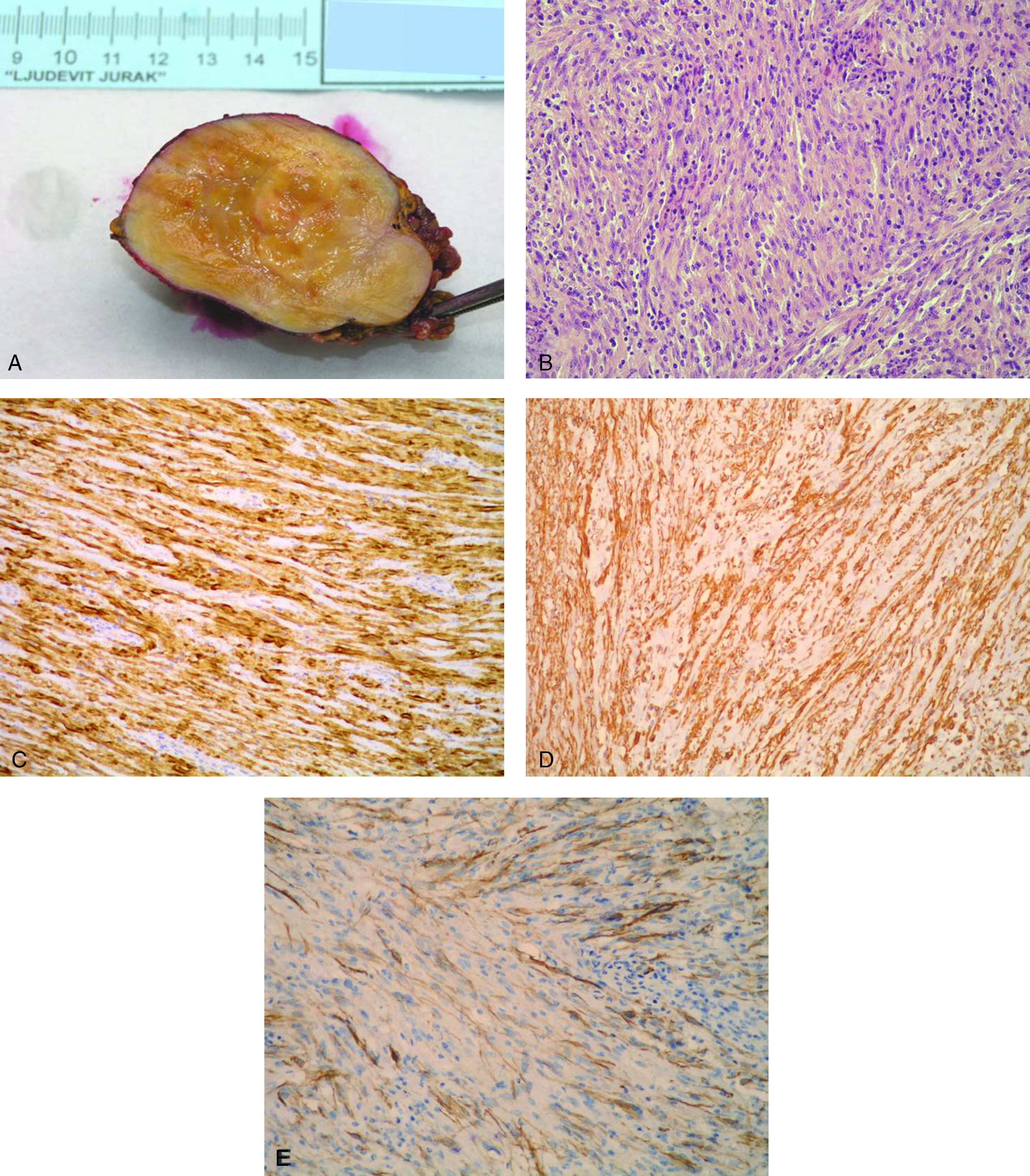
Schwannoma is a benign, usually encapsulated, nerve sheath tumour originating from Schwann cells. Primary schwannomas of the adrenal gland are extremely rare.
Case reportWe present a case of 55 year old female patient with breast cancer, treated with mastectomy and lymphadenectomy with subsequent hormonal therapy. Three months after mastectomy, ultrasonography revealed a hormonally inactive tumour in the left adrenal gland which was suspected to be a metastasis. Adrenalectomy was performed and the microscopic and immunohistochemical examinations revealed the tumour to be a schwannoma.
ConclusionAs adrenal schwannomas are so infrequent, they can pose diagnostic problems and should be differentiated from other spindle cell lesions of the adrenal gland or metastatic tumours.
El schwannoma es un tumor benigno, encapsulado, de la vaina de mielina, originándose en las células de Schwann. El schwannoma primario de la glándula suprarrenal es muy infrecuente.
Caso clínicoSe presenta un caso de una paciente de 55 años, diagnosticada de cáncer de mama y tratada con mastectomía, linfadenectomía y terapia hormonal. Tres meses tras la mastectomía se detectó ecográficamente un tumor, sin actividad hormonal, en la glándula suprarrenal izquierda. Se sospechó una metástasis y se realizó una adrenalectomía. La histología e inmunohistoquímica del tumor fueron consistentes con schwannoma.
ConclusiónDebido a la poca frecuencia del schwannoma suprarrenal pueden ocurrir problemas diagnósticos y deben diferenciarse de otras lesiones de células de la glándula suprarrenal o de tumores metastásicos.