Secretory breast carcinoma was first described by McDivitt and Stewart in 1966, who reported seven cases of this tumoural strain in children and therefore named it “juvenile carcinoma”. However, a couple of decades later, Tavassoli and Norris reported a series of 19 cases and found that most occurred in adults, and therefore they proposed changing its name to “secretory carcinoma.” Since then, approximately 100 cases have been reported in the world literature. We present two cases treated with total mastectomy and sentinel node biopsy with excellent outcome. To our knowledge, only a few cases have been reported in Latin American patients.
El carcinoma secretor dela mama fue inicialmente descrito por McDivitt y Stewartin en 1966, en su artículo, siete casos de este tipo de tumor se presentaron en niños, por lo tanto, se le asignó el nombre de “carcinoma Juvenil”, pero un par de décadas después Tavassoli y Norris encontraron en su serie de 19 casos, que la mayoría eran adultos, y así propusieron cambiar el nombre a Carcinoma secretor. Desde ese momento aproximadamente 100 casos han sido reportados en la literatura. Nosotros presentamos dos casos tratados con mastectomía y ganglio centinela, con excelente pronóstico. Hasta donde sabemos, esta patología se ha reportado escasamente en pacientes latino americanos.
Secretory breast carcinoma is an extremely rare condition/disease characterized by intra- or extracellular discharge and eosinophilic granules in the cytoplasm of neoplastic cells. It presents with marked immunohistochemical and cytogenetic differences to the common ductal carcinoma.
In a retrospective analysis including more than 7000 infiltrating carcinomas in 1994, Lamovec and Bracko, reported only 4 cases of secretory carcinoma. Li et al. in 2012 found only 15 cases out of a sample of 10,000 breast carcinomas.1 We present two cases treated with total mastectomy plus sentinel node biopsy, and as far as we know with excellent prognosis. Our recommendation is that this procedure should be individualized, specially for this rare type of breast cancer.
Case 1Our first case is a 61-year-old female patient, with no relevant family history. Under treatment for diabetes and hypertension, history of surgery in the upper inner quadrant of the right breast in 2012, with no pathology report, with an apparent volume increase of the operated region. Clinically with 50mm×42mm tumor, no axillary nodes. Mammography and ultrasound reported a 45mm tumor in the right breast, classified as BIRADS 5 suggestive of malignancy. Patient was submitted to core needle biopsy which reported ductal carcinoma, grade 2, with no specific pattern. Estrogen receptor 40%, progesterone receptor 0%, Her 2 neu negative (0). Ki-67 5%.
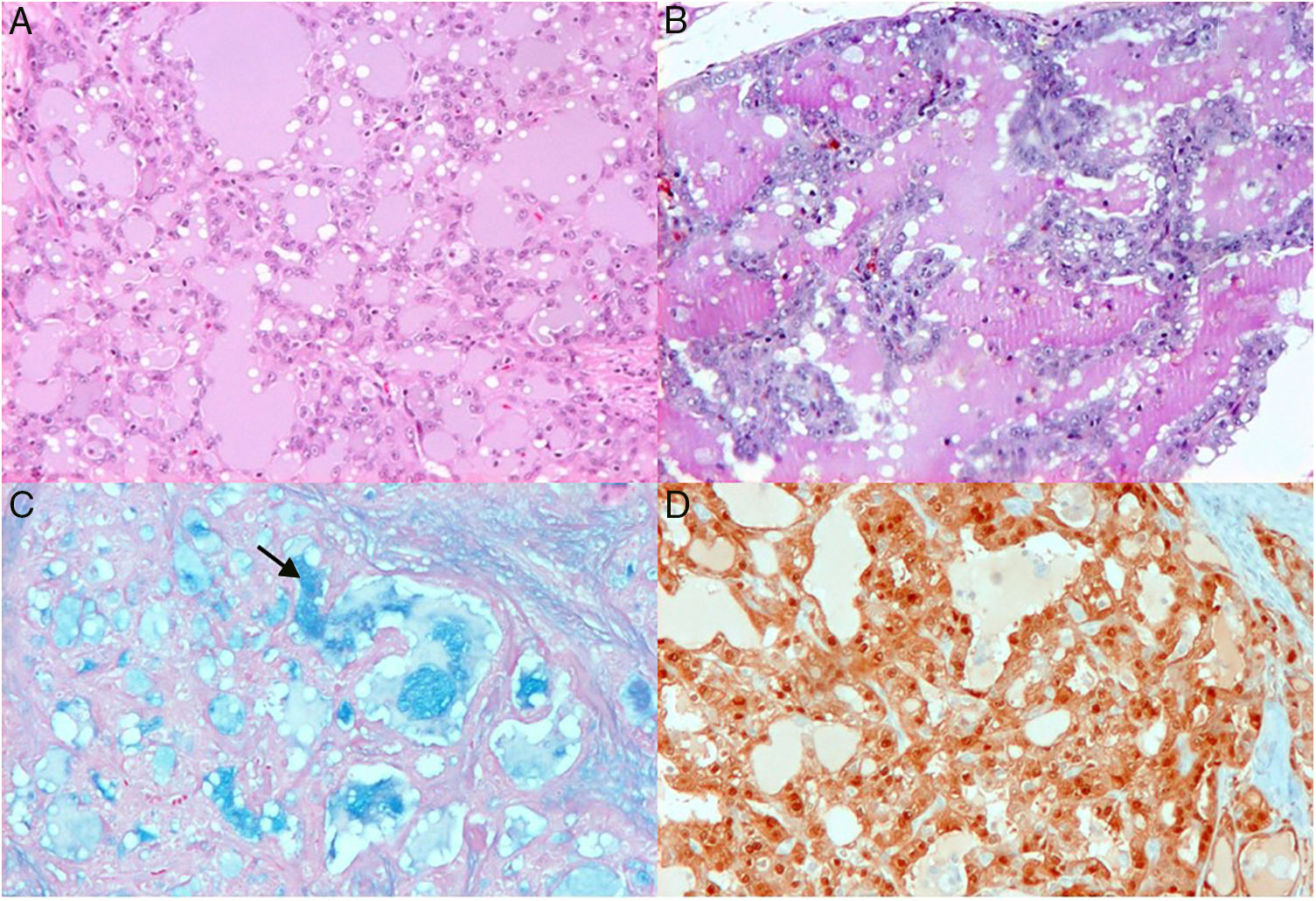
Patient was programmed for total mastectomy and sentinel node biopsy. The definitive pathology report revealed a secretory carcinoma, S-100 positive protein in the nucleus and cytoplasm of neoplastic cells (Fig. 1A–D). A tumor size of 46mm×43mm, 3mm away from the deep border, and four sentinel nodes with mixed hyperplasia (follicular and interfollicular), negative for malignancy. Pathological stage IIA by pT2 pN0. Due to comorbidities, and a good prognosis of the histological type, the patient is considered for hormone therapy and close surveillance. As far as we know, the patient continues asymptomatic and with no recurrences.
Upper pictures show histologic section treated with HE technique, notice microcysts covered by neoplastic cell (A). Dilated glands with abundant osinophilic material in the lumen (B). This is more evident colloidal iron staining (C). S-100 protein is expressed in nucleus and cytoplasm of neoplastic cells (D).
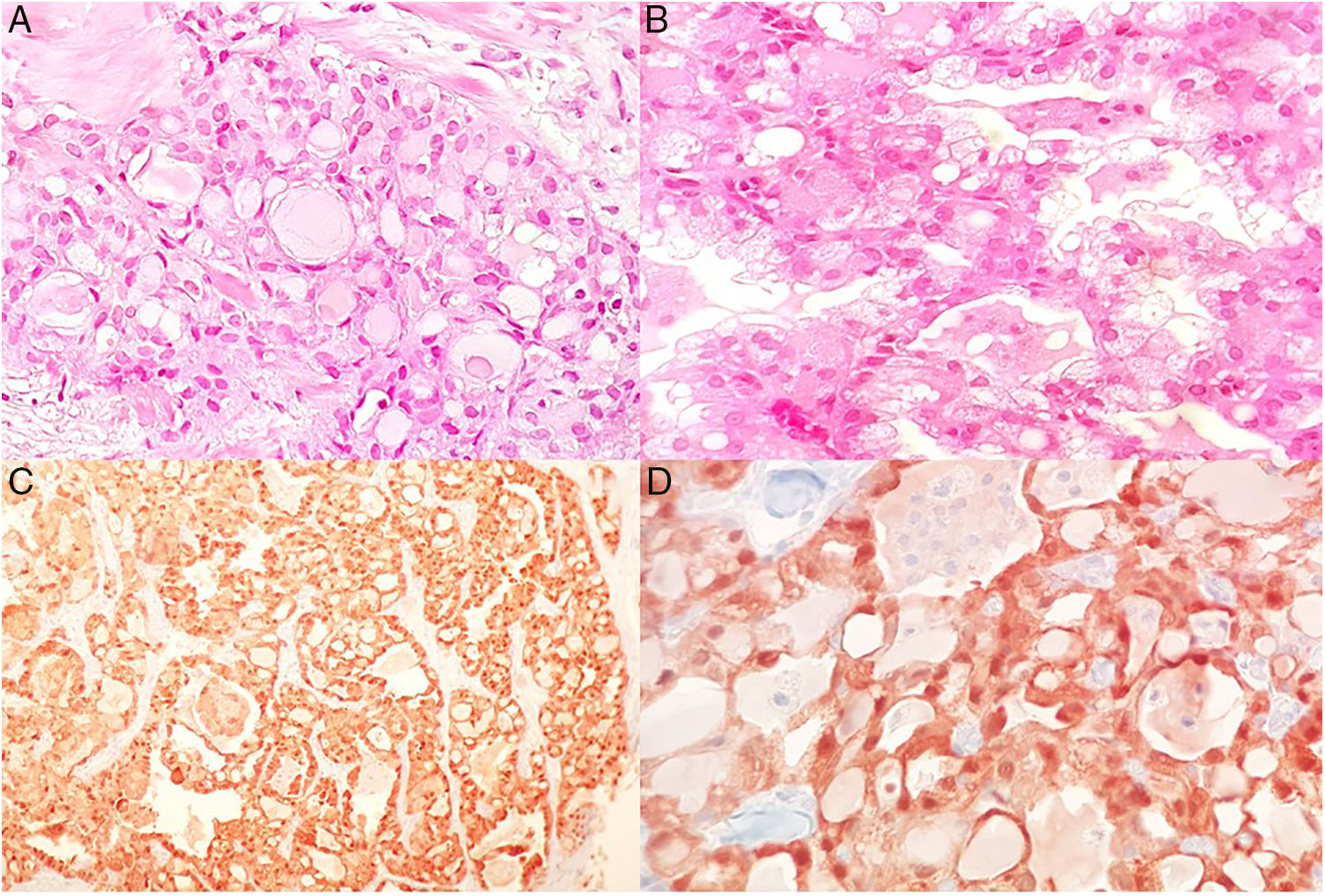
Female, 77 years old, with no relevant family history. Under treatment for diabetes for the last 20 years, multiparous and with breastfeeding history. She presents with self-detected tumor in left breast, painless and without apparent growth, 5 months before medical appointment. Imaging studies categorized BI-RADS 4B for the presence of dense nodule, lobed edges, in the upper outer quadrant. Ultrasound reported a solid, hypoechoic, round nodule, with angled posterior edges, small echogenic halo, circumscribed, parallel to the skin, with an abrupt interface, a slight lateral shadow, peripheral vascularity detected by Doppler ultrasound, elastography reports greater hardness than fat tissue (13kp), measuring 10mm×8.4mm×9.6mm. Physical examination with tumor in the external superior quadrant, 21mm×1mm, clinically with no axillary nodes found. Core Needle biopsy was performed, which reported infiltrating secretory carcinoma. Epithelial cell forming glands, some with cystic dilatation, lumen completely occupied by dense eosinophilic secretion, oval nuclei with moderate pleomorphism and a mitosis in 10 high power fields. Immunohistochemistry: S-100 positive protein in the nucleus and cytoplasm of neoplastic cells (Fig. 2A–D). Estrogen receptors: 0%, progesterone receptors: 0%, Her 2 neu negative (0), Ki-67: <5%. Stage IIA by cT2N0M0T. Patient was considered candidate for total mastectomy+lymphatic mapping+sentinel node biopsy. The surgery was performed without complications, the histopathological definitive report was secretory invasive carcinoma grade 2 of 1.9cm greater diameter, one sentinel node negative for metastasis and no lymphovascular affection.
DiscussionSecretory breast carcinoma was first described by McDivitt and Stewart in 1966.1 The definition given by the World Health Organization is: clear-cell tumor showing prominent secretory activity such as that observed in pregnancy and lactation, whose secreted material is usually positive for Schiff staining test and is present in the cytoplasm and in acinus-like spacesi2. McDivitt and Stewart, described in their article seven cases of this tumoral strain in children, therefore, they assigned the term “juvenile carcinoma”, thus being the most common mammary carcinoma in children.3 In 1980, Tavassoli and Norris found in their series of 19 cases that the majority were adults, and therefore, they proposed to change the name to “secretory carcinoma.” Since then, approximately 100 cases have been reported in the world literature.2
The age of presentation documented ranges from 3 to 83 years3 with a mean of 33 and a median of 40.1 Most cases in the literature are reported in young women,3 in contrast to our two reported cases, which are in the seventh and eighth decades of life. There is a predilection for the female gender in a ratio of 1: 6, although when affecting men, it tends to have a more aggressive course.1 The most common clinical presentation, like any other breast tumor, is usually self-detecting, painless, slow-growing rate, well circumscribed and movable breast nodule. The most frequent localization is in the upper outer quadrant, although it has been found in any breast quadrant,1,3 even in axillary ectopic mammary tissue. It is typically unifocal, although multifocality has also been reported in literature.4 In men and in pre-pubertal patients, the tumor is usually retroareolar and associated with telorrhea, given the small amount of glandular tissue.3 The average tumor size is 3cm and up to 15% of the cases there is lymph node involvement at the time of diagnosis.1 When this happens, more than 3 nodes affection is rare.3
Radiologically, secretory carcinoma, unlike infiltrating carcinoma with no specific pattern, usually presents as a well-circumscribed, rounded or oval mass with microlobulated borders,1,3 and may even mimic benign pathology such as fibroadenoma5 or other malignant disease such as medullary, papillary or mucinous carcinoma.6 For this reason, radiologists usually give a BI-RADS category 4 to this type of lesions. Also, as the age of presentation is usually in relatively young patients, the benefits of mammography may be limited, making breast ultrasound the most useful diagnostic tool. However, when non-palpable secretory carcinomas are treated, they can be documented as asymmetries or even suspicious micro-calcifications.3
Microscopically, it is usually arranged in microcystic, ductal or solid pattern, tumor cells are usually polygonal, with a pale pink vacuolated cytoplasm, with small rounded nuclei, minimal atypia and low mitotic activity. In the extra-cellular spaces and the intra-cytoplasmic vacuoles, an eosinophilic, diastase resistant and Schiff periodic acid positive secretion is present. Sometimes an intra-ductal component can be found, most frequently with cribriform and papillary architecture.2,3 Ring-seal cells have also been reported occasionaly.7,8 At the time of immunohistochemistry, the absence of myo-epithelial cells by p63 staining and/or smooth muscle actin will be documented, as well as the absence of expression of estrogen, progesterone and Her2neu receptors, that is, triple negative subtype.1 Even so, and as in one of our reported cases, there may be expression of hormone receptors. It is well known that this triple negative molecular subtype in infiltrating carcinoma without a specific pattern is associated with a worse prognosis; however, in the secretory carcinoma, even with this immunophenotype, the prognosis is usually highly favorable.3,4 It has also been found that secretory carcinomas are almost always immunopositive to S-100, alpha-lactalbumin and e-cadherin, like any type of infiltrating ductal carcinoma.9 Tognon et al., in 2001, demonstrated a balanced chromosomal translocation t(12; 15) resulting in fusion of the ETV6 and NTRK3 genes.10 Although this translocation is not always present, the fused gene ETV6-NTRK3 is specific for secretory carcinoma, since this gene has not been found in any other type of infiltrating carcinoma of the breast.3
As an extremely rare carcinoma, there is currently no consensus that defines the best treatment option.1,3,11 Although the recommendations vary according to each author, the basis of treatment is undoubtedly surgery.1–4,11 Although the presence of axillary lymph node metastases is usually low, especially in tumors smaller than 2cm, it has been documented, even in tumors smaller than 1.5cm,11 and the presence of 3 or more axillary nodes with metastases is a factor of poor prognosis.11 Reséndiz-Morán and Lazos-Ochoa, in 2005, published a case of decease due to a metastatic secretory carcinoma to bone and to multiple viscera.2 We consider that the loco-regional treatment of the tumor is the surgical treatment, it does not require any other treatment different from other infiltrating breast carcinoma. If the axilla is clinically negative, sentinel node biopsy is considered (cN0), and if the result is positive for metastasis, axillary radical lymphadenectomy should be performed.12 If clinically is node positive or confirmed by cytology or histology (cN1 or cN1 [f]), initial axillary radical lymphadenectomy should be performed for adequate axillary staging. In special cases, like children, local excision with free borders and selective sentinel node biopsy is usually the preferred treatment, trying to preserve the largest pre-pubertal tissue for later development. Although local recurrence does not appear to affect the favorable clinical course of secretory carcinoma, adjuvant radiotherapy may be indicated when conservative surgery is performed3; however, its actual value has not been prospectively evaluated12 and is not recommended in children because of the high risk of complications, such as bone growth alterations and development of pulmonary fibrosis.3
Adjuvant chemotherapy is usually indicated in patients with axillary node metastases.3 However, it should be noted that the NCCN clinical guidelines (NCCN Clinical Practice Guidelines in Oncology version 2.2016) do not include secretory carcinoma within the recommendations for systemic adjuvant treatment.13 This is probably due to its low incidence and its underrepresentation in adjuvant studies in breast cancer. Of course, this does not mean that they should not receive it, but probably it must be individualized in each case and maybe that aggressive systemic treatment should be avoided12 to the extent possible. In the case of expressing hormonal receptors, we consider that hormone therapy should be offered, due to the adequate risk-benefit profile of these drugs at present, although, again, the benefit has not been studied extensively. When ETV6-NTRK3 mutation in secretory carcinomas is found, it is possible that a specific inhibitor of this tyrosine kinase may support the adjuvant treatment for these tumors.3
Overall, the prognosis in children and adolescents is usually excellent, but in adults prognosis is usually unfavorable. When there is no metastasis to axillary lymph nodes, a disease-free survival of more than 15 years has been documented. Even if there is metastasis in 1–3 lymph nodes, the prognosis is usually very favorable. However, when there is metastasis to more than 3 axillary nodes, it increases the probability of systemic recurrence and a poor prognosis.3 Visceral and/or bone metastases have been documented and, when present, they often lead to patient death.2,3
Currently, oncologic therapy should be individualized for every case, though, the good prognosis for these patients could make us consider a less aggressive therapy. These cases will contribute in learning the natural behavior of the disease, in order to generate more specific treatment guidelines. Few cases have been reported in the literature, to our knowledge, there are less than three cases reported in Mexican patients.
Data privacyThe authors followed the procedures established in their center for publication of patient data.
Conflict of interestsThe authors declare no conflict of interests.