Edited by: Dr. Calos Cerdán
Cirugía General y Aparato Digestivo. Unidad de Cirugía Digestiva. Hospital Universitario de la Princesa. Madrid
Dr. Matteo Frasson
Cirugía general. Hospital Universitario y Politécnico La Fe. Valencia
Last update: November 2025
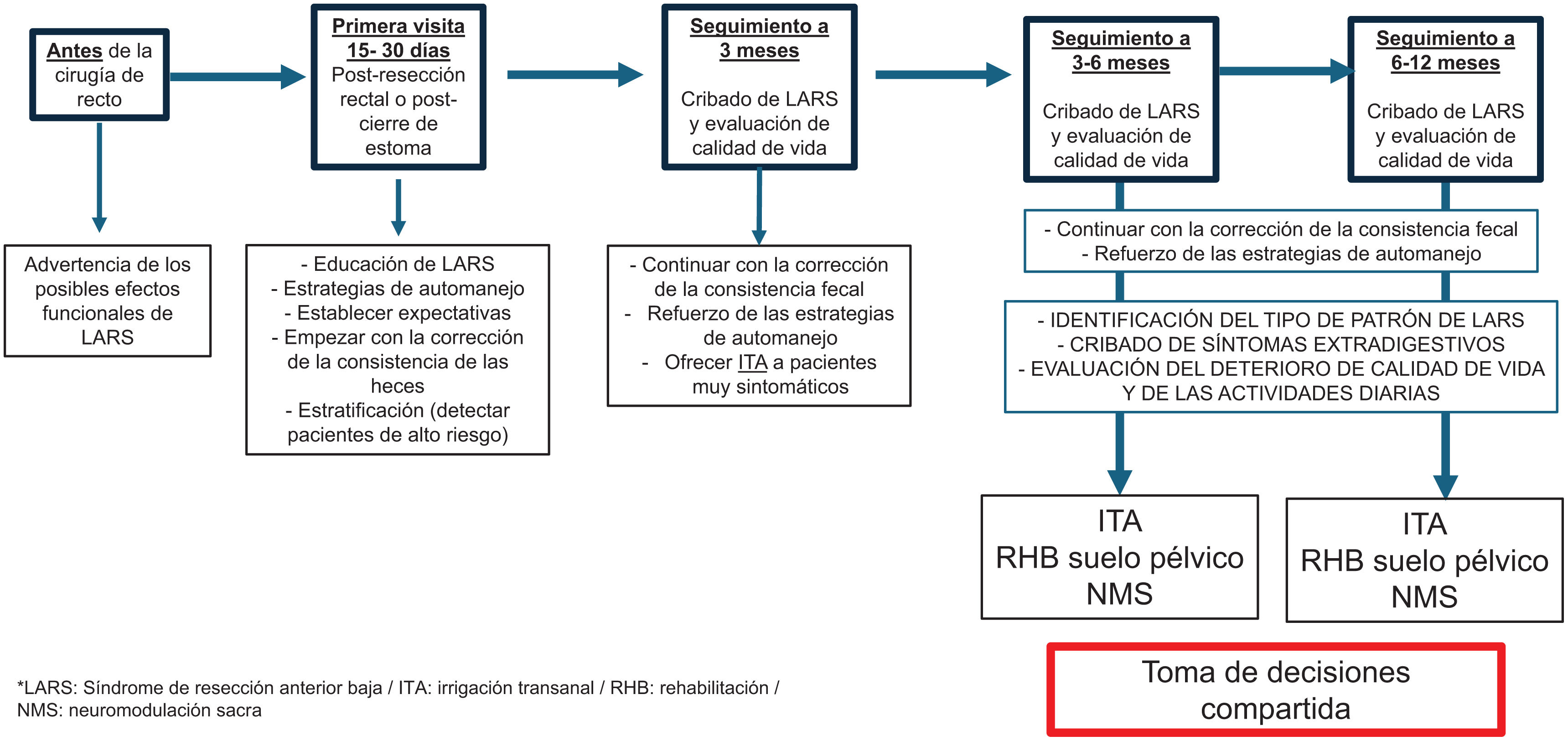
More infoLow anterior resection syndrome is a common but underestimated complication after rectal cancer surgery that significantly impacts the quality of life of the surviving patient. It is characterised by symptoms such as faecal incontinence and voiding dysfunction and affects up to 90% of patients undergoing low anterior rectal resection. The aetiology of the syndrome is multifactorial with no clear determining factor. It includes the use of preoperative radiotherapy, the indication for a protective ileostomy, sphincteric lesions, nerve damage to the rectal autonomic plexuses, and changes in left colon motor function. Although various therapeutic modalities have been shown to be effective in the management of the symptoms of the syndrome, there is still no standard treatment or patient selection pattern. In this article, a critical review of the therapeutic possibilities for patients who have survived rectal surgery will be made.
El síndrome de resección anterior baja es una complicación común pero subestimada después de la cirugía de cáncer de recto, que impacta significativamente en la calidad de vida del paciente sobreviviente. Está caracterizado por síntomas como incontinencia fecal y la disfunción evacuatoria, y afecta hasta el 90% de los pacientes sometidos a resección anterior baja de recto. La etiología del síndrome es multifactorial sin un factor claro determinante. Se incluye el uso de radioterapia preoperatoria, la indicación de una ileostomía de protección, las lesiones esfinterianas, el daño nervioso de los plexos autonómicos rectales y cambios en la función motora del colon izquierdo. Aunque diversas modalidades terapéuticas han demostrado ser efectivas en el manejo de los síntomas del síndrome, aún no existe un tratamiento estándar ni patrón de selección de pacientes. En este artículo se hará una revisión crítica de las posibilidades terapéuticas para pacientes sobrevivientes de la cirugía rectal.