Radium-223 is an alpha-emitting radiopharmaceutical that significantly prolongs overall survival in patients with castration-resistant prostate cancer and symptomatic bone metastases. We report a retrospective analysis of our clinical experience with Radium-223 in the first 68 patients treated.
MethodsThe incidence of hematologic, gastrointestinal, and other adverse events was identified, including events that led to treatment discontinuation or delay. Alterations in bone pain and prostate-specific antigen and serum alkaline phosphatase levels were evaluated. Bone scan changes were identified and correlated with in clinical course.
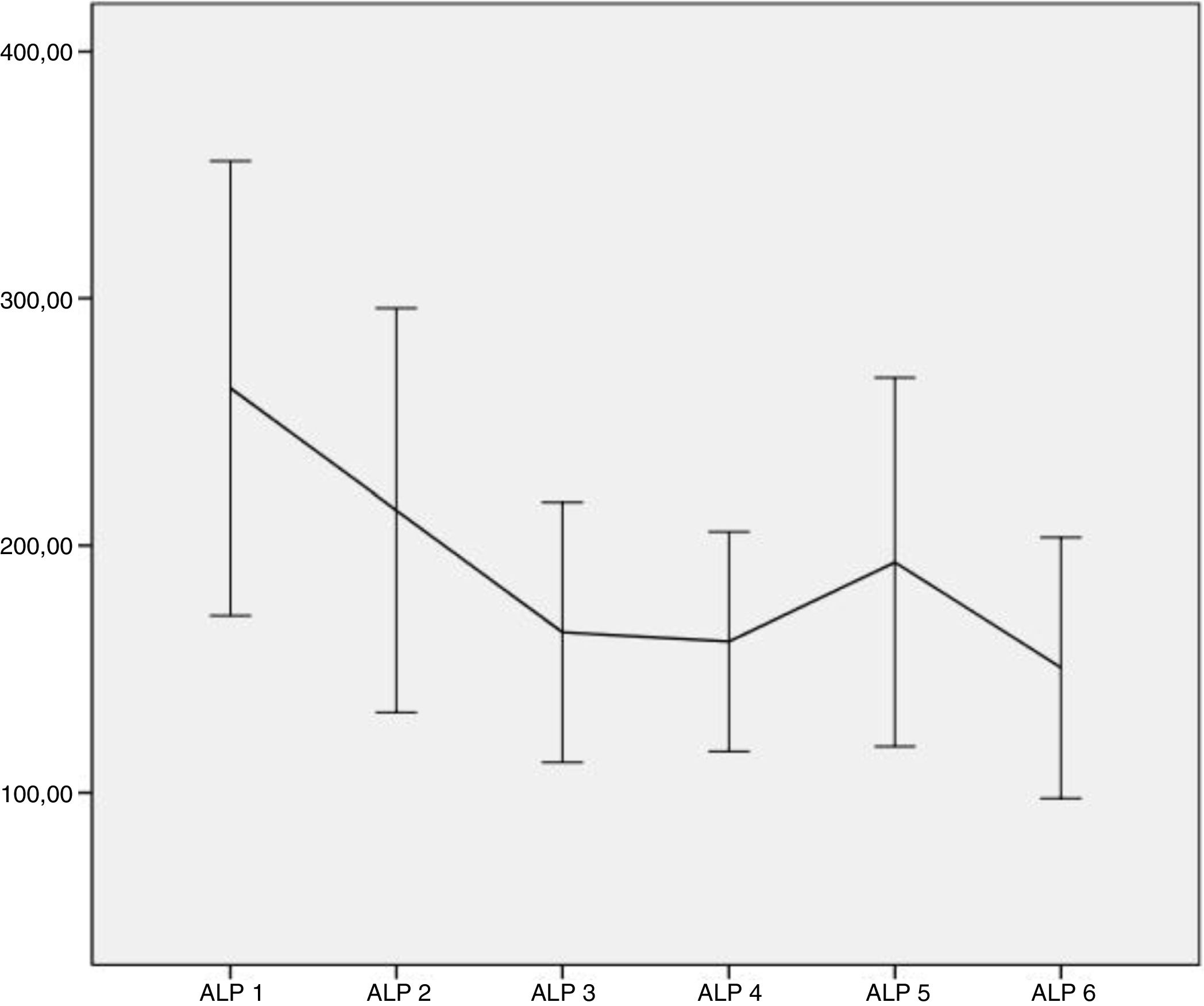
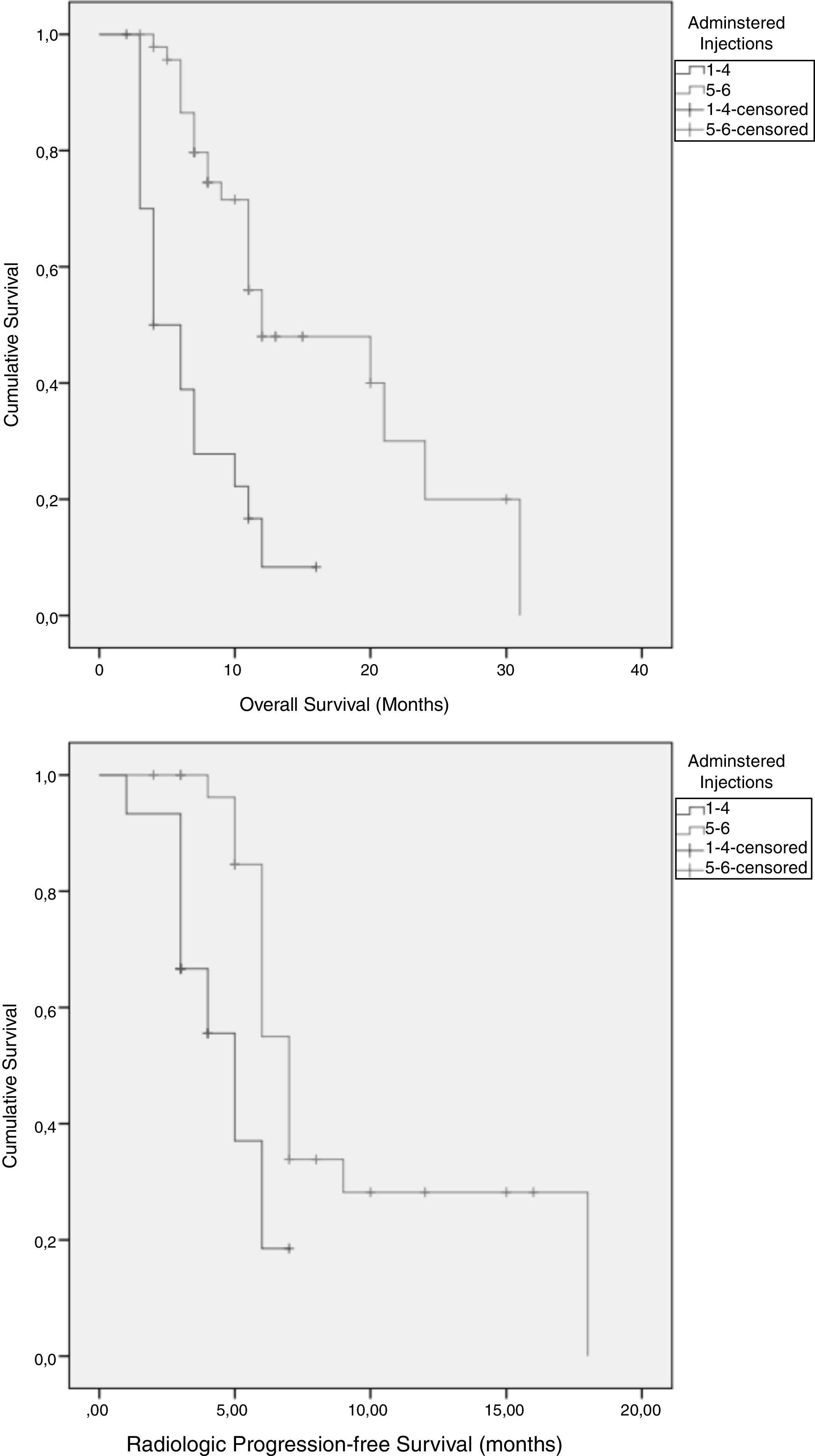
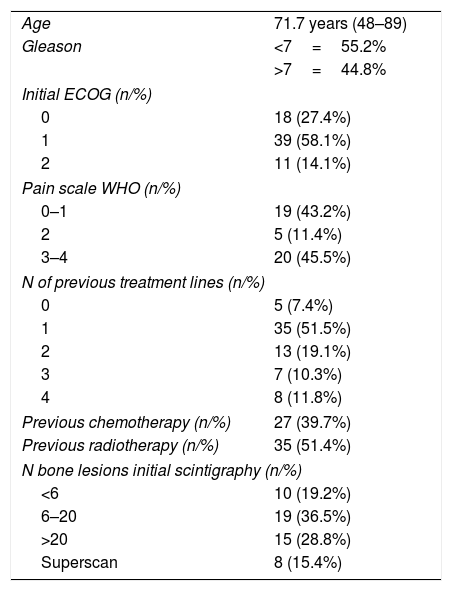
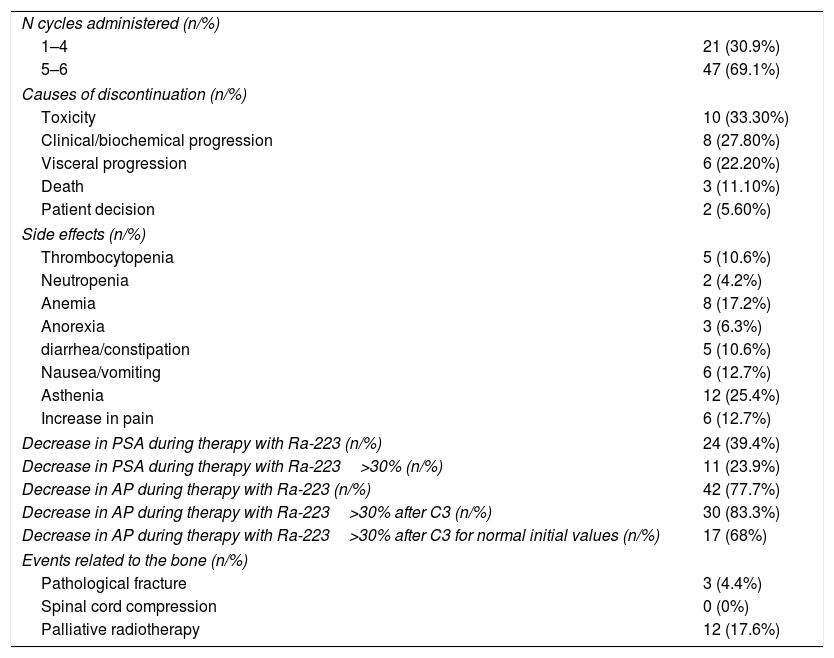
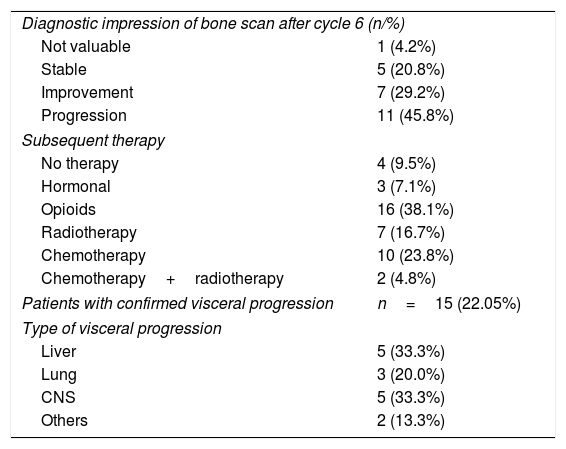
ResultsSixty-eight patients were included in the study. The median number of radium-223 injections was five (range 1–6), with 69% of patients receiving five to six injections. The most common side effects were digestive alterations in 24 patients, anemia in seven patients, and thrombocytopenia in five patients. Clear downward trends in serum alkaline phosphatase were seen, that were less clear in prostate-specific antigen. Mean serum alkaline phosphatase decreased from baseline in 77% of the patients, and prostatespecific antigen in less than 40%. The majority of patients (n=62) experienced an improvement in bone pain intensity or no increase in bone pain intensity. No PSA flare phenomenon was noted.
ConclusionsRadium-223 was generally well tolerated and there were no safety concerns. The adverseevents were mild and manageable. A decline in serum alkaline phosphatase was more frequent than a decline in the prostate-specific antigen. Monitoring changes in serum alkaline phosphatase dynamics may be useful.
El dicloruro de Ra223 es un radiofármaco emisor alfa que prolonga la supervivencia global en pacientes con cáncer de próstata resistente a la castración y metástasis óseas sintomáticas. Presentamos un análisis retrospectivo de 68 pacientes tratados.
MétodoSe evaluó la incidencia de eventos adversos hematológicos, gastrointestinales y otros, incluidos aquellos que llevaron a la interrupción o al retraso del tratamiento. Se determinó la mejora o empeoramiento del dolor óseo, los niveles de antígeno prostático específico y de fosfatasa alcalina sérica. Se valoró la gammagrafía ósea pre y postratamiento y se correlacionó con la evolución clínica.
Resultados68 pacientes fueron incluidos. La media del número de inyecciones de radio-223 fue de cinco (rango 1-6), el 69% de los pacientes recibieron cinco o seis inyecciones. Los efectos secundarios más comunes fueron alteraciones digestivas (24), anemia en 7 y trombocitopenia en 5. Se observaron claras tendencias a la baja en la fosfatasa alcalina sérica. La fosfatasa alcalina sérica media disminuyó desde el inicio en el 77% de los pacientes y el antígeno prostático específico en menos del 40%. La mayoría de los pacientes (62) experimentaron una mejoría en la intensidad o estabilidad del dolor. No se observó ningún fenómeno de llamarada de PSA.
ConclusionesEl radio-223 fue generalmente bien tolerado y no hubo problemas de seguridad. Los eventos adversos fueron leves y manejables. Una disminución en la fosfatasa alcalina sérica fue más común que del antígeno prostático específico. La monitorización en la dinámica de la fosfatasa alcalina sérica puede ser útil.
Article
If you experience access problems, you can contact the SEMNIM Technical Secretariat by email at secretaria.tecnica@semnim.es or by phone at +34 619 594 780.

Revista Española de Medicina Nuclear e Imagen Molecular (English Edition)