In spite of the technological advances in endovascular treatments, median arcuate ligament syndrome (MALS) requires a surgical approach to eliminate the abnormal anatomical structures that cause extrinsic compression to the coeliac artery (CA). If the endovascular treatment is not accompanied by adequate release of the compressive muscular and ligament structures, the symptoms usually recur early on. We present the case of a male patient with this syndrome who presented occlusion of the CA after simple angioplasty requiring decompression surgery and aortocoeliac bypass, which provided satisfactory results (Figs. 1 and 2).
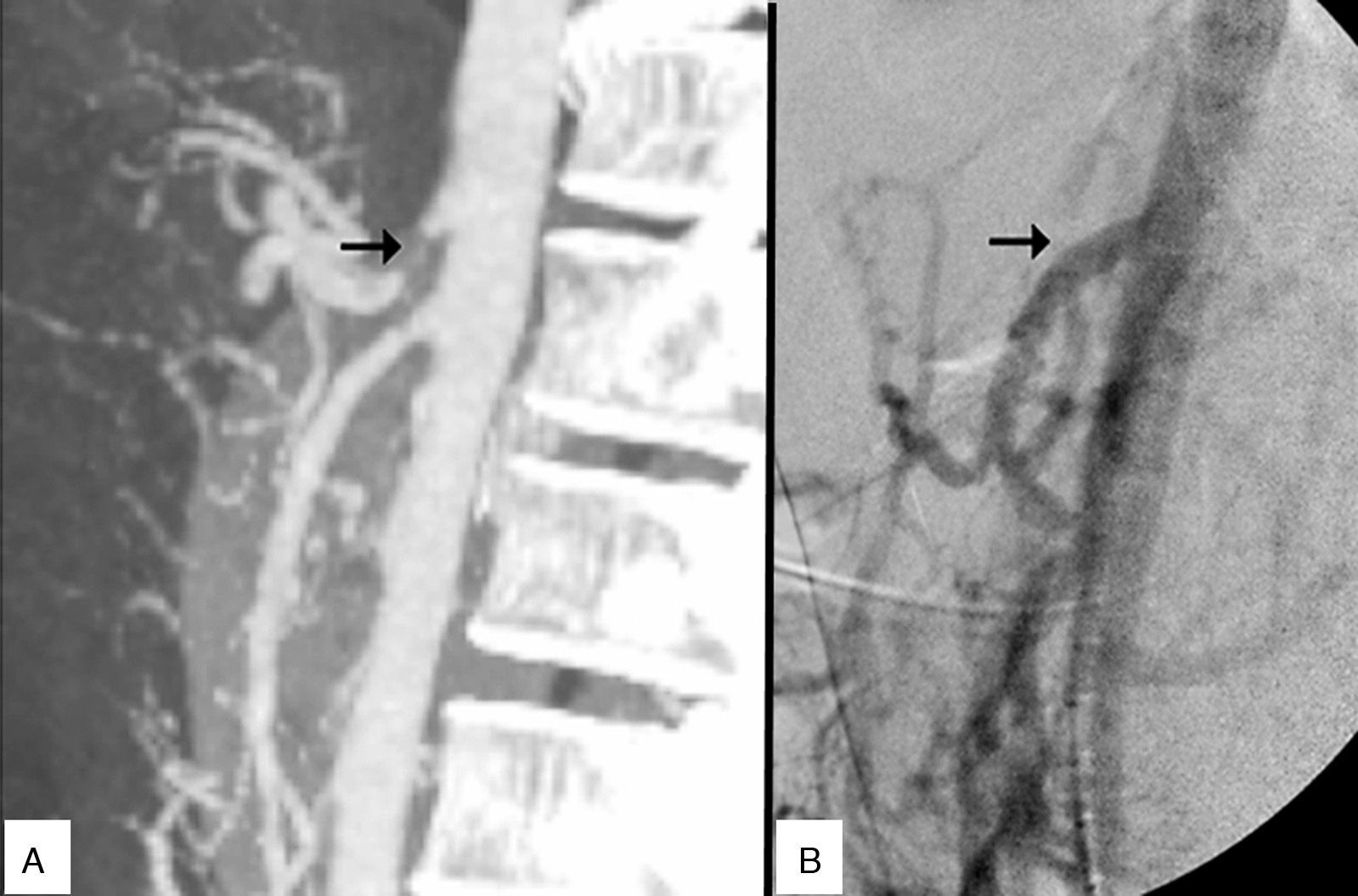
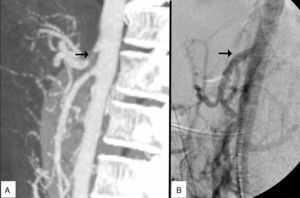
The patient is a 27-year-old man, a carpenter by profession, with a history of intense tobacco habit and type I diabetes mellitus, with poor metabolic control. Over the previous 3 years, he had experienced postprandial epigastric pain and vomiting, leading to a weight loss of 50kg, with no definitive diagnosis. Finally, a study done by the Digestive Tract Department at another hospital provided the diagnosis of MALS. Doppler ultrasound showed turbulent flows with a peak systolic velocity of 440cm/s in the CA. In addition, gadolinium-enhanced magnetic resonance angiography of the visceral aorta confirmed stenosis of the CA of more than 70%.
In the interventional radiology unit at that same hospital, the patient underwent percutaneous transluminal angioplasty of the CA with a balloon catheter measuring 5mm in diameter by 20mm in length, which provided good immediate results and resolution of the symptoms. Nonetheless, the patient quickly presented the same symptoms once again.
Given the unfavourable progress, with frequent visits to the emergency department for intravenous medication for pain control, the patient was referred to the Angiology and Vascular Surgery Department at our hospital. Upon examination, the patient was moderately thin, pulses were symmetric, ankle-brachial indices were normal, and no abdominal bruit was detected. Doppler ultrasound and CT angiography confirmed CA occlusion.
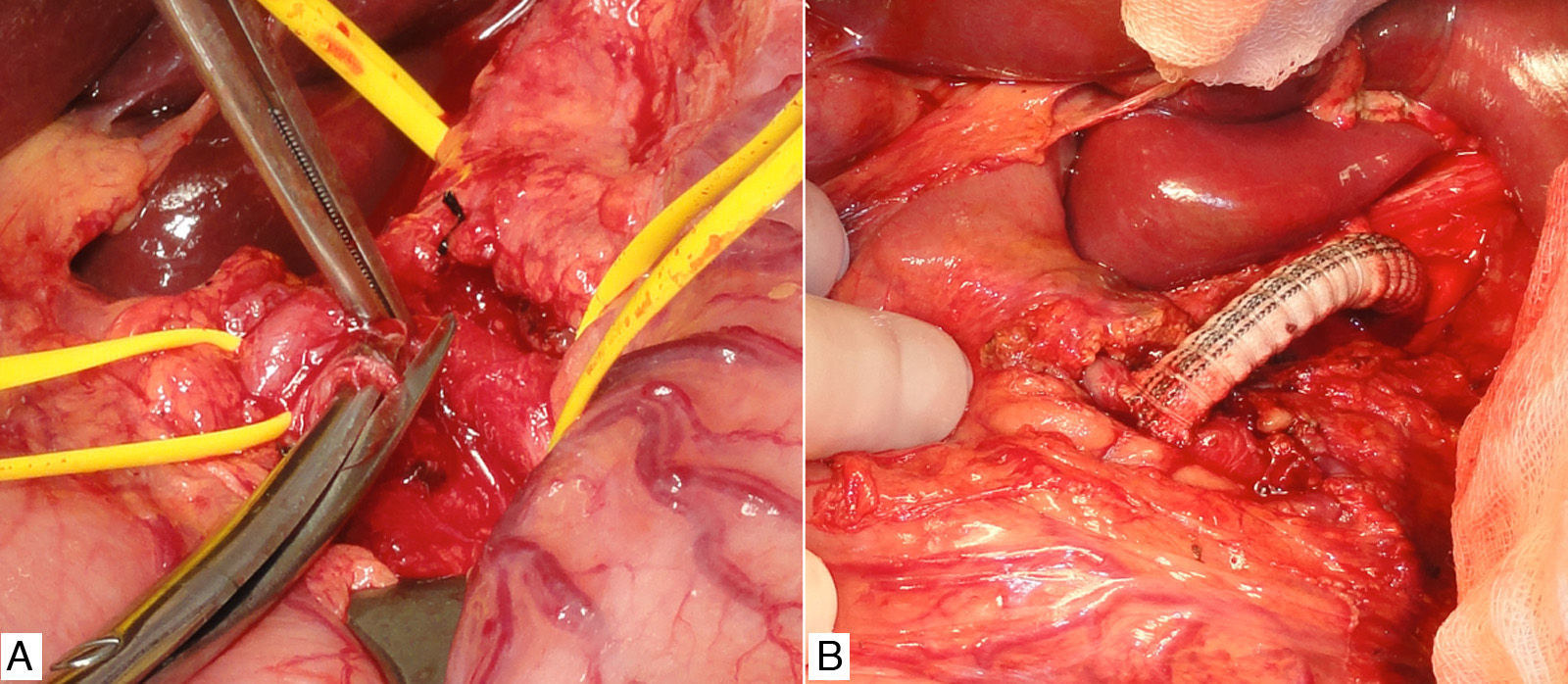
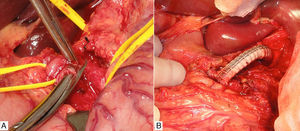
We decided to schedule open surgery, which involved supraumbilical abdominal midline incision and periarterial decompression by means of adhesiolysis of the CA and its 3 branches. In addition, we divided the surrounding crura of the diaphragm and a multitude of neighbouring fibrous adhesions.
Prior to the surgical approach of the CA, intraoperative arteriography demonstrated the obstruction. The revascularisation consisted of an anterograde bypass from the supracoeliac aorta up to the origin of the CA branches with a 7mm Dacron® gelatine-impregnated stent (Grupo Cardiva). After creating the bypass, the intraoperative arteriogram was satisfactory.
The patient was discharged on the third day with no further incidents, and he continued to be asymptomatic at the successive follow-up visits.
The median arcuate ligament connects the crura of the diaphragm to both sides of the aortic hiatus and is situated above the CA.1 An abnormally low insertion of the diaphragm and/or an excessively high origin of the CA can cause median arcuate ligament syndrome. It is more prevalent in women, and its incidence is around 2 out of every 100,000 patients with recurring abdominal pain.2
Radiological studies show that some 50% of asymptomatic persons can have varying degrees of extrinsic pressure on the CA during forced expiration, but this does not mean that they should be diagnosed with MALS. In patients with this syndrome, compression is exerted both during inspiration as well as expiration.3
Chronic compression of the CA causes symptoms that vary greatly. The most common is the existence of chronic abdominal pain and weight loss due to chronic intestinal ischaemia. The most uncommon is the development of splanchnic artery aneurysm with possible rupture.4,5 There have also been rare reports of retrosternal pain associated with physical exercise.6
The treatment of choice for MALS involves surgical decompression, which can be open or laparoscopic,7 to eliminate the fibres that encompass or compress the CA. If there are associated intrinsic lesions of the CA with stenosis or obstruction, it is necessary to associate endovascular treatment using stents or aortocoeliac bypass. Endovascular therapy, without associated surgical decompression, is not recommended due to the high risk for early restenosis, similar to other extrinsic arterial compressions.2
It is important to consider MALS as a possible diagnosis in patients with chronic recurring abdominal pain and vomiting, especially in young patients and when other diseases have been excluded.8 If arteriography of the abdominal aorta were necessary for confirmation in the diagnostic phase, we must avoid the temptation to perform balloon angioplasty on the stenotic lesion since, without associated surgical decompression, early recurrence is very likely and symptoms may even become worse.
Please cite this article as: Novo Martínez GM, Rodríguez Morata A, Alonso Argüeso G, Reyes Ortega JP, Gómez Medialdea R. Dolor abdominal crónico recurrente, a consecuencia del síndrome del ligamento arcuato medio. Cir Esp. 2016;94:301–303.