End TB Strategy aims to reduce the incidence of tuberculosis between 2015 and 2025 milestone by 50%. We analyse whether the decreasing incidence objectives of this strategy can be achieved in Spain with the impact of COVID-19 pandemic and to review the incidence evolution in Western European countries (WEC).
Study designRetrospective longitudinal ecological study with forecasting.
MethodologyAn exponential curve was fitted to the Spanish and WEC data prior the pandemic period and projected with the model until 2025 using a 90% confidence interval to have better precision due to the sample size.
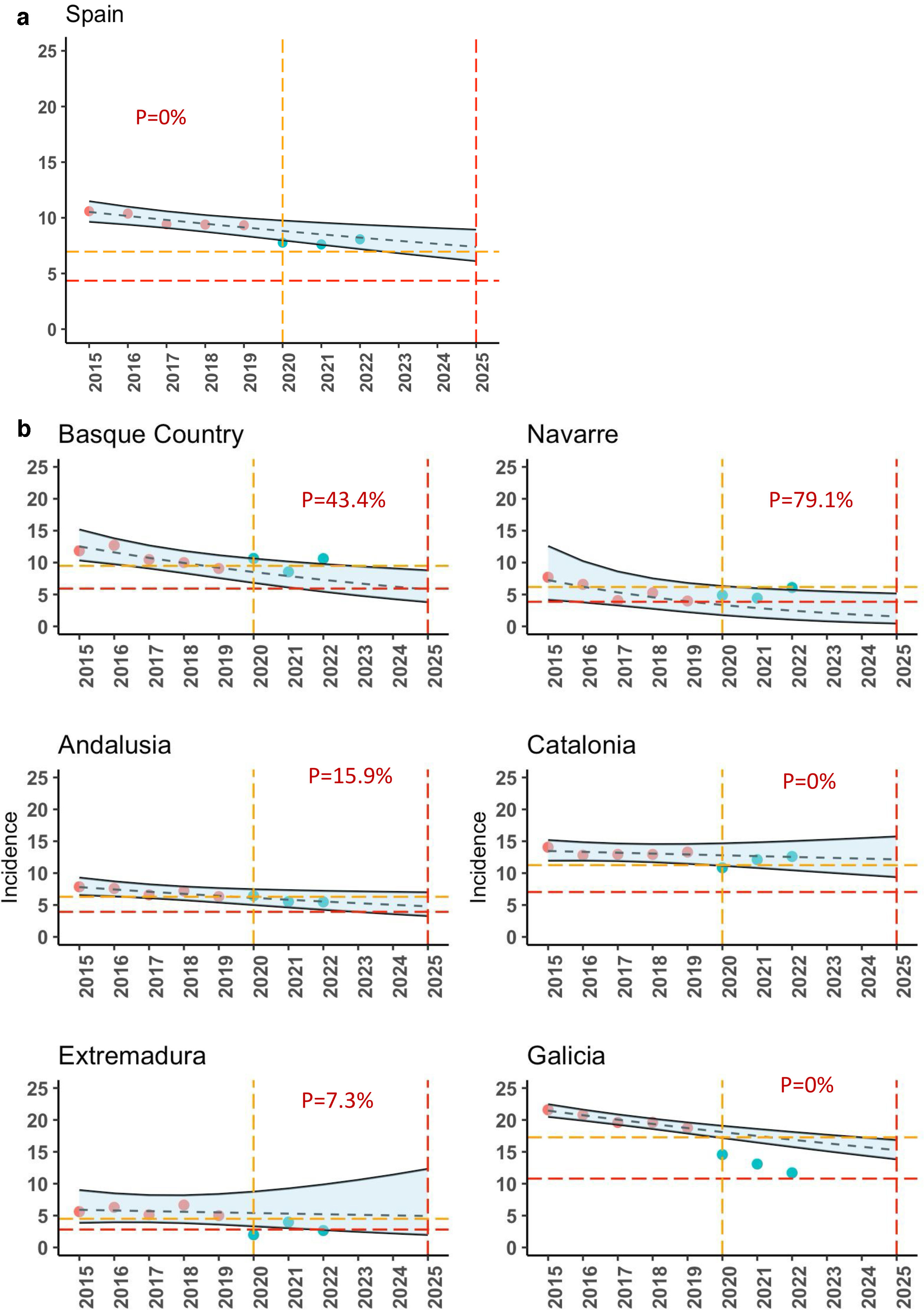
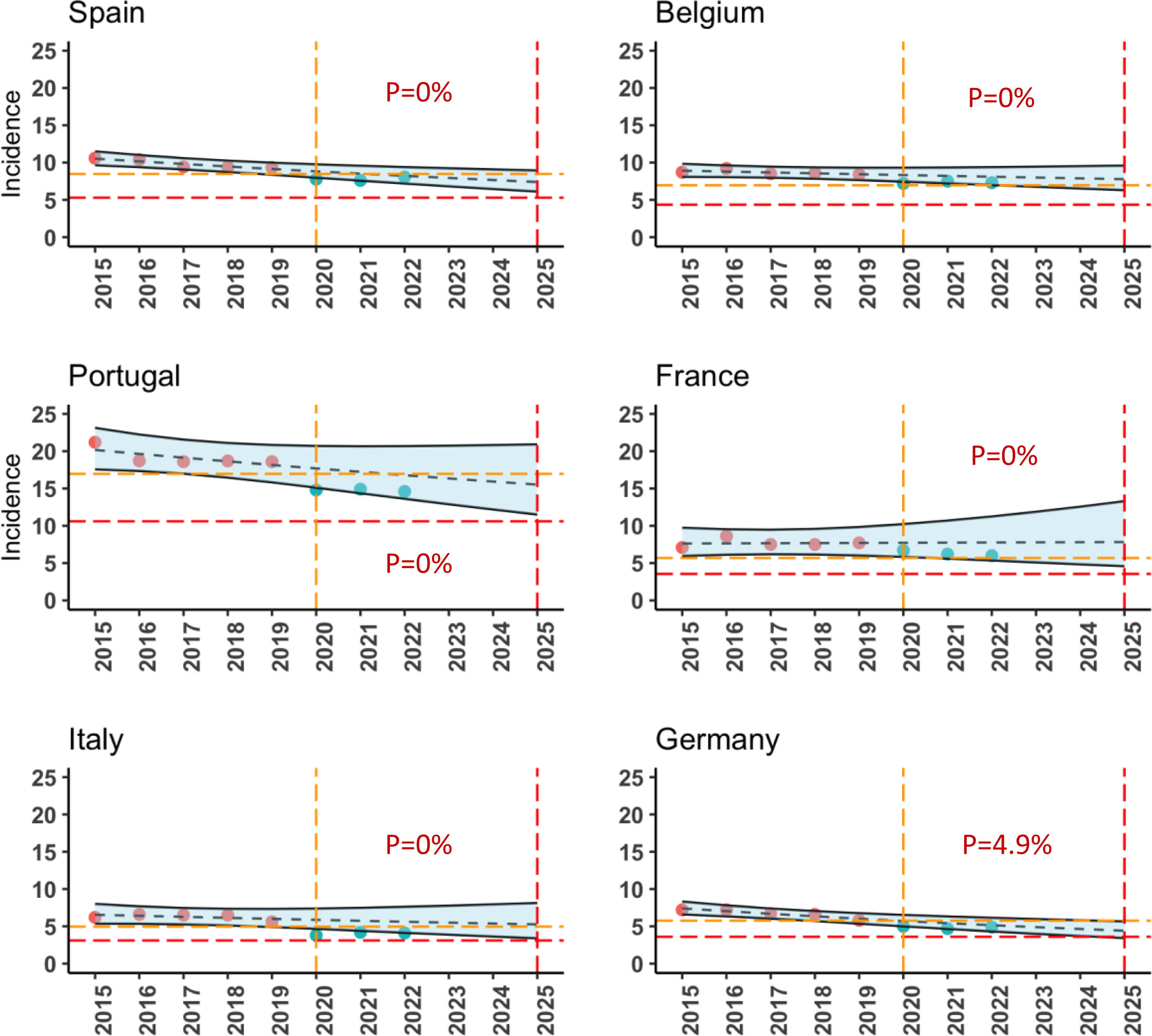
ResultsThe mean annual % change in incidence rates of TB in the three most affected WEC during 2018–2022 decreased by 6.5% in Spain, by 6.0% in Portugal, by 3.8% in Belgium and by 5.7% in France. The annual decrease in Spain in the first year of COVID-19 pandemic was 16.6%. Lower declines than in Spain were observed in most WEC. The probability of achieving the objective of reducing the incidence by 50% between 2015 and 2025 in Spain is, with a certainty of 90%, 0%, but with important differences by regions from 0% to 79%. These probabilities in Portugal, Belgium, France and Italy are also 0%.
ConclusionWith this epidemiological evolution, the main objective of the End TB Strategy for 2025 milestone (50% incidence decline) will not be achieved in Spain. The 80% decline will probably not be reached by 2030 unless surveillance and control are improved, and TB Programmes are provided with sufficient resources. The same situation could be happening in other WEC.
La Estrategia fin de la TB tiene como objetivo reducir la incidencia de la tuberculosis entre 2015 y 2025 en un 50%. Analizamos si los objetivos de disminución de la incidencia de esta estrategia pueden lograrse en España con el impacto de la pandemia de COVID-19 y revisamos su evolución en los países de Europa occidental (PEO).
Diseño del estudioEstudio ecológico longitudinal retrospectivo con pronóstico.
MetodologíaSe ajustó una curva exponencial a los datos de España y de los PEO antes del período pandémico y se proyectó con el modelo hasta 2025 utilizando un intervalo de confianza del 90% para tener una mejor precisión debido al tamaño de la muestra.
ResultadosEl cambio porcentual medio anual en las tasas de incidencia de TB en los 3 PEO más afectados durante 2018-2022 disminuyó en un 6,5% en España, en un 6,0% en Portugal, en un 3,8% en Bélgica y en un 5,7% en Francia. La disminución anual en España en el primer año de la pandemia de COVID-19 fue del 16,6%. Se observaron descensos menores que en España en la mayoría de los PEO. La probabilidad de alcanzar el objetivo de reducir la incidencia en un 50% entre 2015-2025 en España es, con una certeza del 90%, del 0%, pero con importantes diferencias entre regiones, desde el 0% hasta el 79%. Estas probabilidades en Portugal, Bélgica, Francia e Italia también son del 0%.
ConclusiónCon esta evolución epidemiológica el objetivo principal de la Estrategia fin de la TB para el hito de 2025 (reducción del 50% en la incidencia) no se logrará en España. La disminución del 80% probablemente no se alcanzará para 2030, a menos que se mejoren la vigilancia y el control y se proporcionen suficientes recursos a los programas de TB. La misma situación podría estar ocurriendo en otros PEO.
Artículo
Socio de la Sociedad Española de Enfermedades Infecciosas y Microbiología Clínica

Para acceder a la revista
Es necesario que lo haga desde la zona privada de la web de la SEIMC, clique aquí