The lack of consensus and specific guidelines, and the introduction of new treatments in thrombocytopenia management in liver cirrhosis patients, required a series of recommendations by experts to improve knowledge on this disease. This study's aim was to improve the knowledge around thrombocytopenia in liver cirrhosis patients, in order to contribute to the generation of future evidence to improve the management of this disease.
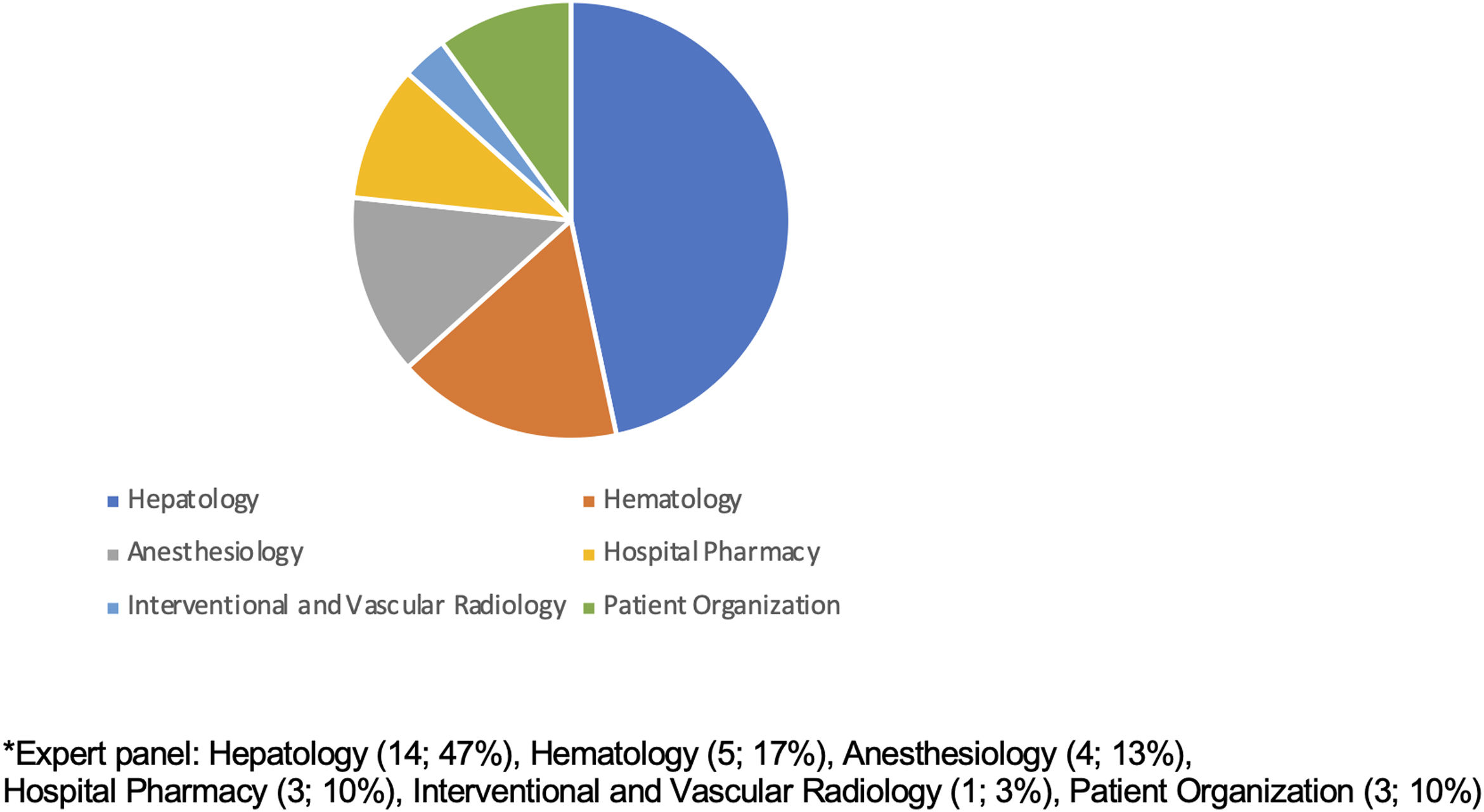
Patients and methodsA modified version of the RAND/UCLA appropriateness method was used. The scientific committee, a multidisciplinary team of 7 experts in managing thrombocytopenia in liver cirrhosis patients, identified the expert panel, and participated in elaborating the questionnaire. Thirty experts from different Spanish institutions were invited to answer a 48-item questionnaire covering 6 areas on a nine-point Likert scale. Two rounds were voted. The consensus was obtained if >77.7% of panelists reached agreement or disagreement.
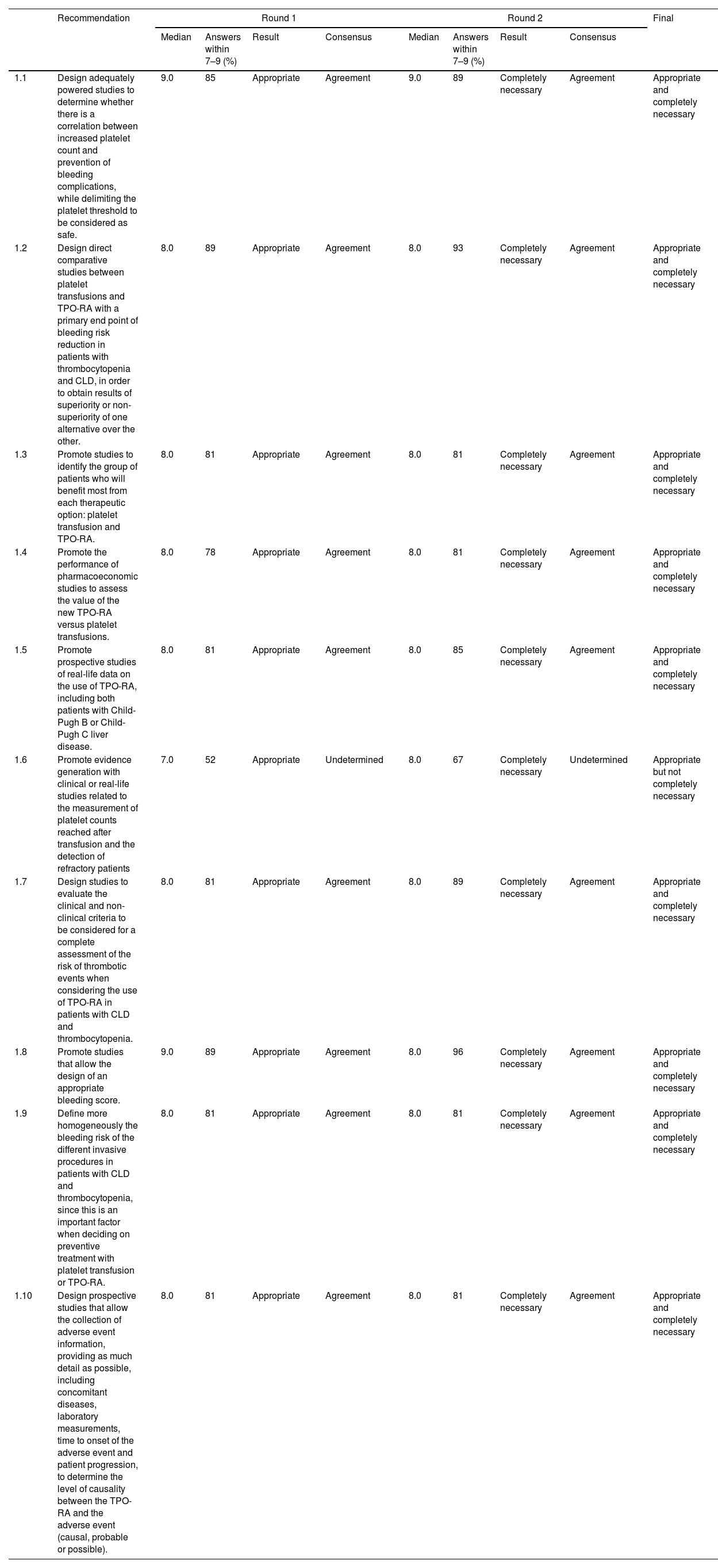
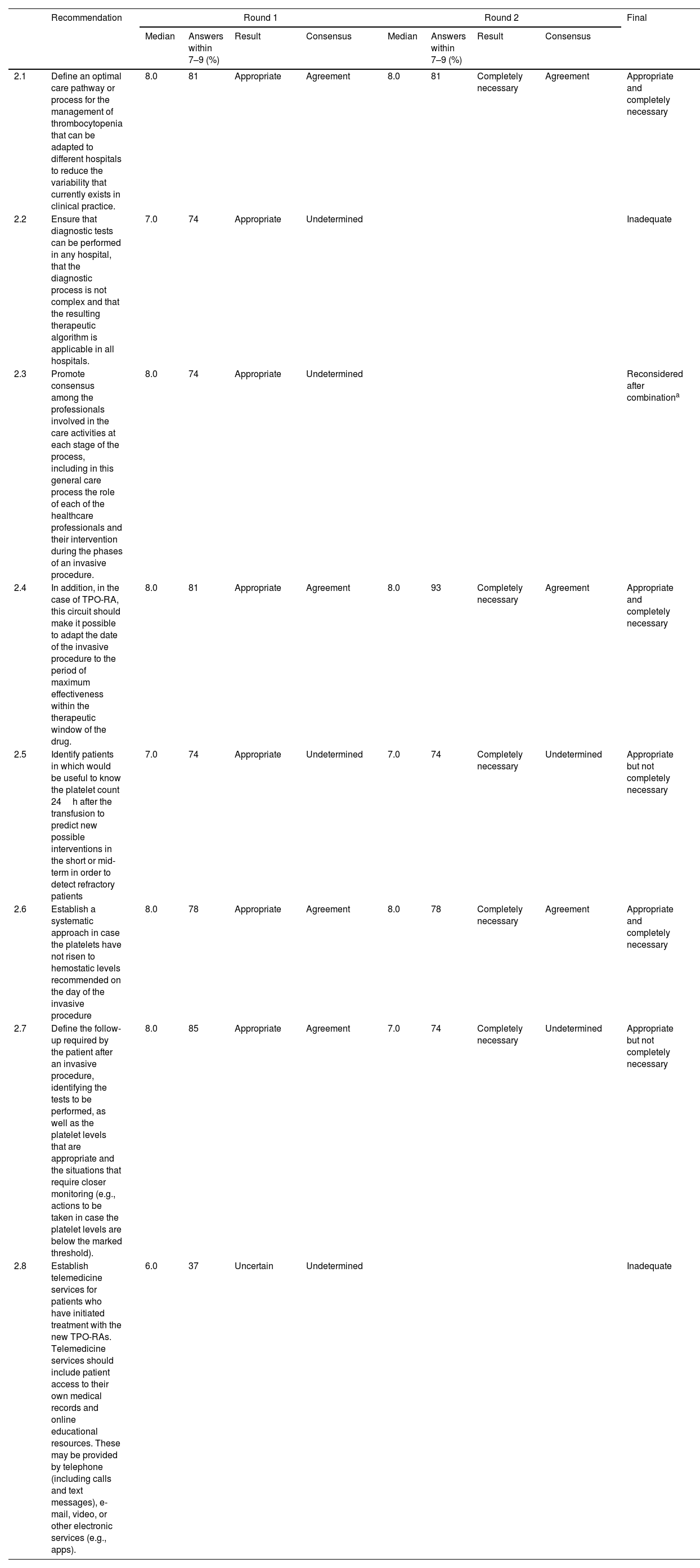
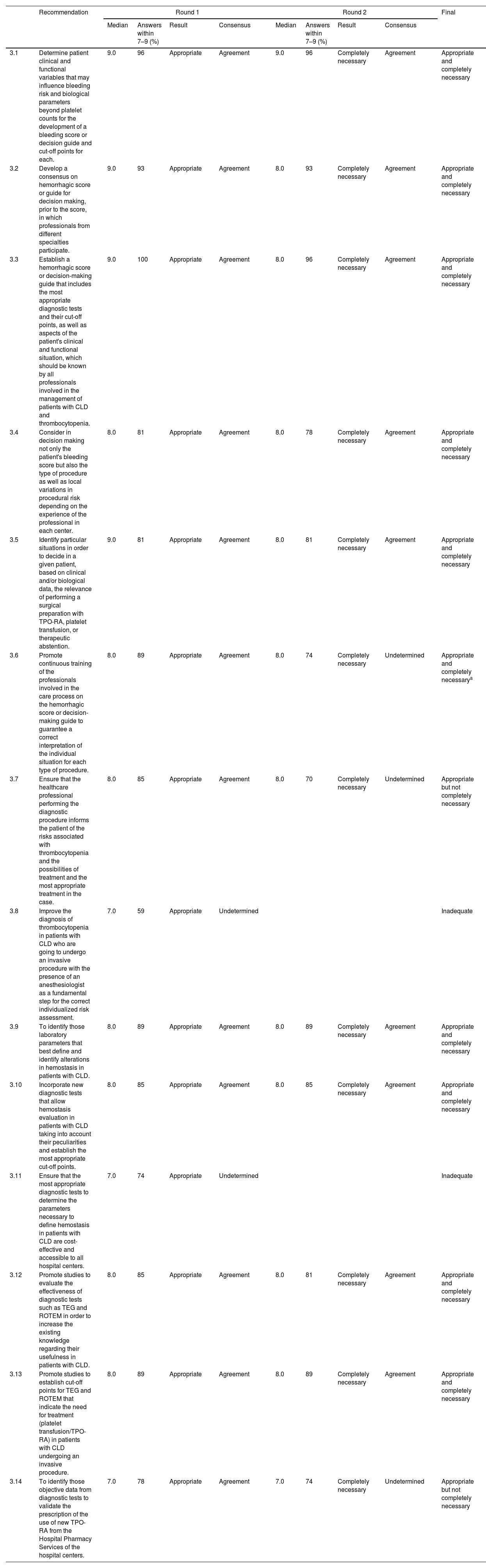
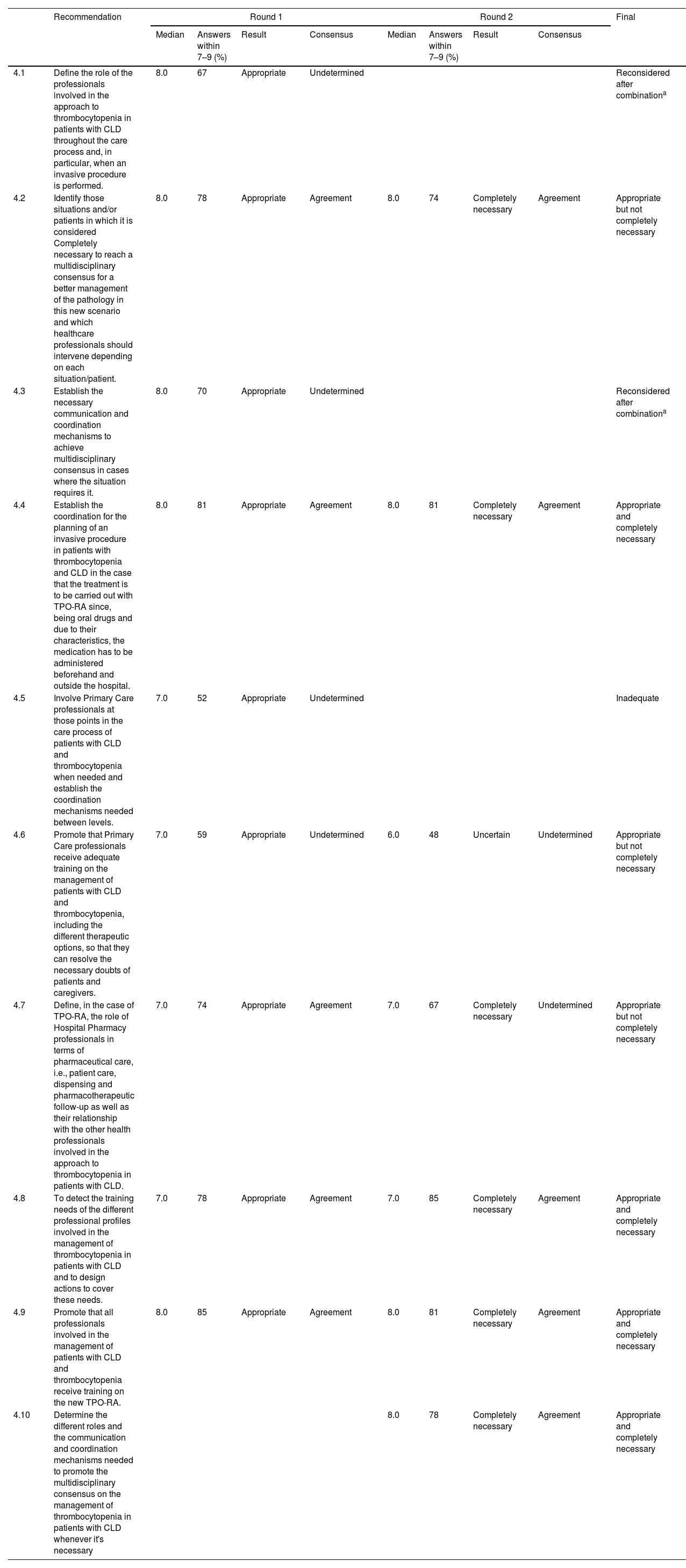
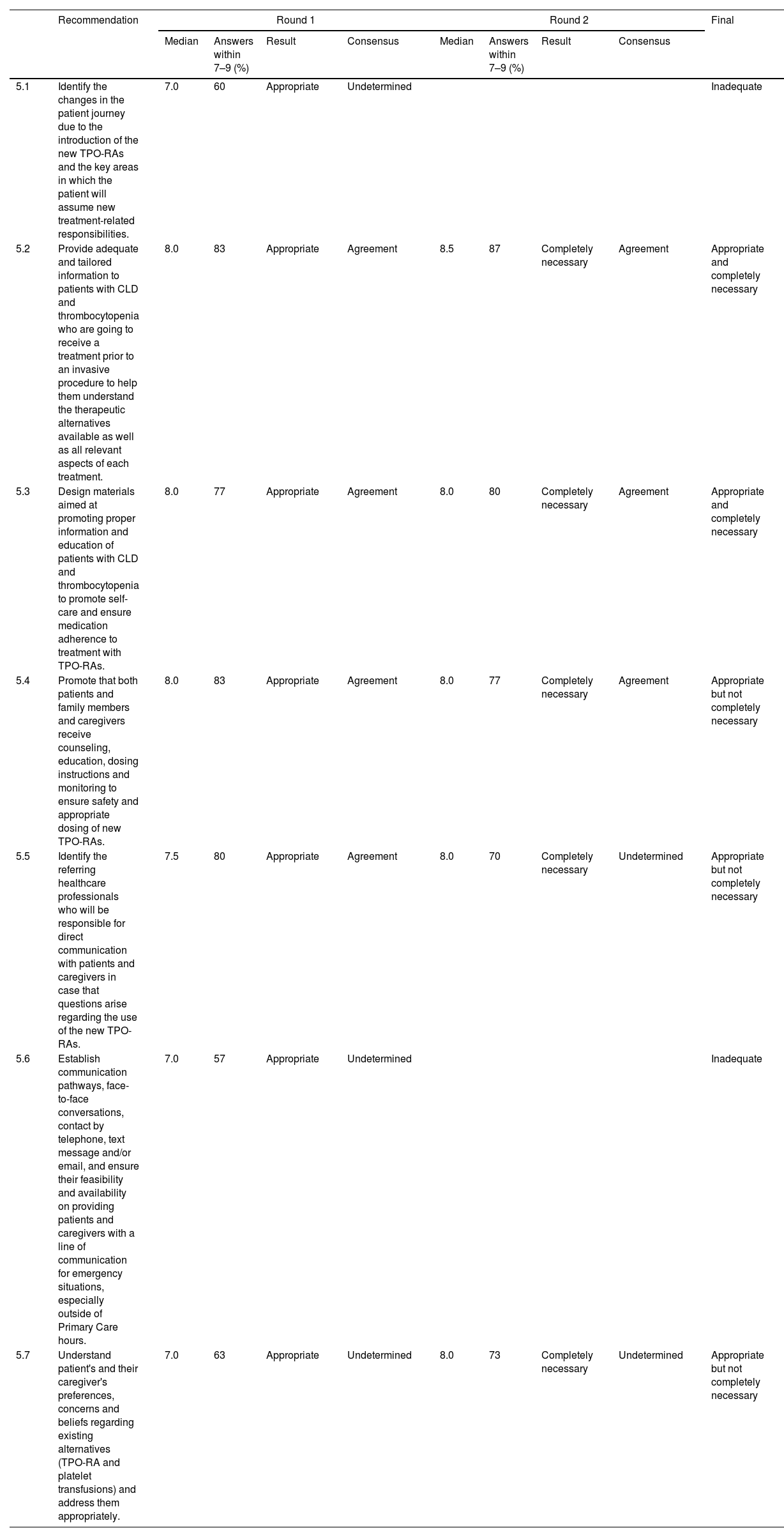
ResultsA total of 48 statements were developed by the scientific committee and then voted by the experts, resulting in 28 defined as appropriate and completely necessary, relating to evidence generation (10), care circuit, (8), hemorrhagic risk assessment, decision-making and diagnostic tests (14), professionals’ role and multidisciplinary coordination (9) and patient education (7).
ConclusionsThis is the first consensus in Spain on the management of thrombocytopenia in liver cirrhosis patients. Experts indicated several recommendations to be carried out in different areas that could help physicians make better decisions in their clinical practice.
La falta de consenso y guías específicas, y la introducción de nuevos tratamientos para el manejo de la trombocitopenia en pacientes con cirrosis hepática, requerían recomendaciones expertas para mejorar el conocimiento sobre dicha patología. El objetivo de este estudio es mejorar el conocimiento sobre la trombocitopenia en pacientes con cirrosis hepática de cara a contribuir en la generación de futuras evidencias que mejoren el manejo de esta patología.
MetodologíaAe utilizó una versión modificada de la metodología Delphi RAND/UCLA. El comité científico, formado por 7 expertos en el manejo de la trombocitopenia en pacientes con cirrosis hepática, identificó un panel de expertos y participó en la elaboración del cuestionario de recomendaciones. Treinta expertos de diferentes hospitales españoles fueron invitados a responder al cuestionario. Los expertos respondieron a 48 ítems divididos en 6 áreas en una escala Likert de 9 puntos. La votación tuvo lugar en 2 rondas, en las que se obtuvo consenso siempre y cuando >77,7% de los panelistas alcanzasen acuerdo o desacuerdo.
ResultadosCuarenta y ocho recomendaciones fueron elaboradas por el comité científico para su votación por parte del panel de expertos. Finalmente 28 recomendaciones fueron consideradas apropiadas y completamente necesarias: 10 de ellas relativas a la generación de evidencia; 8 al circuito de cuidados; 14 a la evaluación de riesgo hemorrágico, la toma de decisiones y los test diagnósticos; 9 al papel de los profesionales y la coordinación multidisciplinar, y 7 a la educación de los pacientes.
ConclusionesSe trata del primer consenso español en el manejo de la trombocitopenia en pacientes con cirrosis hepática. Los expertos definieron un amplio número de recomendaciones que podrían contribuir a la toma de decisiones clínicas y a la mejora en el manejo de estos pacientes en la práctica clínica real.