Pancreaticoduodenectomy (PD) is usually contraindicated in chronic liver disease. The objective of the present study was to analyze PD results in cirrhotic patients, and compare them with non-cirrhotic ones.
MethodsBetween 1994 and 2014 we prospectively collected all patients with a PD for periampullar neoplasms in Hospital Universitari de Bellvitge. We registered preoperative, intraoperative and postoperative variables. We defined patients undergoing PD with liver cirrhosis as the study group (CH group), and those without liver cirrhosis as the control group (NCH group). A case/control study was performed (1/2).
ResultsWe registered 15 patients in the CH group, all with good liver function (Child A), and included 30 patients in NCH group. The causes of hepatopathy were HCV (60%) and alcoholism (40%). For the 3 moments studied, the CH group had a lower blood platelet count and a higher prothrombin ratio, compared with NCH group. Postoperative morbidity was 60% and mean postoperative stay was 25±19 days, with no differences in terms of complications between CH group and NCG group (73% vs 53%, P=.1). Presence of ascites was higher in the CH group compared with NCH group (28 vs 0%, P<.001). There were no differences in terms of hemorrhage or pancreatic fístula. Four patients of the CH group and 2 patients of the NCH group were reoperated on (26.7 vs 6.7%, P=.1). There was no postoperative mortality.
ConclusionsPD is a safe procedure in cirrhotic patients with good liver function although it presents high morbidity.
La duodenopancreatectomía cefálica (DPC) es el tratamiento de elección de los tumores del área periampular. Esta intervención presenta una elevada morbilidad postoperatoria, y suele estar contraindicada en los pacientes con hepatopatía crónica (CH). Analizar los resultados de la DPC en pacientes cirróticos, y compararlos con los de pacientes no cirróticos.
MétodosEntre abril de 1994 y noviembre de 2014 registramos de forma prospectiva a todos los pacientes a los que se les realizó una DPC por cáncer del área periampular en el Hospital Universitari de Bellvitge. Se recogieron de forma prospectiva variables preoperatorias, intraoperatorias y del postoperatorio inmediato. Se definió grupo de estudio a los pacientes tratados mediante DPC y afectos de cirrosis hepática (grupo CH), y grupo control a pacientes intervenidos sin cirrosis hepática (grupo NCH); se realizó un estudio caso/control (1/2).
ResultadosRegistramos a 15 pacientes del grupo CH, todos ellos con una buena función hepática (Child A), y a 30 del grupo NCH. La causa de la hepatopatía fue VHC (60%) y enolismo (40%). En los 3 instantes estudiados, los pacientes del grupo CH presentaron una cifra de plaquetas en sangre inferior y una ratio de protrombina superior, respecto al grupo NCH. La morbilidad postoperatoria fue del 60%, con una estancia media de 25±19 días; sin diferencias significativas en la incidencia de complicaciones entre el grupo CH y NCH (73 vs. 53%; p=0,1). La presencia de ascitis durante el postoperatorio fue superior en el grupo CH respecto al NCH (28 vs. 0%; p<0,001). No hubo diferencias entre ambos grupos en la aparición de complicaciones hemorrágicas, ni de fístula pancreática. Se reintervino a 4 pacientes del grupo CH y a 2 del grupo NCH (26,7 vs. 6,7%; p=0,1). No hubo mortalidad postoperatoria.
ConclusionesLa DPC es una intervención segura entre los pacientes hepatópatas con buena función hepática preoperatoria, a pesar de comportar una elevada morbilidad.
The treatment of choice for tumors in the periampullary region is pancreaticoduodenectomy (PD). This surgical technique has a morbidity rate of 40%–60% and a postoperative mortality rate of about 5%, even in reference hospitals.1,2 It has been demonstrated that the results of this procedure are worse in patients with associated comorbidity, obesity,3 hypoalbuminemia or advanced age.4 Several studies have reported a higher number of postoperative complications after abdominal surgery in cirrhotic patients.5–8 To date, however, there are few published studies that analyze the results of oncologic surgery in patients with liver disease.5,9 At most hospitals, in fact, hepatic cirrhosis (HC) is a contraindication for PD. The objective of this study is to analyze the postoperative results of PD in cirrhotic patients and to compare these with results from non-cirrhotic patients.
MethodsFrom April 1994 to November 2014, we prospectively registered patients who had undergone PD for periampullary cancer at Bellvitge University Hospital, in Spain. We prospectively collected demographic variables, laboratory data and preoperative, intraoperative and immediate postoperative data. The extension studies of the patients affected by periampullary tumors, criteria for irresectability and surgical technique have been previously discussed.10 The HC group included patients diagnosed with HC prior to surgery and those who, at the time of surgery, had a cirrhotic-appearing liver. The diagnosis of HC was based on clinical-radiological or histologic results. Portal hypertension (PH) was defined as the presence of collateral circulation in the preoperative staging study. Patients affected by HC were evaluated individually, recording their functional hepatic reserve in accordance with the Child–Pugh classification11; only patients with good hepatic reserve (Child A) underwent surgery. The patients were administered desmopressin acetate (Minurin®) during the induction of anesthesia in cases of thrombocytopenia. The presence of HC involved no modifications to the surgical technique in terms of radical surgery. We were more meticulous about hemostasis of the surgical field and surgical ligatures were used more frequently than in non-cirrhotic patients. The following data were collected for each patient: alanine aminotransferase (IU), albumin, bilirubin, platelet count and prothrombin rate from the preoperative analysis and the first and seventh days post-op.
Postoperative MorbidityPostoperative morbidity was defined as any complication recorded during hospitalization. Postoperative mortality was that which occurred during hospitalization or within 90 days of surgery. Pancreatic fistula was defined as liquid discharge through the drain tubes that was rich in amylase after the 3rd day post-op, in accordance with the classification of the International Group for the Study of Pancreatic Fistulas (ISGPF).12 Postoperative ascites was defined as the discharge of ascitic fluid through the drains with a volume greater than 10mL/kg of weight/day after the 4th post-operative day.13 The presence of other complications was also recorded, as previously mentioned.10 All patients were monitored by a member of the surgical team after being discharged from the hospital.
Statistical AnalysisAfter having collected prospective data, a retrospective case-control (1/2) analysis was performed between the study group (HC group) and the non-HC (NHC) control group. Specifically, the study group was defined as patients who were treated with PD and were affected with hepatic cirrhosis (HC group); the control group included patients who had undergone PD due to periampullary cancer with no HC (NHC group). Patients of the control group were selected from the patients who had been treated immediately before and after each case of the study group; they were paired for age, sex and diagnosis in order to have the greatest possible similarity between the study groups. Furthermore, because in the study group (HC) no mortality was observed, we ruled out from the control group all those patients with postoperative mortality in order to increase their similarity. Afterwards, a descriptive analysis of the entire series was completed according to measurements of central tendency (mean, median) and dispersion (standard deviation and interquartile range). Then, we performed a comparative study between the qualitative variables using Chi-squared or Fisher's exact tests and the quantitative variables with the Mann–Whitney U. SPSS statistical software was used and a P<.05 was considered statistically significant in all cases. Lastly, the HC group was divided between patients with and without PH for their comparison.
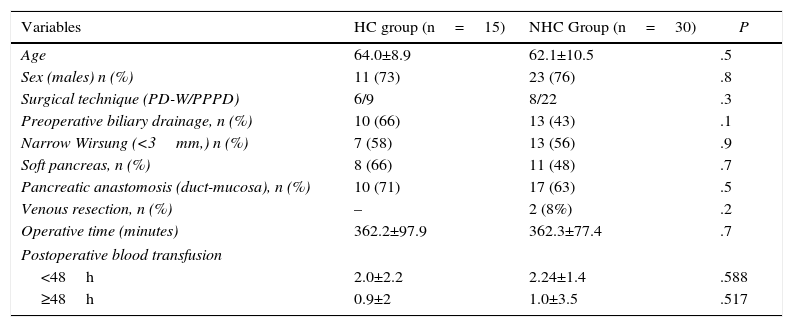
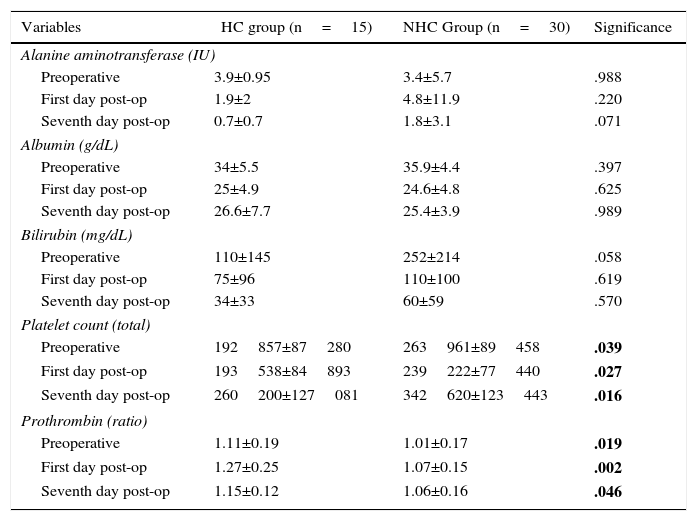
ResultsPatientsDuring the study period, 15 patients with HC (case group) were treated for periampullary cancer, and 30 patients were selected according to the criteria mentioned (control group, NHC). The overall study series (n=45) demonstrated that the majority of the patients were men (75%), with a mean age of 63±10 years. Pylorus-preserving PD (PPPD) was carried out in 69% of the patients, and PD with hemigastrectomy (Whipple procedure) (PD-W) was done in 31%. The diagnosis of HC was confirmed by hepatic biopsy in 9 patients, while in 6 the diagnosis was based on clinical radiological data. Liver function was satisfactory (Child A) in all patients. The causes of hepatopathy were HCV (60%) and alcoholism (40%). When we compared the 2 patient groups, they were comparable in terms of preoperative and intraoperative data (Table 1). We found no evidence of difference in mean operative time or postoperative transfusion between the 2 study groups. Last of all, in the HC group, we registered 8 patients with PH (73%) and 3 without PH (27%); the first 4 patients in the series were excluded due to lack of data. The liver function analysis demonstrated that the patients of the HC group presented lower levels of preoperative bilirubin than patients of the NHC group, although the differences were not significant (Table 2). In the 3 instances studied, the patients of the HC group presented lower blood platelet counts and higher prothrombin times compared to the NHC group.
Description and Comparison of Patient Groups According to Preoperative and Intraoperative Variables.
| Variables | HC group (n=15) | NHC Group (n=30) | P |
|---|---|---|---|
| Age | 64.0±8.9 | 62.1±10.5 | .5 |
| Sex (males) n (%) | 11 (73) | 23 (76) | .8 |
| Surgical technique (PD-W/PPPD) | 6/9 | 8/22 | .3 |
| Preoperative biliary drainage, n (%) | 10 (66) | 13 (43) | .1 |
| Narrow Wirsung (<3mm,) n (%) | 7 (58) | 13 (56) | .9 |
| Soft pancreas, n (%) | 8 (66) | 11 (48) | .7 |
| Pancreatic anastomosis (duct-mucosa), n (%) | 10 (71) | 17 (63) | .5 |
| Venous resection, n (%) | – | 2 (8%) | .2 |
| Operative time (minutes) | 362.2±97.9 | 362.3±77.4 | .7 |
| Postoperative blood transfusion | |||
| <48h | 2.0±2.2 | 2.24±1.4 | .588 |
| ≥48h | 0.9±2 | 1.0±3.5 | .517 |
PPPD: pylorus-preserving pancreaticoduodenectomy; PD-W: Whipple procedure.
Evolution of Perioperative Liver Function.
| Variables | HC group (n=15) | NHC Group (n=30) | Significance |
|---|---|---|---|
| Alanine aminotransferase (IU) | |||
| Preoperative | 3.9±0.95 | 3.4±5.7 | .988 |
| First day post-op | 1.9±2 | 4.8±11.9 | .220 |
| Seventh day post-op | 0.7±0.7 | 1.8±3.1 | .071 |
| Albumin (g/dL) | |||
| Preoperative | 34±5.5 | 35.9±4.4 | .397 |
| First day post-op | 25±4.9 | 24.6±4.8 | .625 |
| Seventh day post-op | 26.6±7.7 | 25.4±3.9 | .989 |
| Bilirubin (mg/dL) | |||
| Preoperative | 110±145 | 252±214 | .058 |
| First day post-op | 75±96 | 110±100 | .619 |
| Seventh day post-op | 34±33 | 60±59 | .570 |
| Platelet count (total) | |||
| Preoperative | 192857±87280 | 263961±89458 | .039 |
| First day post-op | 193538±84893 | 239222±77440 | .027 |
| Seventh day post-op | 260200±127081 | 342620±123443 | .016 |
| Prothrombin (ratio) | |||
| Preoperative | 1.11±0.19 | 1.01±0.17 | .019 |
| First day post-op | 1.27±0.25 | 1.07±0.15 | .002 |
| Seventh day post-op | 1.15±0.12 | 1.06±0.16 | .046 |
In bold: statistically significant result.
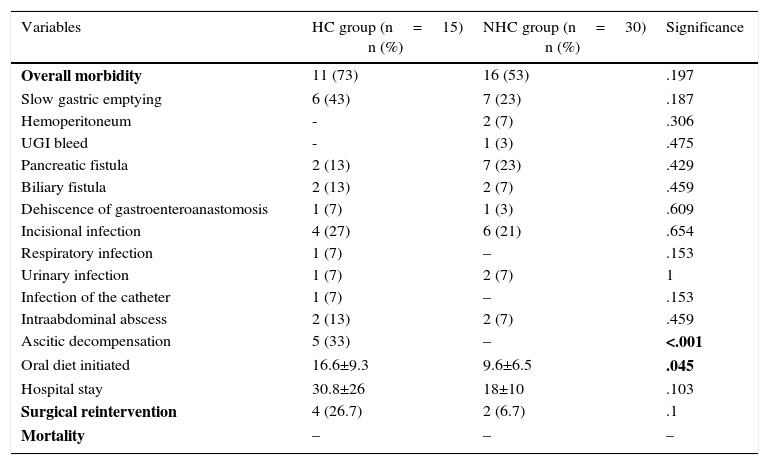
With regard to postoperative morbidity in the entire series, 60% of the patients had some type of complication. Mean postoperative hospital stay was 25±19 days and was longer in the HC group than in the NHC group (30 vs 18 days), with differences that were not statistically significant. When the 2 groups were compared, evidence of complications was seen in 11 patients of the HC group (73%) and in 16 (53%) of the NHC group (Table 3). As for hemorrhagic complications, none of the patients from the HC group presented hemoperitoneum, while 2 patients (7%) from the NHC group had hemoperitoneum. One patient from the NHC group presented UGI bleed (3%). Pancreatic fistulas were observed in 2 (13%) patients in the HC group and in 7 (23%) patients of the NHC group. Five patients of the HC group (33%) presented ascites during post-op. Two presented ascites refractory to diuretic treatment, so the placement of a peritoneal-venous shunt was indicated. A total of 6 patients were reoperated on, 4 (26.7%) from the HC group and 2 from the NHC group (6.7%), with differences that were not statistically significant. The causes of reoperation among the cirrhotic patients were intraabdominal abscess, evisceration, dehiscence of the duodenojejunal anastomosis and ischemic colitis, which required colectomy. The 2 non-cirrhotic patients were surgically treated for hemoperitoneum and coleperitoneum, respectively. There was no postoperative mortality in either of the 2 groups of patients studied. When the HC group was studied, the preoperative PH was not associated with a higher rate of postoperative complications than the group of cirrhotic patients without preoperative PH (62 vs 66%). We reoperated on 25% of the patients with PH (2/8), compared to 33% of patients without PH (1/3), which was not a statistically significant difference.
Postoperative Complications.
| Variables | HC group (n=15) n (%) | NHC group (n=30) n (%) | Significance |
|---|---|---|---|
| Overall morbidity | 11 (73) | 16 (53) | .197 |
| Slow gastric emptying | 6 (43) | 7 (23) | .187 |
| Hemoperitoneum | - | 2 (7) | .306 |
| UGI bleed | - | 1 (3) | .475 |
| Pancreatic fistula | 2 (13) | 7 (23) | .429 |
| Biliary fistula | 2 (13) | 2 (7) | .459 |
| Dehiscence of gastroenteroanastomosis | 1 (7) | 1 (3) | .609 |
| Incisional infection | 4 (27) | 6 (21) | .654 |
| Respiratory infection | 1 (7) | – | .153 |
| Urinary infection | 1 (7) | 2 (7) | 1 |
| Infection of the catheter | 1 (7) | – | .153 |
| Intraabdominal abscess | 2 (13) | 2 (7) | .459 |
| Ascitic decompensation | 5 (33) | – | <.001 |
| Oral diet initiated | 16.6±9.3 | 9.6±6.5 | .045 |
| Hospital stay | 30.8±26 | 18±10 | .103 |
| Surgical reintervention | 4 (26.7) | 2 (6.7) | .1 |
| Mortality | – | – | – |
In bold: statistically significant result.
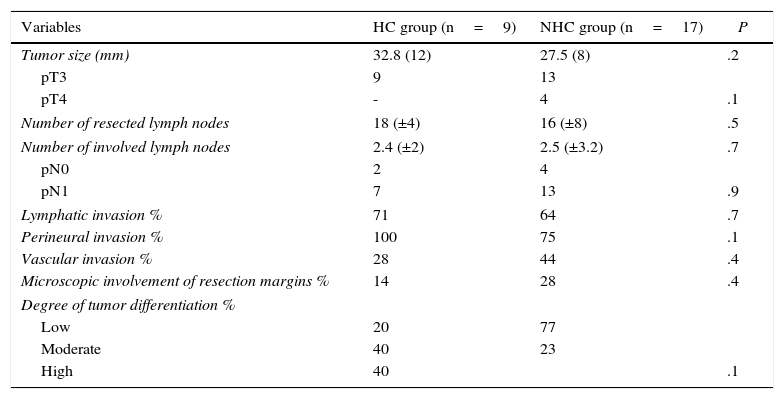
The distribution of diseases that were the cause for resection was similar. In the HC group, the pathologies were adenocarcinoma of the pancreas (60%), ampulla (33%) and common bile duct (7%). The NHC group pathologies were adenocarcinoma of the pancreas (63%), ampulla (23%), common bile duct (10%) and duodenum (3%). When we studied the group of patients with adenocarcinoma of the pancreas, we found no differences between the cirrhotic and non-cirrhotic patients in the pathology study. As for follow-up, 33% of the patients from the HC group and 33% of the NHC group were alive at the end of the study (Table 4). Mean actuarial survival of the patients with pancreatic adenocarcinoma was 28 months; meanwhile, it was 36 months in the HC group and 19 in the NHC group, with differences that were not statistically significant.
Pathology Study of Resected Pancreatic Adenocarcinomas.
| Variables | HC group (n=9) | NHC group (n=17) | P |
|---|---|---|---|
| Tumor size (mm) | 32.8 (12) | 27.5 (8) | .2 |
| pT3 | 9 | 13 | |
| pT4 | - | 4 | .1 |
| Number of resected lymph nodes | 18 (±4) | 16 (±8) | .5 |
| Number of involved lymph nodes | 2.4 (±2) | 2.5 (±3.2) | .7 |
| pN0 | 2 | 4 | |
| pN1 | 7 | 13 | .9 |
| Lymphatic invasion % | 71 | 64 | .7 |
| Perineural invasion % | 100 | 75 | .1 |
| Vascular invasion % | 28 | 44 | .4 |
| Microscopic involvement of resection margins % | 14 | 28 | .4 |
| Degree of tumor differentiation % | |||
| Low | 20 | 77 | |
| Moderate | 40 | 23 | |
| High | 40 | .1 | |
Gastrointestinal surgery in cirrhotic patients has classically been associated with high postoperative morbidity and mortality, depending on the surgical technique and hepatic function prior to surgery.5,14,15 Our study aims to analyze the viability of oncological pancreatic surgery in cirrhotic patients and postoperative complications.
At our hospital, patients with obstructive jaundice are admitted to the Gastrointestinal Surgery Department. This peculiarity is particularly beneficial in liver disease patients because, in our opinion, the preparation before surgery should be meticulous. In accordance with our criteria, we opted for preoperative biliary drainage in patients with albumin levels under 30g/dL or in those patients with uncompensated disease. We have only considered resection in Child A cirrhotic patients, and we have contraindicated Child B or C patients. If we review published series, several authors have demonstrated poor results after this surgery in patients with poor hepatic reserve. For instance, in 2011, Warnick et al.16 showed their experience in pancreatic surgery (PD and other pancreatic resections) by comparing 32 cirrhotic versus 32 non-cirrhotic patients. They included 2 Child B and 30 Child A patients, with a postoperative mortality of 100 and 3%, respectively. Two years later, El Nakeeb et al.11 published the longest series of PD in liver disease patients, comparing 67 cirrhotic versus 375 non-cirrhotic patients. These included 4 Child B and 63 Child A patients. Postoperative mortality was 50 and 9.5%, respectively. And, last of all, in 2015, Regimbeau et al.17 published their experience in 35 PD for pancreatic adenocarcinoma in cirrhotic patients within a multicenter study of 14 French work groups. The cirrhotic patients were compared with 70 non-cirrhotic patients. The authors included 11 Child B and 24 Child A cases, with a postoperative mortality of 55 and 4%, respectively. The morbidity of the Child B subgroup was 100%. As we have previously commented, the patients with Child B in our series were ruled out for surgery, and only those with Child A were treated surgically. Given the published results, pancreatic resection should be contraindicated in Child B cases.
When we compared the preoperative lab work, the cirrhotic patients had a higher prothrombin ratio and a lower platelet count than the non-cirrhotic group, which was probably related to the hepatopathy and PH. Furthermore, albumin levels were similar in both groups, and the bilirubin in blood was lower among the cirrhotic patients. These data reveal that the cirrhotic patients were treated in a correct nutritional situation, with satisfactory hepatic function. Our findings are similar to those published by El Nakeeb,11 who found no differences in bilirubinemia or in preoperative transaminase levels between the two groups, in spite of registering lower preoperative albumin in the cirrhotic patients, which is probably related with the indication for surgery in Child B patients.
Surgery in cirrhotic patients creates more stress for surgeons due to the greater risk of bleeding in the surgical field. Meticulous hemostasis is always essential in these surgeries and should be even more painstaking in this group of patients. In the previously mentioned French multi-center study,17 no differences were found in operative time or intraoperative blood loss, which is similar to our findings. Contrarily, other authors demonstrated greater blood loss in cirrhotic patients,11 so they recommend the prophylactic use of vitamin K or the intraoperative transfusion of fresh plasma in extreme situations. In accordance with our criteria, some authors recommend achieving hemostasis by means of surgical ligatures.11
The Egyptian group added an analysis to determine the importance of PH in this type of surgery.11 They compared 16 patients with PH to 51 without PH. According to their results, this would involve greater blood loss (300mL vs 100mL) and a higher consumption of blood products. In addition, they showed evidence of higher postoperative mortality, although this difference was not statistically significant (25 vs 7.8%). In the study by the group at Kings College Hospital in London, Sethi18 demonstrated satisfactory results in 4 patients with hepatic cirrhosis and PH treated with PD. They even mention a case with preoperative and intraoperative portal pressure of 23 and 17mmHg, respectively. Three groups publish their results with the use of preoperative transjugular intrahepatic portosystemic shunt (TIPPS) to palliate the effects of PH in abdominal surgery. Nonetheless, these are very short series and the placement of TIPPS does not seem to provide clear benefits,19 while it seems to result in a higher rate of hepatic encephalopathy or cardiac insufficiency.20,21 In our series, 8 patients presented PH in the preoperative imaging study, with results similar to the patients without PH.
When we reviewed the results of the 15 cirrhotic patients treated surgically, we observed postoperative ascites in 5 (33%). This was also reported by El Nakeeb,11 although to a lesser degree (10%). As for other postoperative hepatic function data, we did not find evidence of deterioration in the lab results among the cirrhotic patients. We saw similar differences to those found preoperatively, with a lower platelet count and a more prolonged prothrombin time among cirrhotics. The postoperative hepatic failure recorded by other authors was 6%–14%.11,15 In our experience, we have seen no such cases, which was probably related with the preoperative selection.
When we compared both groups, we found evidence of a higher rate of postoperative complications in the HC group versus the NHC group (73 vs 53%), although the difference was not statistically significant. Other authors report postoperative complication rates between 46 and 86%.11,16,18 There were 6 reoperations: 4 in the HC group and 2 in the NHC group. This resulted in a slightly longer mean postoperative hospital stay in the HC group compared to the NHC group (30 vs 18 days), although the differences were not significant. The El Nakeeb11 study reported a longer mean postoperative hospital stay in the HC group than in the NHC group.
In the 3 longest series published to date about pancreatic surgery in cirrhotic patients, postoperative mortality was 9%–17%.11,16,17 When comparing PD in cirrhotic and non-cirrhotics, Regimbeau17 found significant differences in postoperative mortality (17 vs 5%; P=.04), and El Nakeeb11 demonstrated higher postoperative mortality in the group of cirrhotic patients (11.9 vs 1.6%; P<.0001). The study by Warnick16 showed non-significant differences between both groups in terms of postoperative mortality (9 vs 0%); however, as we have commented, said study includes patients treated with other types of pancreatic resections, such as duodenum-preserving pancreatic head resection, or distal pancreatectomies. In our study, postoperative mortality in the study group was zero. Proper preoperative patient selection, which excluded Child B patients, contributed to favorable postoperative progress.
As for adenocarcinoma of the pancreas, the publication of the French group17 demonstrated long-term results that were similar in the Child A group and in the control group for both recurrence and survival rates. In our experience, there were also no long-term differences between the HC and NHC groups, and there even seemed to be slightly higher survival in the HC group, although without statistically significant difference (32 vs 15 months; P=.06). The analysis of the surgical specimens in the French group revealed fewer resected lymph nodes among the cirrhotic patients (14 vs 20). In our experience, neither the resected lymphadenopathies nor the tumor size were different between the two groups.
PD is a feasible surgical intervention in cirrhotic patients, even though it involves elevated postoperative morbidity. The selection of patients with optimal hepatic function is the key for obtaining positive results.
Conflict of InterestsThe authors have no conflict of interests to declare.
Please cite this article as: Busquets J, Peláez N, Gil M, Secanella L, Ramos E, Lladó L, et al. ¿Es la duodenopancreatectomía cefálica una intervención segura en el paciente cirrótico? Cir Esp. 2016;94:385–391.