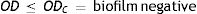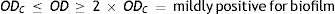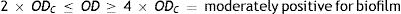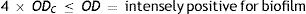Infections related to Candida albicans biofilms and subsequent antifungal resistance have become more common with the increased use of indwelling medical devices. Regimens for preventing fungal biofilm formation are needed, particularly in high-risk patients. In this study, we investigated the biofilm formation rate of multiple strains of Candida albicans (n=162 clinical isolates), their antifungal susceptibility patterns, and the efficacy of certain antifungals for preventing biofilm formation. Biofilm formation was graded using a modified Christensen's 96-well plate method. We further analyzed 30 randomly chosen intense biofilm-forming isolates using the XTT method. Minimum biofilm inhibition concentrations (MBIC) of caspofungin, micafungin, anidulafungin, fluconazole, voriconazole, posaconazole, itraconazole, and amphotericin B were determined using the modified Calgary biofilm method. In addition, the inhibitory effects of antifungal agents on biofilm formation were investigated. Our study showed weak, moderate, and extensive biofilm formation in 29% (n=47), 38% (n=61), and 23% (n=37) of the isolates, respectively. We found that echinocandins had the lowest MBIC values and that itraconazole inhibited biofilm formation in more isolates (26/32; 81.3%) than other tested agents. In conclusion, echinocandins were most effective against formed biofilms, while itraconazole was most effective for preventing biofilm formation. Standardized methods are needed for biofilm antifungal sensitivity tests when determining the treatment and prophylaxis of C. albicans infections.
Las infecciones relacionadas con las biopelículas de Candida albicans y la consiguiente resistencia antifúngica se han vuelto fenómenos habituales con el uso creciente de dispositivos médicos permanentes. Son necesarios regímenes para prevenir la formación de biopelículas fúngicas, en especial en los pacientes de alto riesgo. En este estudio se investigó la tasa de formación de biopelículas de numerosas cepas de Candida albicans (162 aislados clínicos), sus patrones de sensibilidad a los antifúngicos y la eficacia de algunos de estos agentes para prevenir la formación de biopelículas. La formación de biopelículas se clasificó utilizando el método de Christensen modificado de 96 pocillos. Posteriormente se analizaron 30 aislados de formación intensa de biopelículas elegidos al azar, utilizando el método XTT. Se calcularon las concentraciones mínimas de inhibición de biopelículas (minimum biofilm inhibition concentrations, MBIC) de la caspofungina, la micafungina, la anidulafungina, el fluconazol, el voriconazol, el posaconazol, el itraconazol y la anfotericina B, utilizando el método modificado de biopelículas de Calgary. Además, se investigaron los efectos inhibitorios de los agentes antifúngicos sobre la formación de biopelículas. Nuestro estudio encontró una formación débil, moderada e intensa de biopelículas en el 29% (n=47), 38% (n=61) y 23% (n=37) de los aislados, respectivamente. Encontramos que las equinocandinas mostraron los menores valores MBIC, y que el itraconazol inhibió la formación de biopelículas en más aislados (26/32; 81,3%) que otros agentes ensayados. En conclusión, las equinocandinas resultaron más eficaces frente a las biopelículas formadas, mientras que el itraconazol resultó más eficaz para prevenir la formación de biopelículas. Se necesita contar con métodos estandarizados para efectuar las pruebas de sensibilidad a los antifúngicos en términos de formación de biopelículas a la hora de determinar el tratamiento y la profilaxis de las infecciones por C. albicans.
Candida species are the most frequently isolated human fungal pathogens and cause a wide spectrum of diseases, including superficial mucosal, severe deep, and systemic infections42,44. The increased use of biomaterial implantations, bone marrow and solid organ transplantations, immunosuppressive treatment, and the use of broad-spectrum antibiotics are associated with severe systemic infections44.
One of the most important virulence factors of Candida infections involves the formation of biofilms42. Candida species have differing abilities for biofilm formation and frequently play a role in biofilm-associated infections42. In particular, Candida albicans is an intensely biofilm-forming fungus12,23.
Cells in the biofilm are protected from the external environment, immune system, and antifungal treatment; in fact, the most important characteristic of biofilms is their high antimicrobial resistance42. Different antimicrobials have been used in infections due to C. albicans14,31,43, whose biofilms show high resistance to fluconazole, a frequently preferred treatment2,34,42. Amphotericin B shows dose-dependent activity, while echinocandins have in vitro action against biofilms20,24,42,43. Although most new azoles affect planktonic cells, they do not sufficiently inhibit sessile cells20,24,43.
Because planktonic and sessile forms of microorganisms have different antimicrobial susceptibilities, treatment strategies based on minimal inhibitory concentration (MIC) results of planktonic cells may fail.
The incidence of life-threatening biofilm-related infections has grown with an increasing number of oncological and immunosuppressed patients and the use of indwelling medical equipment; therefore, prophylactic treatment strategies are needed15,16. Few studies have investigated the benefit of administering antifungals at subinhibitory concentrations for infection prophylaxis and their inhibitory effects on Candida biofilm formation.
In this study, we determined the rate of biofilm formation in C. albicans strains that frequently cause systemic infections. We then compared the antifungal susceptibility profiles of planktonic and sessile forms and the efficacy of antifungals at subinhibitory concentrations for preventing biofilm formation in selected isolates.
Materials and methodsMicroorganismsIn this study, we included 162 C. albicans isolates from blood cultures of patients in Ankara, Adana, and Istanbul hospitals of Başkent University. Only one isolate from each patient within a month was selected. The strains were classified as C. albicans or its related species based on germ tube formation and colony morphologies on Corn Meal Tween 80 agar (BD, France/USA).
We randomly chose 30 strains that formed extensive biofilms based on biofilm density (modified Christensen method), C. albicans ATCC 90028, and C. albicans ATCC 10231 for subsequent experiments.
Growth conditionsStrains were kept in Skim Milk (BD, France/USA) at −80°C. For each experiment, the strains were passed on Sabouraud dextrose agar (SDA, BD, France, USA) twice for purity control, then selected colonies were suspended in Yeast Nitrogen Base (YNB) broth and incubated at 37°C until the yeast cells entered the budding phase. The yeast cells were washed twice with sterile phosphate buffer solution and adjusted to a standard turbidity of 1–3×107CFU/ml in YNB broth using a spectrophotometer (Phoenix Spec, BD, USA/Ireland)21,30. Yeast cell suspensions were prepared for all experiments as described above.
Measurement of biofilm densityOne hundred microliters of prepared yeast suspension were transferred into 96-well, flat-based microplates (Costar3599, USA). The plates were incubated at 37°C in static conditions to allow biofilms to form. Wells were washed with 250μl PBS (pH 7.4) three times after incubation to remove non-adherent cells4. A modified Christensen method was used to quantify biofilm formation and density. The optical density (OD540nm) values for every well were determined with an ELx800 (Bio-Tek Instruments Inc., USA) microplate spectrophotometer. Each strain occupied three wells, and negative controls (media blanks) were included in at least 6 wells. All experiments were repeated at least twice on different days.
Four biofilm density categories were used to grade the biofilms based on established cut-off values (ODc), which were derived from the mean values of negative controls (mean ODnc) summed with three standard deviations of the negative controls (3×SDnc): ODc=mean ODnc+(3×SDnc). The biofilm density categories used are as follows:
The biofilm density grade of each strain was determined in relation with its mean OD value4,40.
Biofilm measurements in plates with needle lidsThe biofilm-forming abilities of 30 clinical isolates with intense biofilm-forming capacity, as well as C. albicans ATCC 10231 and C. albicans ATCC 90028, were investigated using the XTT method in 96-U-well microplates (Thermo-Nunc, TSP-screening, Denmark) with needle lids. All strains were tested in 3 wells and incubated under flow conditions at 37°C for 48h, then the needle lid was washed twice.
Three hundred mg/l XTT (Applichem, Germany) and 0.13mM menadione (Sigma, Germany) were distributed to produce last-well concentrations39 and the needle lids were closed. The plates were incubated at static conditions in the dark for 2h at 37°C. The needle lid was opened, OD490nm values were measured as described above, and resulting color changes were observed visually.
Antifungal susceptibility testsIn vitro antifungal susceptibilities of planktonic cells of the 30 strains showing intense biofilm formation, C. albicans ATCC 90028, and C. albicans ATCC 10231 were determined by the microdilution method in accordance with CLSI M27-A3 (S4) criteria46. Standard powder forms of antifungal agents including caspofungin (Merck, USA), micafungin (Astellas, Japan), anidulafungin (Pfizer, USA), fluconazole (Sigma), voriconazole (Sigma), posaconazole (Merck, USA), itraconazole (Sigma), and amphotericin B (Sigma) were used.
The antifungal susceptibility tests of sessile cells were performed in accordance with a modified Calgary biofilm method9. We generated 48 h biofilms in needle lids of 96-well microplates as mentioned above. Antifungal stock solutions were diluted with YNB broth supplemented with 100mM sucrose (Merck, Germany), two-fold serial dilutions were made. The antifungal concentration ranges were adjusted as follows: 0.06–32μg/ml for echinocandins, 0.25–128μg/ml for amphotericin B and itraconazole, 0.5–256μg/ml for posaconazole, 1–512μg/ml for voriconazole, and 4–2048μg/ml for fluconazole.
Needle lids with formed biofilms were washed and placed in prepared plates with antifungal agents and incubated at 37°C for 24h. After incubation, the needle lid was washed twice with PBS and transferred into the microplate containing 200μl fresh YNB with sucrose. The plate was sonicated at high power for 5min (Ultrasonic Cleaner, Daihan Scientific Co., Korea), and incubated at static conditions at 37°C for 24h. The resulting material was analyzed visually and with a microplate spectrophotometer at 630nm. The concentrations of amphotericin B that inhibited all growth and of echinocandins and azoles that significantly limited fungal growth (≥50%) were the minimal biofilm inhibition concentration (MBIC) values. The MICs of the planktonic forms and MBICs of sessile cells of the 32 strains were compared, and the decreased antifungal susceptibility of sessile cells (MBIC/MIC ratio) was calculated.
Biofilm inhibition experimentFor this experiment, quadrupled MIC values determined by CLSI reference methods were chosen as the starting concentration for each antifungal compound, and two-fold serial dilutions were made across each row.
Each yeast suspension was inoculated into each well so that yeast cells were incubated with MIC/2 – MIC/128 sub-MICs of the antifungals. Needle lids were placed on the microplates, and the plates were incubated under flow conditions on an orbital shaker for 48h at 37°C for cell adherence. After incubation, the needle lid was washed twice with PBS. Sonication was performed at high power for 5min. Dilutions and spot inoculations to SDA were performed from the wells, and the agar plates were incubated at 37°C for 48h. In addition, the needle lid was removed, and the plate was incubated at 37°C for 24h.
Wells were analyzed visually and by using a microplate spectrophotometer at 630nm. The CFU/ml values were calculated for colonies grown after spot inoculations. The biofilm inhibitory concentration (BIC) for each antifungal agent that inhibited all growth for each strain was recorded. Lower inhibition levels were not considered.
Statistical analysisData were analyzed using SPSS (version 17.0; SPSS Inc., Chicago, IL, USA). The Friedman test was used for comparing the medians of the groups. Results were presented as mean±standard deviation, median, interquartile range, or geometric means. A two-proportion Z test was used to compare the proportions, which were presented as number (n) and percentage (%). The relationships and effects among antifungals were analyzed using the Spearman's rho correlation coefficient. Statistical significance was set at p<0.05.
ResultsBiofilm formationMany of the strains showed moderate biofilm formation (38%, n=61), 47 (29%) strains showed weak biofilm formation, 37 (23%) strains showed intense biofilm formation, and 17 strains (10%) did not form biofilms. Intense biofilm formation was also observed in C. albicans ATCC 10231, while weak biofilm formation was observed in C. albicans ATCC 90028.
The biofilm capacity of 30 intensely biofilm-forming strains and the two standard strains were further analyzed using the XTT colorimetric method in microplates with needle lids by showing the metabolic activity of the sessile cells expressed as density levels between 1+ and 3+. In addition, yeast colonies were visually observed on needles in some strains.
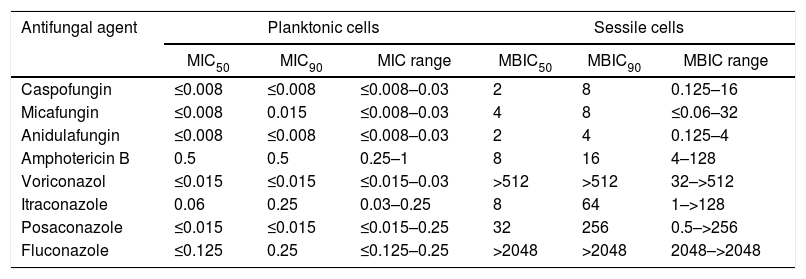
Planktonic cell antifungal susceptibilityTable 1 shows the antifungal susceptibility results of the 32 strains. Only 5 clinical isolates (16.7%) and C. albicans ATCC 10231 exhibited dose-dependent susceptibility to itraconazole.
Antifungal susceptibility test results for planktonic (MIC) and sessile (MBIC) cells (μg/ml) of 32 C. albicans strains
| Antifungal agent | Planktonic cells | Sessile cells | ||||
|---|---|---|---|---|---|---|
| MIC50 | MIC90 | MIC range | MBIC50 | MBIC90 | MBIC range | |
| Caspofungin | ≤0.008 | ≤0.008 | ≤0.008–0.03 | 2 | 8 | 0.125–16 |
| Micafungin | ≤0.008 | 0.015 | ≤0.008–0.03 | 4 | 8 | ≤0.06–32 |
| Anidulafungin | ≤0.008 | ≤0.008 | ≤0.008–0.03 | 2 | 4 | 0.125–4 |
| Amphotericin B | 0.5 | 0.5 | 0.25–1 | 8 | 16 | 4–128 |
| Voriconazol | ≤0.015 | ≤0.015 | ≤0.015–0.03 | >512 | >512 | 32–>512 |
| Itraconazole | 0.06 | 0.25 | 0.03–0.25 | 8 | 64 | 1–>128 |
| Posaconazole | ≤0.015 | ≤0.015 | ≤0.015–0.25 | 32 | 256 | 0.5–>256 |
| Fluconazole | ≤0.125 | 0.25 | ≤0.125–0.25 | >2048 | >2048 | 2048–>2048 |
MIC: minimal inhibitory concentration, MBIC: minimal biofilm inhibition concentration, MIC50: MIC value, which inhibits 50% of the tested strains, MBIC50: MBIC value, which inhibits 50% of the tested strains, MIC90: MIC value, which inhibits 90% of the tested strains, MBIC90: MBIC value, which inhibits 90% of the tested strains, MIC range: range of MIC values of all tested strains, MBIC range: range of MBIC values of all tested strains.
Table 1 also shows biofilm susceptibility results. Echinocandins had the lowest MBIC values (MBIC50 of 2μg/ml for caspofungin and anidulafungin and 4μg/ml for micafungin; MBIC90 of 4μg/ml for anidulafungin and 8μg/ml for caspofungin and micafungin. The lowest MBIC90 (4μg/ml) was obtained with anidulafungin. Among the azoles, itraconazole had the lowest MBIC50 and MBIC90 values (8μg/ml and 64μg/ml, respectively).
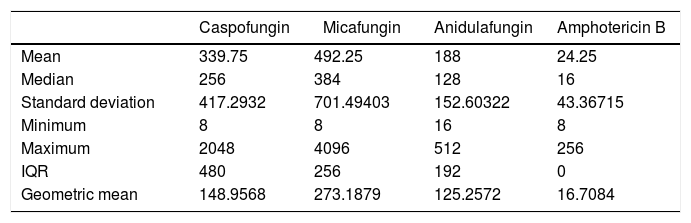
Comparison of antifungal susceptibilities of planktonic and sessile cellsAmong the 32 strains tested, MBIC values of the antifungals were greater than their MIC values (Table 1). Table 2 shows MBIC/MIC ratios of the antifungals, showing that the median MBIC/MIC ratio of amphotericin B had the lowest MBIC/MIC ratio among the antifungals (p<0.05). The MBIC/MIC ratio of micafungin was significantly higher than the other antifungal agents. The MBIC/MIC ratio of posaconazole was significantly lower than fluconazole or voriconazole, whose ratios were the highest among the azoles tested (p<0.05).
MBIC/MIC ratio values of sessile and planktonic cells
| Caspofungin | Micafungin | Anidulafungin | Amphotericin B | |
|---|---|---|---|---|
| Mean | 339.75 | 492.25 | 188 | 24.25 |
| Median | 256 | 384 | 128 | 16 |
| Standard deviation | 417.2932 | 701.49403 | 152.60322 | 43.36715 |
| Minimum | 8 | 8 | 16 | 8 |
| Maximum | 2048 | 4096 | 512 | 256 |
| IQR | 480 | 256 | 192 | 0 |
| Geometric mean | 148.9568 | 273.1879 | 125.2572 | 16.7084 |
| Voriconazole | Itraconazole | Posaconazole | Fluconazole | |
|---|---|---|---|---|
| Mean | 38016 | 419.25 | 4745 | 26368 |
| Median | 32768 | 64 | 1536 | 32768 |
| Standard deviation | 21065.79006 | 998.40773 | 9376.42182 | 8511.94567 |
| Minimum | 2048 | 8 | 32 | 8192 |
| Maximum | 65536 | 4096 | 32768 | 32768 |
| IQR | 32768 | 224 | 3072 | 16384 |
| Geometric mean | 29404.5135 | 92.4916 | 1327.9637 | 24726.1500 |
MIC: minimal inhibitory concentration, MBIC: minimal biofilm inhibition concentration, IQR: interquartile range.
The analysis of MBIC/MIC ratio correlations of the antifungals revealed that caspofungin significantly correlated with micafungin (Spearman's rho=0.572, p<0.001) and anidulafungin (Spearman's rho=0.414, p<0.05) and had similar effects on the 32 strains. Posaconazole showed significant correlations with voriconazole (Spearman's rho=0.402, p<0.05) and itraconazole (Spearman's rho=0.684, p<0.001). In summary, antifungals in the same group showed similar effects on the yeast strains; however, no significant correlations of the effects among the different groups (echinocandins, azoles, and amphotericin B) were observed.
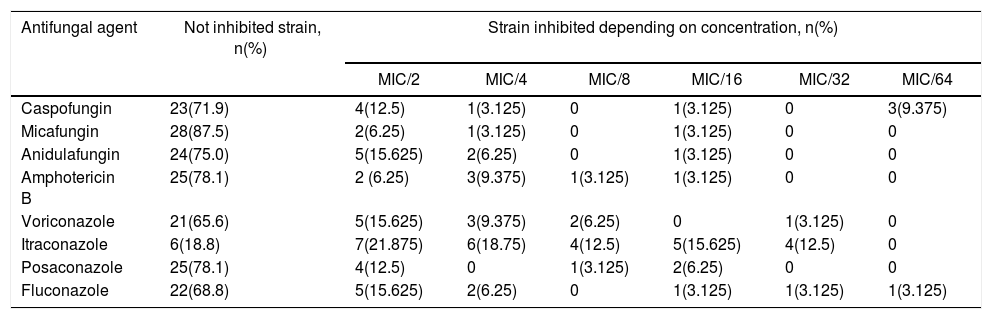
Biofilm inhibition experimentsTable 3 shows the number of C. albicans strains inhibited by different antifungal agents at sub-inhibitory concentrations. The concentration that inhibited biofilm formation 100% at needle-tips has been determined as biofilm inhibitory concentration (BIC). BIC results for echinocandins ranged from 0.0001 to 0.004μg/ml for caspofungin, 0.001 to 0.004μg/ml for micafungin, and 0.001 to 0.008μg/ml for anidulafungin. The BIC range of amphotericin B was 0.03–0.25μg/ml.
Proportion of C. albicans strains inhibited by different antifungal agents at sub-inhibitory concentrations
| Antifungal agent | Not inhibited strain, n(%) | Strain inhibited depending on concentration, n(%) | |||||
|---|---|---|---|---|---|---|---|
| MIC/2 | MIC/4 | MIC/8 | MIC/16 | MIC/32 | MIC/64 | ||
| Caspofungin | 23(71.9) | 4(12.5) | 1(3.125) | 0 | 1(3.125) | 0 | 3(9.375) |
| Micafungin | 28(87.5) | 2(6.25) | 1(3.125) | 0 | 1(3.125) | 0 | 0 |
| Anidulafungin | 24(75.0) | 5(15.625) | 2(6.25) | 0 | 1(3.125) | 0 | 0 |
| Amphotericin B | 25(78.1) | 2 (6.25) | 3(9.375) | 1(3.125) | 1(3.125) | 0 | 0 |
| Voriconazole | 21(65.6) | 5(15.625) | 3(9.375) | 2(6.25) | 0 | 1(3.125) | 0 |
| Itraconazole | 6(18.8) | 7(21.875) | 6(18.75) | 4(12.5) | 5(15.625) | 4(12.5) | 0 |
| Posaconazole | 25(78.1) | 4(12.5) | 0 | 1(3.125) | 2(6.25) | 0 | 0 |
| Fluconazole | 22(68.8) | 5(15.625) | 2(6.25) | 0 | 1(3.125) | 1(3.125) | 1(3.125) |
MIC: minimal inhibitory concentration.
BIC ranges of the azoles were as follows: 0.0005–0.008μg/ml for voriconazole, 0.06–0.015μg/ml for itraconazole, 0.002–0.008μg/ml for posaconazole, and 0.004–0.06μg/ml for fluconazole.
Most of the antifungal agents tested inhibited biofilm formation at similar proportions. Itraconazole completely inhibited biofilm formation in more strains (26/32, 81.3%; p<0.05) than other antifungals. Micafungin showed significantly less inhibition (4/32 strains) when compared to caspofungin, itraconazole, voriconazole, and fluconazole (p<0.05); however no significant differences among anidulafungin, amphotericin B, and posaconazole were observed.
DiscussionThe increased prevalence of biofilm-related infections in high-risk patients is a significant health concern. In our study, we detected biofilm formation in 90% of 162 C. albicans isolates obtained from blood cultures, which is a higher rate than that observed in previous studies6,7,29,36. The choice of assay may influence this finding, since the Christensen method stains dead cells, living cells, and the extracellular matrix, while the XTT assay only measures the metabolism of living cells.
Mature biofilms are complex structures containing sessile cells with different metabolic activities42. Moreover, studies have shown disagreement regarding the significant correlation between metabolic activity as measured by XTT and cell numbers determined by other methods11,22,23,34. Therefore, previous data, together with our own findings, prompt us to consider if comparing results obtained from different assays can accurately characterize biofilm formation in these strains.
Plates with needle lids are frequently used to detect bacterial biofilms and their antimicrobial susceptibilities; however, few studies utilizing this approach with Candida cells have been published10,28,30,41. Studies reported that the optimal cell number for Candida biofilm formation was 107CFU/ml, and sucrose significantly increased C. albicans biofilm formation on needles23,30. Therefore, we adjusted the yeast inocula to McFarland 1 (30×107CFU/ml for bacteria) and diluted the suspension 1 in 30 with growth medium because yeast cells are larger in size than bacteria and few cells are needed for biofilm formation8. Therefore, we optimized the conditions and determined significant biofilm using the Calgary method9.
In our study, the planktonic forms of all 30 extensive biofilm-forming isolates were susceptible to echinocandins and azoles as reported in previous epidemiological studies26,32.
Several studies have investigated the effect of fluconazole on sessile C. albicans cells and found MBIC ranges of 4–1024μg/ml2,3,25,27,28,34. In contrast, we found higher MBIC50 and MBIC90 values (≥2048μg/ml). Only few studies have tested the effect of itraconazole on sessile cells. We observed similar MBIC values (1–128μg/ml) as indicated in these studies13,27,28. The published MBIC range for voriconazole is 0.5–>265μg/ml20,27,37; however, only a few studies have investigated the efficacy of posaconazole on sessile cells20,43. Similarly to previous results, we found that sessile cells were resistant to voriconazole and posaconazole.
The published amphotericin B MBIC range is 0.015–2μg/ml2,3,28,37. We observed higher MBIC values (4–128μg/ml) and greater amphotericin B resistance among our clinical isolates compared to data in the aforementioned studies.
Many studies have found that echinocandins effectively inhibit C. albicans sessile cells at MBIC of 0.03–4μg/ml of caspofungin, ≤0.03–2μg/ml of micafungin, and 0.015–16μg/ml of anidulafungin3,17,18,20,24,37,38. We determined the MBIC ranges for caspofungin and micafungin as 0.125–16μg/ml and ≤0.06–32μg/ml, respectively. Although these overall ranges are similar to those published in other studies, the maximum values that we observed were higher, with the exception of anidulafungin17,20,38.
Although there are no standard breakpoints for the susceptibility of sessile cells, the MBIC values of echinocandins were partially within the planktonic susceptibility range of CLSI. Among the intense biofilm-forming and standard strains, 11 were susceptible to caspofungin, 9 were susceptible to anidulafungin, and 5 were susceptible to micafungin. In addition, the MBIC50 of caspofungin was within the therapeutic concentration range (2μg/ml) as indicated by Cocuaud et al.5, and caspofungin was effective against 50% of the clinical isolates we tested.
Overall, the MBIC values found in our investigation were higher than those found in some studies2,4,10,13,17,28,34,37 but similar to others13,24,27,28,34. The higher MBIC values we observed may be due to our 48-h biofilm formation period, the relatively short 24-h exposure to each antifungal, and the intense biofilm-forming capabilities of our strains. In addition, methodological differences and the isolation of our strains from blood, as opposed to other culture sources, may be significant factors.
In our study, we compared MBIC/MIC ratios of the antifungal agents to determine their efficacy on biofilm maintenance directly, which has not been widely reported43. The lowest statistically significant MBIC/MIC ratio was determined for amphotericin B, although the MBIC/MIC ratio is based on planktonic MIC values. Although the MBIC/MIC ratio was low for amphotericin B, its MBIC values were above the susceptibility ranges provided by CLSI, highlighting its ineffectiveness at therapeutic concentrations and toxicity at high doses in infections involving planktonic cells33.
Very low planktonic MIC values found for echinocandins in our study resulted in higher MBIC/MIC ratios when compared to amphotericin B. The importance of our finding is that the MBIC values of the echinocandins were closest to the planktonic susceptibility limits of CLSI criteria.
We expected azoles to be less effective on sessile cells, but the MBIC/MIC ratio of fluconazole and voriconazole was >32000 and higher than anticipated. The MBIC/MIC ratio of posaconazole was only 1536, echoing the results of Tobudic et al., who reported decreased efficacy of posaconazole during all biofilm phases of C. albicans43.
Administering lower doses of antifungal agents over a longer period of time in some high-risk patient groups for treatment or prophylaxis is of interest, as blood levels drop below clinical subinhibitory concentrations or in vitro MIC values15,16. To date, only a few studies have explored the inhibitory effect of antifungal agents on Candida biofilm formation and with varying methodologies, such as enabling biofilm formation post-exposure to an antifungal agent or co-incubating the cells with an antifungal agent1,16,24. In our study, C. albicans cells were incubated with MIC/2–MIC/128 sub-MICs of each antifungal agent, and the concentration that inhibited biofilm formation 100% at needle-tips was the BIC. We encountered some methodological difficulties, such as a fluctuating growth density observed in some strain-antifungal combinations, and instead measured the 100% biofilm inhibitory concentration. In general, total inhibition was seen close to MIC values (MIC/2 and MIC/4). We also found that needle lids were not suitable for measuring biofilm formation inhibition due to the fluctuant growth pattern of cells and difficulty in obtaining reproducible results.
The biofilm formation of some strains was not inhibited and sometimes increased close to the MIC of the antifungal agent. Schadow et al. first showed increased Staphylococcus epidermidis biofilm formation at sub-MIC values of rifampin19,35, and since then various bacteria-antibiotic combinations have been observed19. Dose-effect efficacy curves of an agent are usually biphasic and characterized by biofilm formation at low doses and biofilm inhibition at high doses. Some antibiotics act as antagonists for biofilm formation at low or high doses but as agonists at moderate doses. Similarly, we observed significant inhibition of biofilm formation sub-MIC levels in some strain-antifungal combinations but less inhibition at some higher concentrations. This fluctuant growth pattern was seen in several yeast-echinocandin combinations in contrast to amphotericin B or most of the azoles, with the exception of posaconazole.
Strain-specific factors may have influenced our study because this effect was not seen in all strains. Sub-inhibitory concentrations of an antimicrobial agent often cause physiological changes in the microorganism, such as stimulation or inhibition of enzyme or toxin production15,16,45,47. Simitsopoulou et al.38 reported a paradoxical increase in biofilm formation of C. albicans strains at concentrations higher than the MIC, which led to uncertainty regarding the ability of echinocandins to reach sub-inhibitory concentrations in the biofilms, induce chitin formation, and antifungal resistance.
To our knowledge, no previous studies have investigated Candida biofilm capabilities using 8 different antifungal agents with the Calgary method in the number of strains used in our research. However, our methodology could be improved by combining the BioTimer and Calgary approaches to obtain earlier test results and enable more timely clinical decision-making regarding prophylaxis and treatment. We also evaluated the use of needle lids to investigate inhibition of Candida biofilm formation and found that, despite its ease of use, this method does not yield reproducible results due to fluctuant growth patterns. Thus, we recommend needle lids be combined with or replaced by methods that measure metabolism or depend on the morphological analysis of the cells.
Our results indicate that echinocandins are suitable for treating biofilm-related infections, and that itraconazole may be used for prophylaxis of such infections involving C. albicans. The increased prevalence of biofilm-associated fungal infections of C. albicans needs the development of new treatment and prophylaxis strategies, especially in high-risk patients. An antifungal ability to limit biofilm formation should be considered when selecting an appropriate agent. In addition, even agents effective at sub-MIC levels could lead to antifungal resistance and should be used judiciously16. Therefore, further in vivo and in vitro studies exploring the use of sub-MICs of antifungal agents in prophylaxis strategies and the application of standardized biofilm measurement methods are needed to determine the unique dosage and exposure time of an antifungal for its maximum anti-biofilm effectiveness and patient safety.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestThe authors declare that they have no conflicts of interest.
This study was approved by the Baskent University Institutional Review Board (Project no: KA13/150). Our research was supported by the Baskent University Research Fund, Astellas Pharma/Japan (micafungin), Merck Sharp & Dohme Corp./USA (caspofungin, posaconazole), and Pfizer/USA (anidulafungin).