We aim to describe the use of emergency electroencephalogram (EmEEG) by the on-call neurologist when nonconvulsive status epilepticus (NCSE) is suspected, and in other indications, in a tertiary hospital.
Subjects and methodsObservational retrospective cohort study of emergency EEG (EmEEG) recordings with 8-channel systems performed and analysed by the on-call neurologist in the emergency department and in-hospital wards between July 2013 and May 2015. Variables recorded were sex, age, symptoms, first diagnosis, previous seizure and cause, previous stroke, cancer, brain computed tomography, diagnosis after EEG, treatment, patient progress, routine control EEG (rEEG), and final diagnosis. We analysed frequency data, sensitivity, and specificity in the diagnosis of NCSE.
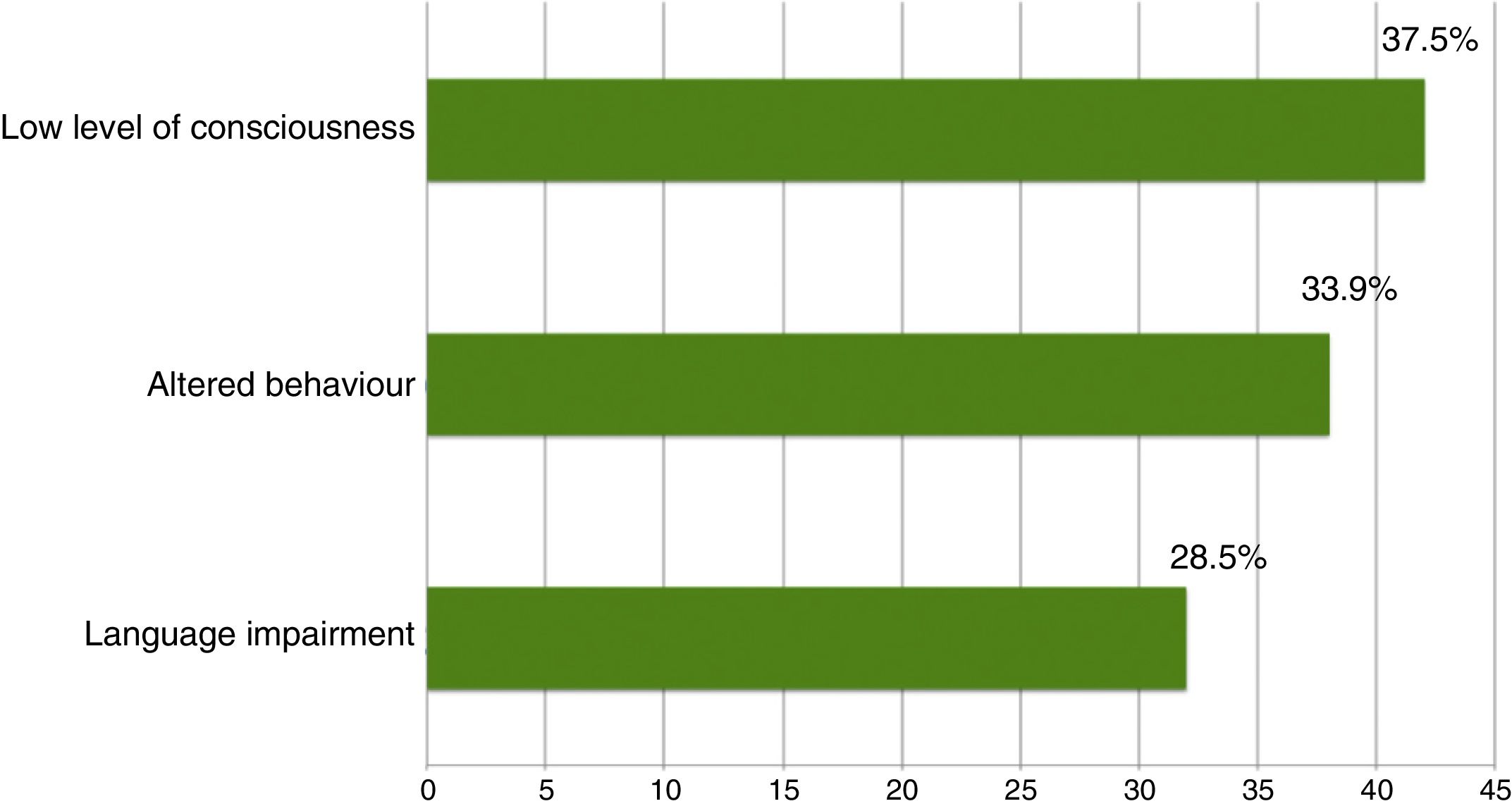
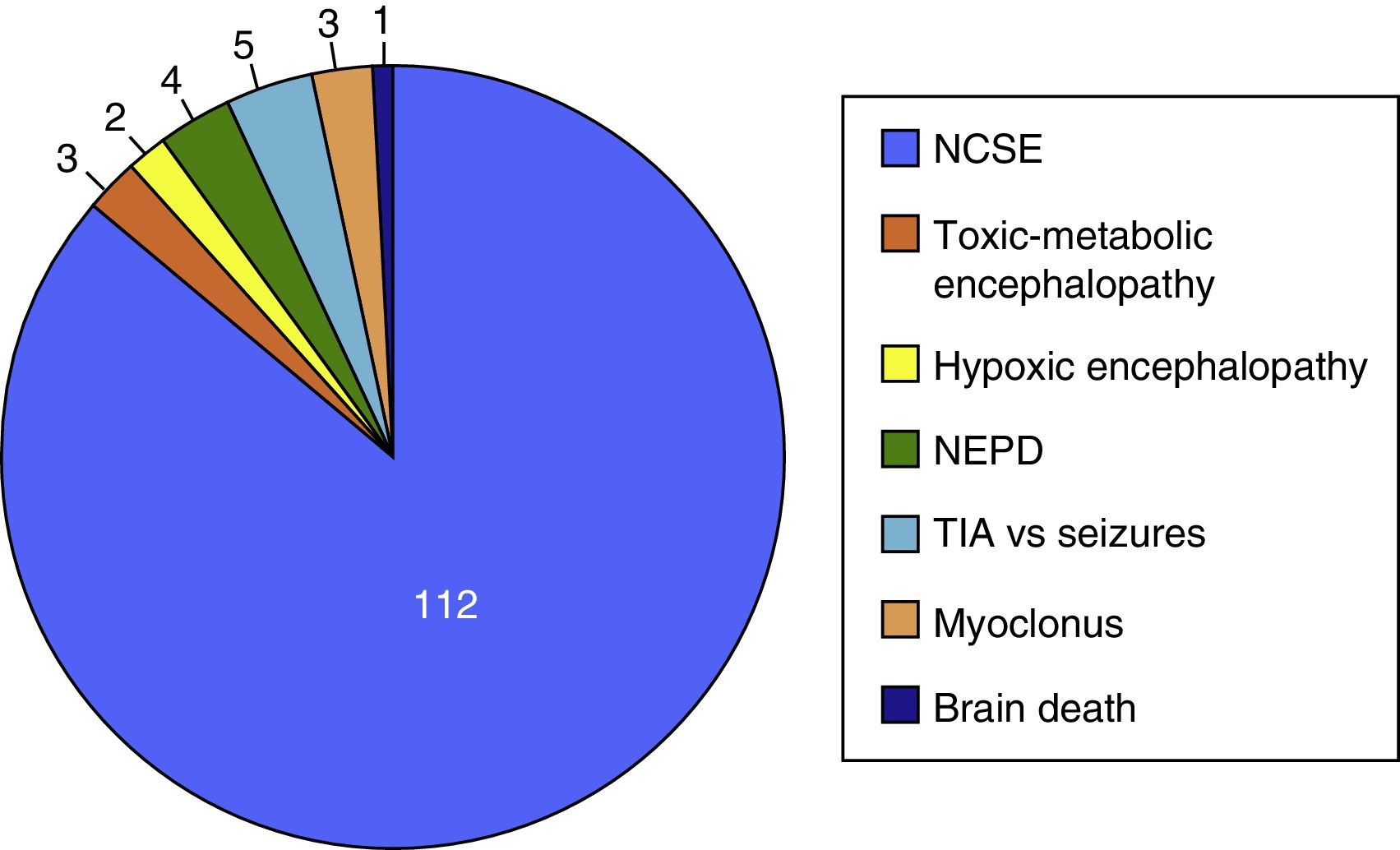
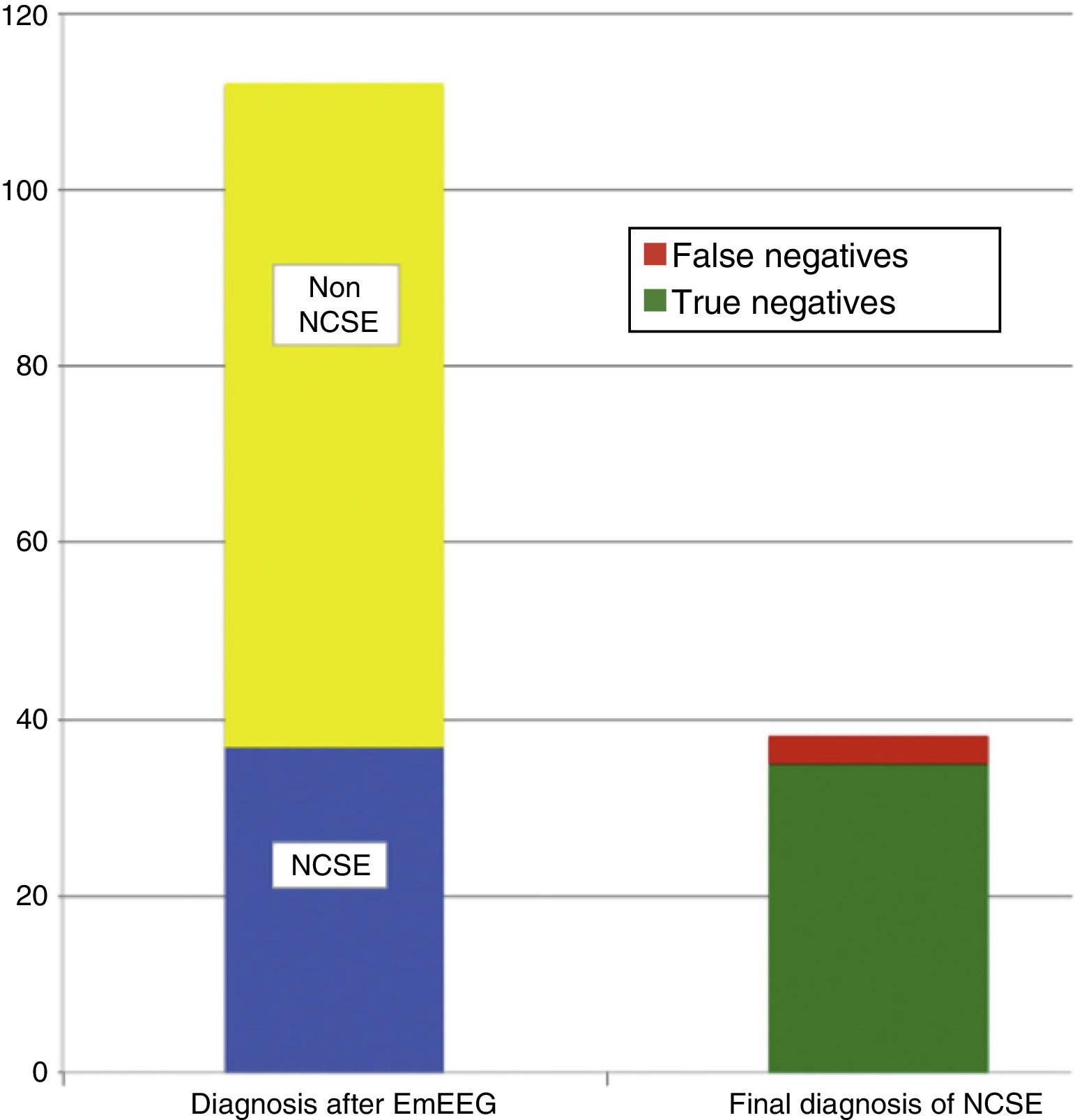
ResultsThe study included 135 EEG recordings performed in 129 patients; 51.4% were men and their median age was 69 years. In 112 cases (83%), doctors ruled out suspected NCSE because of altered level of consciousness in 42 (37.5%), behavioural abnormalities in 38 (33.9%), and aphasia in 32 (28.5%). The EmEEG diagnosis was NCSE in 37 patients (33%), and this was confirmed in 35 (94.6%) as the final diagnosis. In 3 other cases, NCSE was the diagnosis on discharge as confirmed by rEEG although the EmEEG missed this condition at first. EmEEG performed to rule out NCSE showed 92.1% sensitivity, 97.2% specificity, a positive predictive value of 94.6%, and a negative predictive value of 96%.
ConclusionsOur experience finds that, in an appropriate clinical context, EmEEG performed by the on-call neurologist is a sensitive and specific tool for diagnosing NCSE.
Estudio que describe la experiencia en el uso del electroencefalograma urgente (EEGurg) por la guardia de neurología ante la sospecha de estatus epiléptico no convulsivo (EENC) y en otras indicaciones en un hospital terciario.
Sujetos y métodosEstudio observacional retrospectivo de los registros de EEG de 8 canales realizados con carácter urgente en pacientes hospitalizados y en Urgencias e interpretados por la guardia de neurología entre julio del 2013 y mayo del 2015. Se recogieron las siguientes variables: sexo, edad, síntomas, diagnóstico inicial, epilepsia previa y causa, ictus previo, neoplasia, tomografía computarizada cerebral urgente, diagnóstico tras EEGurg, actitud terapéutica, evolución, EEG convencional (EEGc) de control y diagnóstico definitivo. Se analizaron los datos de frecuencia, así como la sensibilidad y especificidad en el diagnóstico de EENC.
ResultadosSe recogieron 135 registros de EEGurg realizados a 129 pacientes, el 51,4% hombres, edad mediana 69 años. En 112 casos (83%) la indicación fue descartar EENC por alteración del nivel de consciencia 42 (37,5%), del comportamiento 38 (33,9%) y del lenguaje 32 (28,5%). En 37 (33%) registros se informó como EENC, siendo este el diagnóstico definitivo en 35 (94,6%). En otros 3 casos, el EENC se diagnosticó en el EEGc de control tras pasar desapercibido en la valoración del EEGurg por la guardia. El EEGurg en el diagnóstico del EENC presenta una sensibilidad del 92,1%, una especificidad del 97,2%, un valor predictivo positivo del 94,6% y un valor predictivo negativo del 96% tomando como referencia el diagnóstico al alta.
ConclusionesEn nuestra experiencia, en un contexto clínico adecuado, el EEGurg utilizado por la guardia de neurología es una herramienta sensible y específica en el diagnóstico del EENC.
Non-convulsive status epilepticus (NCSE) is defined as an epileptic seizure lasting longer than 30minutes and with electroencephalogram (EEG) results showing continuous or recurrent epileptiform activity.1–3 As a clinical syndrome, NCSE includes a series of symptoms related to alterations in mental state,4,5 behaviour,6 language,7 emotions,8 sensory perception,9 and consciousness.10–12 From the perspective of EEG, there has historically been controversy regarding NCSE diagnosis,13,14 with continuous attempts by the International League Against Epilepsy (ILAE) to define the electrical characteristics of NCSE and to overcome the obstacles to establishing a universally accepted definition.15,16 This lack of consensus is rooted in the difficulty of assessing EEG patterns associated with NCSE in different contexts of age, brain development, comorbidity, encephalopathies, and epileptic syndromes.17 A series of EEG patterns associated with the corresponding clinical presentations has recently been published,18 defining EEG criteria for the diagnosis of NCSE19 which are applicable to everyday clinical practice.20 De novo NCSE manifests in nearly 30% of cases21; therefore, even in patients with no history of epilepsy, NCSE should be considered in the differential diagnosis of such conditions as ischaemic or haemorrhagic stroke, post-traumatic amnesia, transient global amnesia, autoimmune or infectious encephalitis, toxic-metabolic encephalopathy, and psychiatric disorders17 as a complication of these conditions and in cases of acute brain damage.22 In many cases, these are severe and potentially treatable conditions requiring emergency care in order to start specific treatment early, which improves short- and long-term outcomes.23,24 Several authors propose monitoring with emergency EEG (EmEEG) and a therapeutic trial of intravenous benzodiazepines during the recording if NCSE is suspected.25–27 The literature establishes no widely accepted time interval between EmEEG (considered to refer to the 24-hour availability of EEG recording and interpretation) being ordered and performed.27–29 In the past, NCSE was considered a relatively benign condition with very low morbidity and mortality rates30; therefore, it was preferred not to treat some cases to avoid the risks associated with antiepileptic drugs (AED) in patients with other disorders.17,31 NCSE in itself is currently considered to be less responsive to treatment than status epilepticus26; prolonged NCSE is associated with refractory symptoms,32,33 poor prognosis, and cognitive sequelae.31,34,35 Our aim is to describe the experience of the on-call neurologists from our hospital with the performance of EmEEG due to suspicion of NCSE and other situations, and to analyse its usefulness in the diagnosis of NCSE.
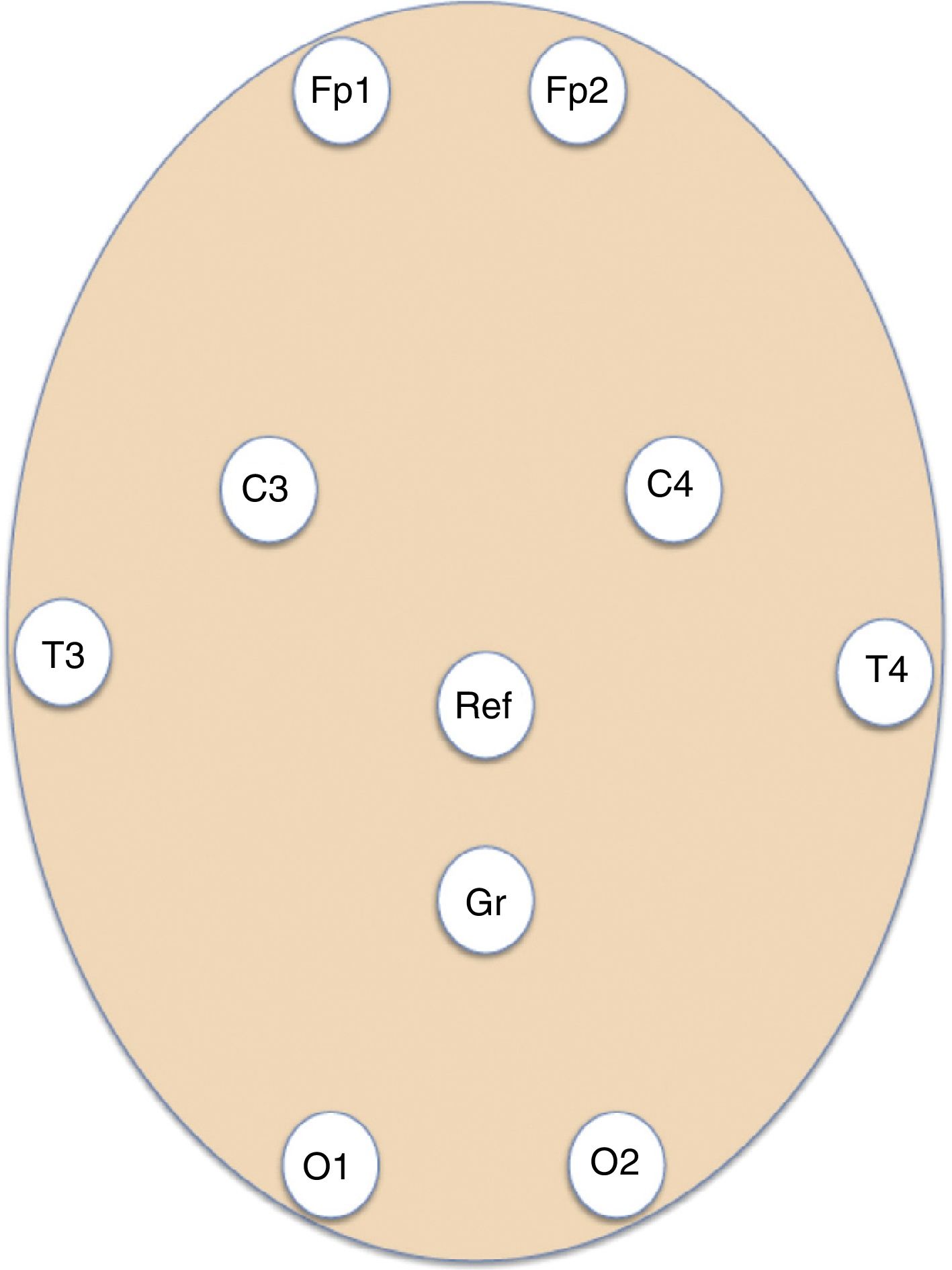
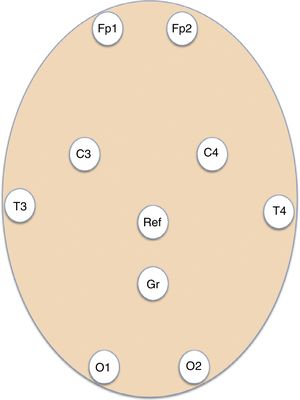
Patients and methodsWe conducted a retrospective, longitudinal, observational study of patients assessed by the on-call emergency neurology team and admitted to hospital wards and critical care units at a tertiary hospital providing neurosurgery to a population of 1 071 666 people. Patients underwent EmEEG studies, which were recorded and interpreted by the on-call neurologists between July 2013 and May 2015. Our hospital's on-call neurology team comprises a neurology consultant and 2 residents: a second-year (or higher) neurology resident, and either a third-year (or higher) neurology resident or a clinical neurophysiology resident. The second resident in the team had been trained in EmEEG recording. Furthermore, epilepsy experts at our hospital organised continuous training workshops on EEG interpretation for neurology residents and consultants before the start of the study. EmEEGs were performed with a portable digital EEG device (NicoletOne EEG, VYASIS Healthcare) (Fig. 1). Self-adhesive electrodes (Fp1, Fp2, T3, T4, C3, C4, O1, O2) were placed, as well as reference and ground electrodes (Fig. 2), enabling a system with 8 EEG channels and one ECG channel (Fig. 3). The duration of recording varied according to the patient, ranging from 20minutes to several hours. Demographic data on patients’ sex and age were gathered from clinical histories; regarding initial management at the emergency department, we gathered data on symptoms leading to indication of EmEEG, and clinical diagnosis. We recorded data on previous history of epilepsy, aetiology and type of epilepsy, previous stroke, active or remitting neoplasm, and emergency complementary tests (results from brain computed tomography and laboratory analysis). After the EmEEG, we also analysed: interpretation of the EmEEG recording by the on-call team, therapeutic trial with benzodiazepines during the EmEEG, diagnosis after EmEEG, therapeutic approach after EmEEG, patient progression, results of the conventional follow-up EEG (cEEG), and final diagnosis at discharge. Conventional EEG refers to an EEG recording of 16 or more channels performed during normal consultation hours, obtained by specialised nursing staff and interpreted by EEG experts. We analysed frequency data and sensitivity and specificity in the diagnosis of NCSE during the on-call shift, taking final diagnosis at discharge as a reference. Descriptive statistics were analysed using the SPSS statistical software version 15.0 for Windows.
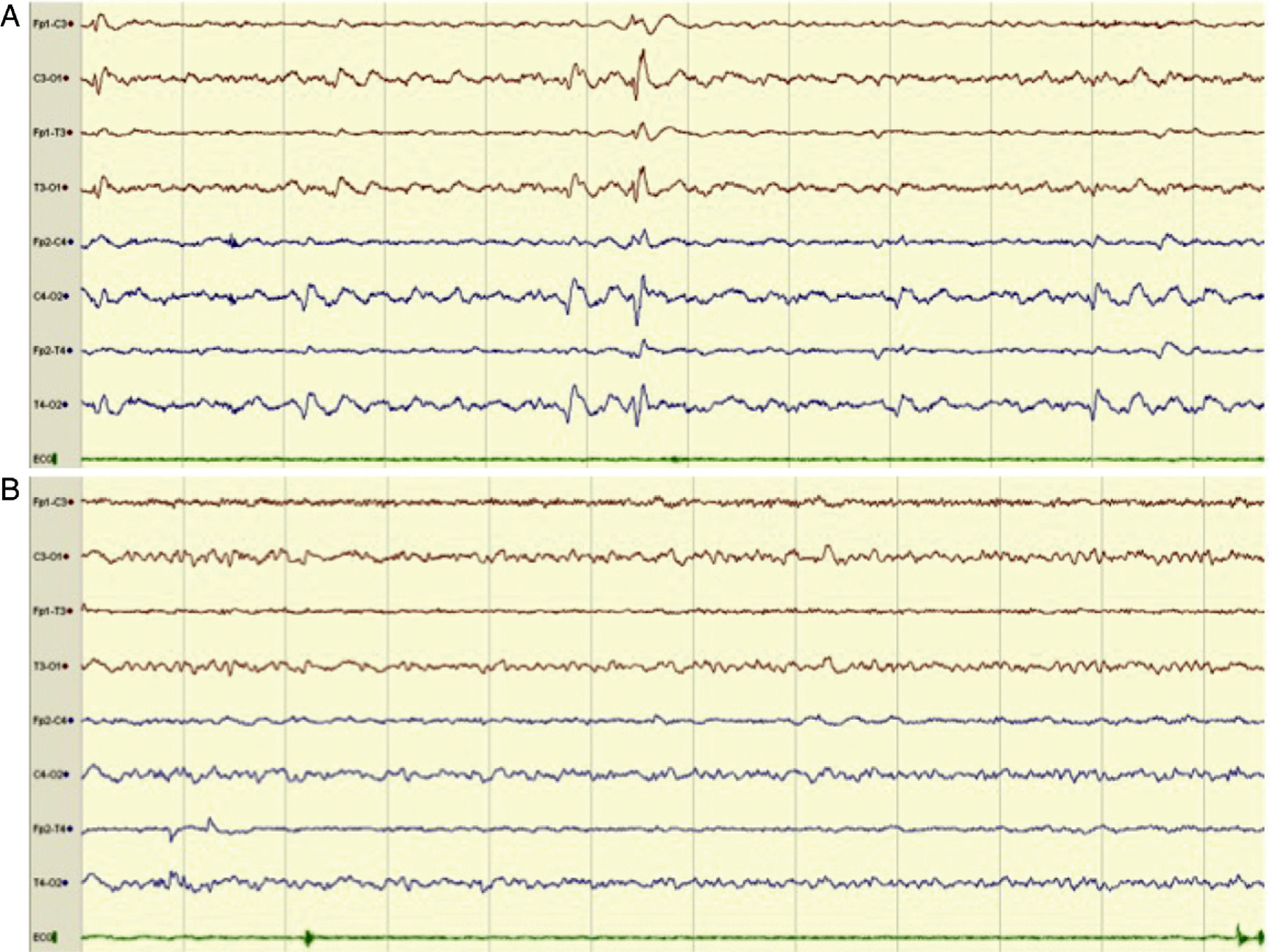
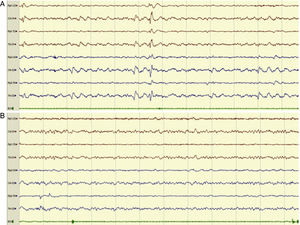
Example of an 8-channel emergency EEG recording showing initial epileptiform anomalies (EA) of generalised expression, predominating in posterior regions (compatible with NCSE), in a patient with episodes of disorientation (A) and the subsequent disappearance of the EAs (B). We observed a posterior alpha background activity and anterior fast rhythms, accompanied by clinical improvement, after the administration of IV diazepam.
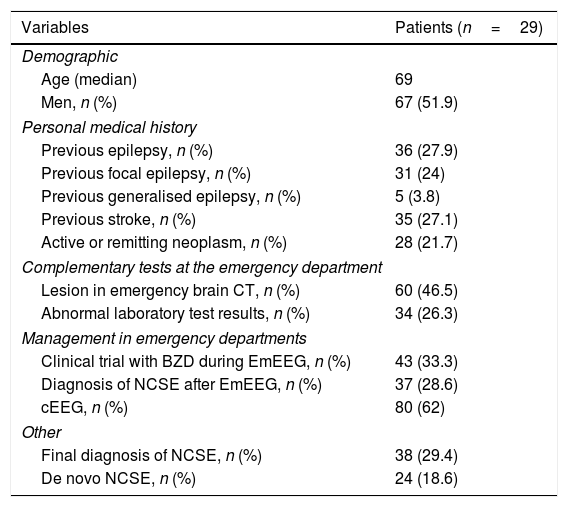
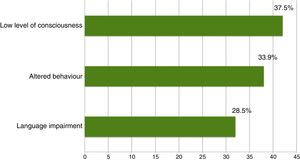
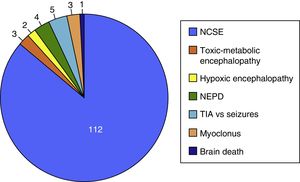
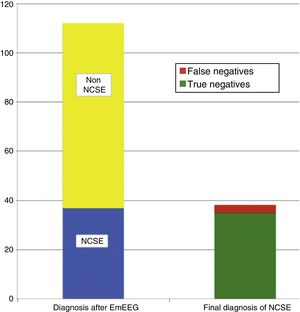
The on-call neurology team performed 135 EmEEG recordings in 129 patients. Table 1 includes the characteristics of the sample: 67 (51.9%) were men; median age was 69 years. In most cases (112, 83%), EmEEG was indicated to rule out NCSE. Fig. 4 shows the symptoms which led to EEG monitoring. Regarding the main indication, symptoms leading to suspicion of NCSE were altered level of consciousness in 42 cases (37.5%), abnormal and inappropriate behaviours in 38 (33.9%), and language impairment in 32 (28.5%). Fig. 5 shows the indications other than NCSE leading to the performance of the EmEEG (assessment of non-epileptic paroxysmal disorders, myoclonia, toxic-metabolic and hypoxic encephalopathies, differential diagnosis of stroke vs focal epileptic seizures, and diagnosis of brain death). Where NCSE was suspected, 37 recordings (33%) were considered compatible with NCSE, which was the final diagnosis at discharge in 35 (94.6%) of these patients. In contrast, there were 3 cases where NCSE was the final diagnosis after the follow-up cEEG in patients for whom the on-call neurology team did not consider this diagnosis after the EmEEG (Fig. 6). For the final diagnosis (whether NCSE or not), we referred to patient discharge reports, which include information on clinical progression and response to treatment. In some cases, diagnosis was also supported by follow-up cEEG recordings. In 72 patients (64%), NCSE was correctly ruled out. Taking as reference the final diagnosis at discharge (NCSE or other), the on-call neurology team's interpretation of the EmEEG during the study period has a sensitivity of 92.1%, a specificity of 97.2%, a positive predictive value of 94.6%, and a negative predictive value of 96% in the diagnosis of NCSE (Table 2).
Characteristics of patients who underwent EmEEG recordings.
| Variables | Patients (n=29) |
|---|---|
| Demographic | |
| Age (median) | 69 |
| Men, n (%) | 67 (51.9) |
| Personal medical history | |
| Previous epilepsy, n (%) | 36 (27.9) |
| Previous focal epilepsy, n (%) | 31 (24) |
| Previous generalised epilepsy, n (%) | 5 (3.8) |
| Previous stroke, n (%) | 35 (27.1) |
| Active or remitting neoplasm, n (%) | 28 (21.7) |
| Complementary tests at the emergency department | |
| Lesion in emergency brain CT, n (%) | 60 (46.5) |
| Abnormal laboratory test results, n (%) | 34 (26.3) |
| Management in emergency departments | |
| Clinical trial with BZD during EmEEG, n (%) | 43 (33.3) |
| Diagnosis of NCSE after EmEEG, n (%) | 37 (28.6) |
| cEEG, n (%) | 80 (62) |
| Other | |
| Final diagnosis of NCSE, n (%) | 38 (29.4) |
| De novo NCSE, n (%) | 24 (18.6) |
In daily practice, neurologists manage patients with altered levels of consciousness, acute affective disorders, unusual behaviour, and altered sensory perception or speech; in all these cases, the underlying condition may be NCSE.4–12 Several publications report an NCSE frequency of approximately 25% in patients at emergency units and neurocritical care units with altered levels of consciousness and behavioural problems.4,5,10–12 The complexity of NCSE due to its clinical and electrical heterogeneity, and its broad differential diagnosis,36–39 frequently lead to underdiagnosis of this complex disorder. Performing an EmEEG when NCSE is suspected is of great diagnostic use, facilitating treatment with AEDs, which is more effective when started early24,26,32,33 and contributes to a better prognosis by reducing the risk of cognitive sequelae.20,34 For several decades, researchers have attempted to implement EEG as a standard test in emergency and neurocritical care units.28,40 Recent, prestigious publications describe the situations in which EmEEG would be indicated: coma of unknown origin, focal neurological signs not explained by other factors, prolonged low level of consciousness following status epilepticus, and suspicion of psychogenic status epilepticus. However, having physicians and nursing staff dedicated exclusively to encephalography is infrequent, and approximately only 2% of emergency departments in the USA have EmEEG,28 probably because it is inefficient in terms of healthcare management. In relation to this, several studies have been published over the past few years describing devices that can take simple EEG recordings; these facilitate interpretation by medical staff not specialising in epilepsy, and have shown promise in the diagnosis of NCSE.29,42–45 Since July 2013, our hospital's on-call neurology team has included 2 residents in addition to the neurology consultant; all team members are on-site 24hours a day. The structure of the team enables EmEEG to be performed in cases where it is deemed necessary41; recordings are accompanied in some cases by a therapeutic trial of benzodiazepines, especially when NCSE is suspected. During the study period, more than one third of the patients who underwent an EmEEG were diagnosed with NCSE, and intensive treatment with AEDs was started early. In most of these cases, clinical progression and the results of a follow-up cEEG performed between 12 and 48hours after the EmEEG were consistent with the diagnosis at discharge of resolved NCSE. In contrast, the final diagnosis in 3 patients was NCSE, which went unnoticed despite the EmEEG performed by the on-call neurology team. However, of the cases where NCSE was initially suspected but ruled out by EmEEG (72, 64%), most were true negatives (69, 96%). In our experience, EmEEG as a diagnostic test may be useful to rule out NCSE in patients whose symptoms lead to an initial suspicion of NCSE. EmEEG allowed unnecessary treatments to be avoided in these patients, who might have received empirical treatment with AEDs in a classic clinical approach. These data show a sensitivity of 92.1% and a specificity of 97.2% which, in our opinion, would justify maintaining EmEEG as part of our clinical approach. However, certain limitations require us to interpret these data with caution. The variability of EEG patterns associated with NCSE make diagnosis difficult on occasions, even for experts in epilepsy46,47; therefore, the clinical context and the change in the recording after the administration of benzodiazepines are essential. An expert-assessed, follow-up cEEG was not always performed; therefore, there might be cases of false positives in which, in absence of NCSE, the diagnosis may have been the same at discharge due to favourable clinical progression in patients with benign symptoms of confusion or low level of consciousness which would have resolved spontaneously, regardless of AED treatment. We detected 3 false negatives where the results of the cEEG and the persistence of symptoms made us reconsider diagnosis of NCSE, despite initially having ruled it out. However, there may be more cases in critical care units whose broad-spectrum empirical treatment including AEDs may have masked a transient NCSE not diagnosed after EmEEG, which may have gone unnoticed in the diagnosis at discharge. The quality of 8-channel EEG recordings is not optimal, although there are reports in the literature with more limited recordings.43,44 Furthermore, since 2014, most of the EmEEGs performed at our hospital yielded similar recordings to the cEEG performed with electro-caps with up to 32 channels. However, we have found that although the limitations described demand caution in these conclusions, EmEEGs are useful in the everyday management of patients with suspected NCSE. These results would justify the constant availability of EmEEG as a diagnostic tool at our hospital, and invite further research, including a blind analysis of the EmEEG recording by epileptologists to obtain more accurate sensitivity and specificity data.
FundingThe authors have received no funding for this study.
Conflicts of interestNone.
Please cite this article as: Máñez Miró JU, Díaz de Terán FJ, Alonso Singer P, Aguilar-Amat Prior MJ. Uso de la electroencefalografía urgente por el neurólogo de guardia: utilidad en el diagnóstico del estatus epiléptico no convulsivo. Neurología. 2018;33:71–77.
This article was presented as an oral communication in epilepsy and during the outstanding communication session at the 67th Annual Meeting of the Spanish Society of Neurology. It was also presented in poster format at the 13th Annual Meeting of the Madrid Association of Neurology.