INTRODUCTION
Nutte described the first case of contact urticaria with latex in 1979 1 in a housekeeper who used natural rubber latex globes working at home. Since then specific-IgE mediated allergy to latex has been considered an important growing health problem because of the increase of his frequency, the severity of the reactions and the fact of being considered as an occupational disease 2-4, that includes risk groups of patients as natural rubber industry workers and health care workers as the most importants 5-7, but hairdressers 8, cookers and other professionals who use latex products at work also.
Another very important risk group is children with great and exposure to latex products in early childhood, especially those with spina bifida 9,10 and other urogenital disorders that had undergone multiple operations. Mechanical ventilation 11 and atopy 12 had been described as risk factors for latex allergy too.
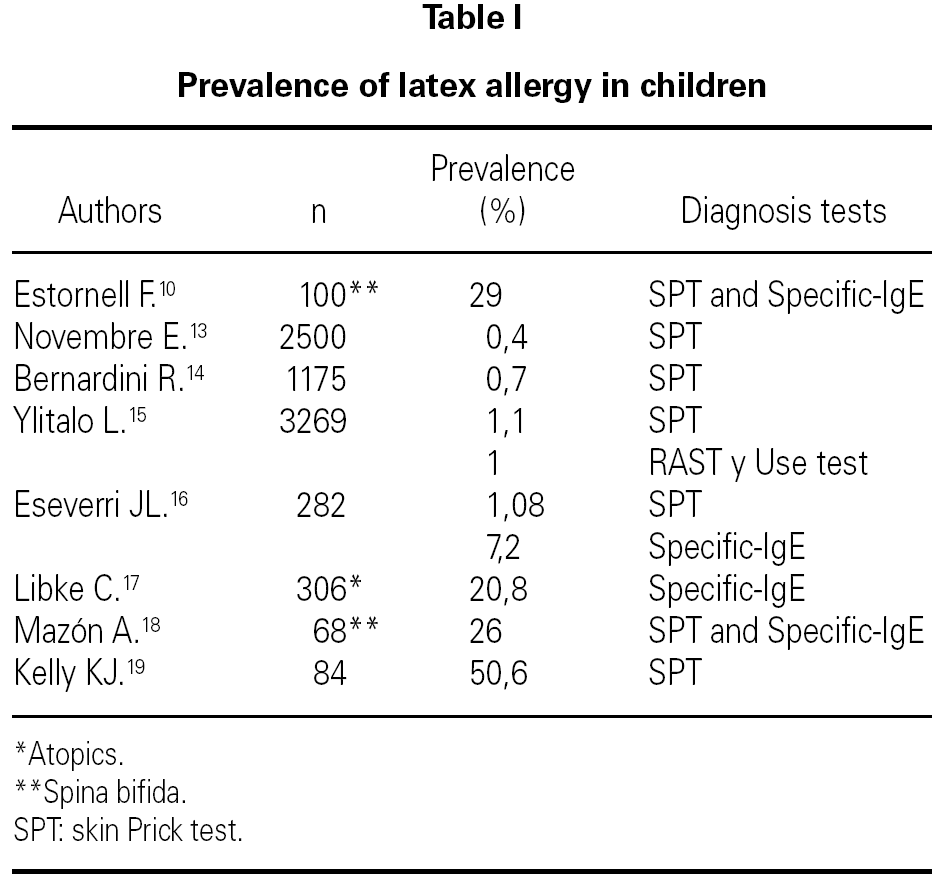
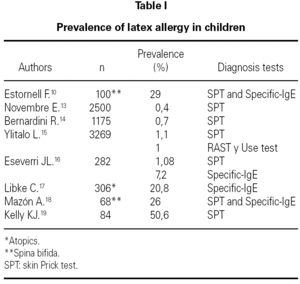
The prevalence of natural rubber latex (NRL) allergy in children is very different depending on the diagnostic tests that were used for the authors and the patients included in the study 13-19 (table I).
Several studies were realized in spina bifida patients finding prevalence from 26 % to 50 % 18,19.
A lot of aspects of this disease are still unknown; one of them is the clinical evolution of those children.
OBJECTIVE
The objective was the clinical and epidemiological evaluation of NRL allergy and latex avoidance instructions in our allergic children.
METHODS AND PATIENTS
Selection of patients
17 consecutive patients were selected from the children who came to our hospital from December 1999 to June 2000, with a clinical history of latex allergy, fruit allergy or chronic urticaria. We realized them:
A prick test with latex commercial extract (concentration 1/10 w/v).
Determination of specific- IgE antibodies to latex by CAP Pharmacia system.
Classification of patients
The patients were classified in two groups following the next criteria:
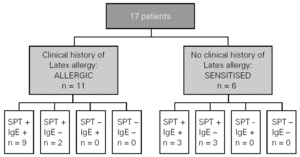
Those children with one positive test (SPT or specific IgE) and a positive clinical history of latex allergy were considered allergic.
Those with one positive test and without a suggestive clinical history of allergy to latex in fact they tolerate currently contact with latex products, were classified as sensitised.
After the diagnosis we gave them oral and written latex avoidance instructions. They were followed up for a period from 1 to 7 years (median of 3 years).
At the end of this period:
We asked them about contacts with latex, at medical and home environment.
We realized them a skin prick test (SPT) with latex commercial extract (concentration 1/10 weight/ volume). Histamine dihydrochloride (10 mg/ml) served as positive control and physiological saline as negative control. A wheal of 3 mm or larger was regarded as positive, according to EAACI guidelines 20.
We realised latex specific-IgE antibodies determination by the CAP Pharmacia system. Specific IgE values greater than 0.35 Ku/L were regarded as positive.
STATISTICS
Median, minimum and maximum values were used to describe continuous variables, and total and relative frequencies to describe categorical variables. Chi-square test or Fisher's exact test was used to compare the population of allergic and sensitised. Wilcoxon matched-pair test was used to compare the results of SPT and latex specific-IgE at the diagnosis and at the end of the follow up. The patients who had undergone multiple operations and those who did not have, allergic and sensitised children were analysed separately. The Mann-Whitney U test was used to compare the SPT and specific-IgE levels at the end of the study in children who had contact with latex after the diagnosis and those who did not have. P-values below 0.05 were considered significant.
RESULTS
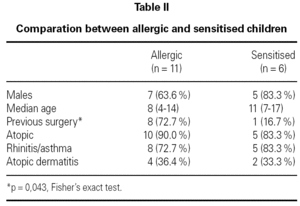
We studied the clinical and epidemiological characteristics of our patients. Twelve of our children were males (70.6 %) and 5 were females (29.4 %). The median age was 10 years (range 4 to 17).
Nine of the patients (52.9 %) had undergone surgery before the diagnosis of sensitisation to latex; the median of the number of operations was two. One of these 9 children was a patient with spina bifida.
Fifteen children (88.2 %) were atopic (respiratory allergy, food allergy or atopic dermatitis). Thirteen patients (76.5 %) had rhinitis and asthma due to pollen hypersensitivity, and 5 (29.4 %) had respiratory allergy to animals epithelium.
Atopic dermatitis was present in 6 of the 17 children (35.3 %) and 11 (64.7 %) had fruit allergy (kiwi, banana, avocado) or nuts (chestnut, hazelnut).
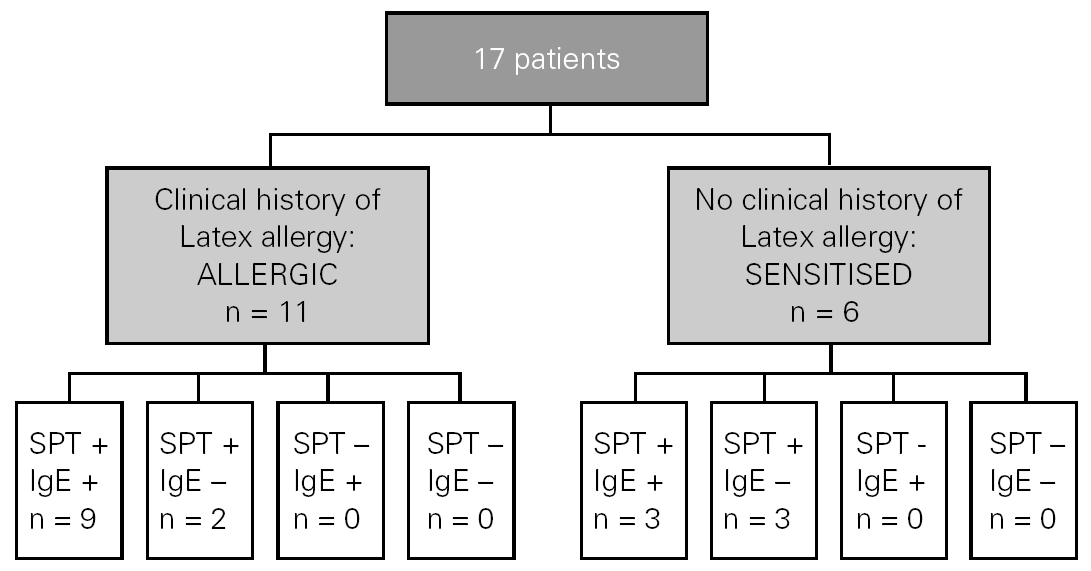
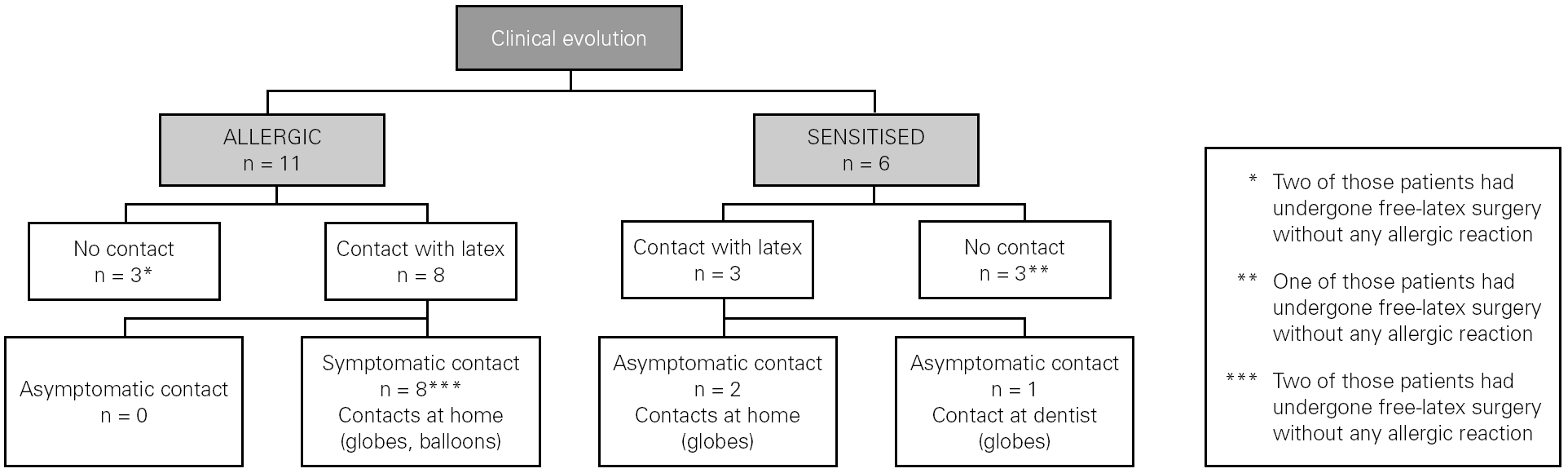
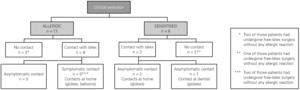
Eleven patients (64.7 %) were diagnosed as allergic to latex, and 6 (35.3 %) as sensitised, with the criteria described before (fig. 1). All the patients who referred a clinical history of immediate reactions after contact with latex had a positive SPT to latex extract.
Figure 1.--Clasification of the patients and diagnostic tests results. SPT: skin prick test to latex; IgE: specific-IgE antibodies to latex by CAP Pharmacia system.
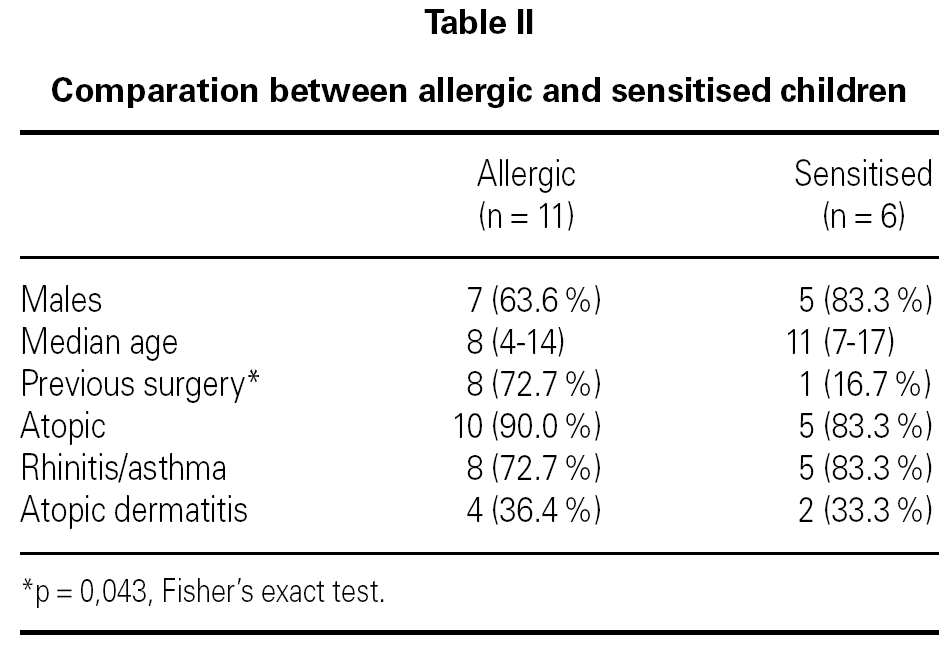
We did not find significant differences in sex, atopic dermatitis, allergic rhinitis or asthma and food allergy between allergic and sensitised patients. The number of patients with history of multiple operations before the diagnosis was significantly higher in allergic than in sensitised children (p = 0.043, OR 13.3 (95 % confidence interval 1.07-166.4) (table II).
The age of the allergic children was higher than the sensitised group, but this difference did not was statistical significant.
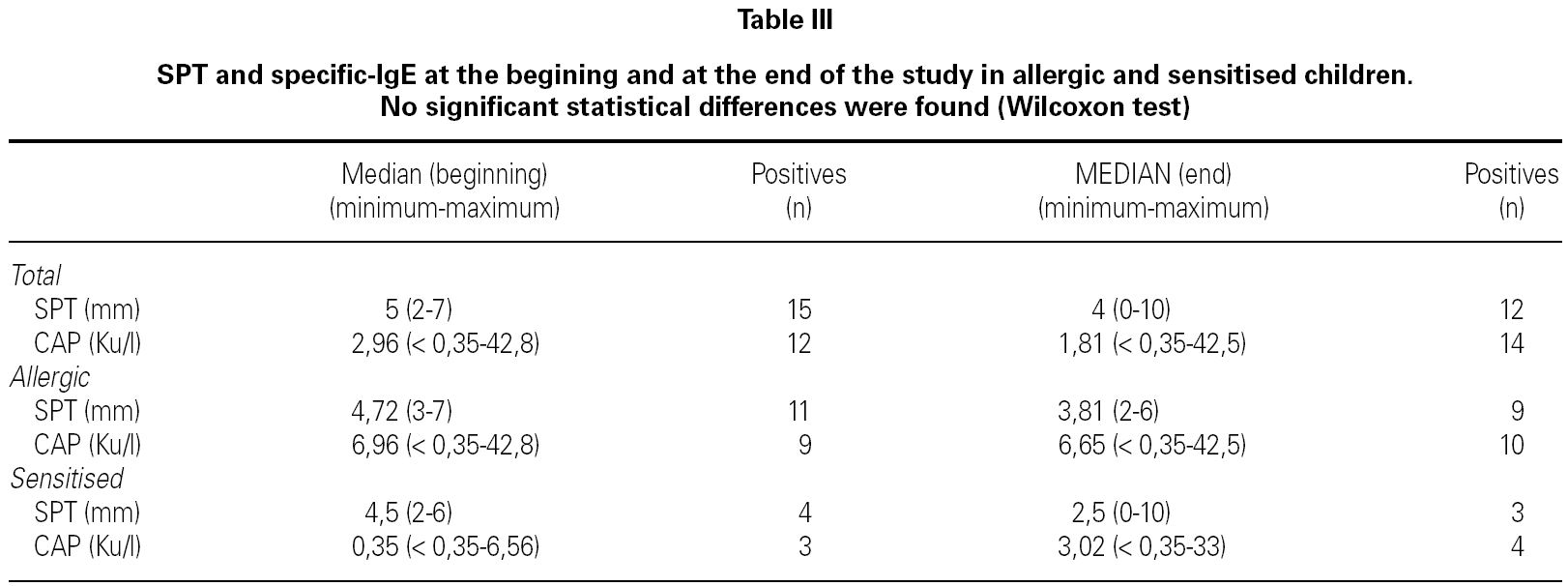
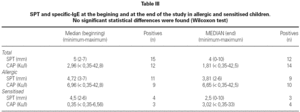
No differences were found between the results of the SPT and latex specific-IgE obtained at the beginning and at the end of the study. We analysed separately three groups: patients who had a history of multiple operations, allergic and sensitised and no differences were found in each group (tables III and IV).
During the follow up 5 patients had undergone free-latex surgery after the diagnosis without any allergic complications (4 were allergic and 1 sensitised).
The frequency of latex contacts was higher in allergic (72.7 %) than in sensitised (50 %) (fig. 2). Only 3 out of the allergic patients completely avoided latex products. In two of them the SPT and specific-IgE levels decreased, but only one had a negative SPT to latex at the end of the study. The third patient did not have relevant changes in his diagnostic tests.
Figure 2.--Clinical evolution of the patients during the follow-up.
The other allergic patients, 8 (73 %), had contact with latex; all of them were contacts at home (gloves, balloons) and all of them were symptomatic. The symptoms were urticaria and angioaedema.One of these 8 patients had a negative SPT to latex at the end of the study, but a positive determination of specific-IgE antibodies. Half of the sensitised patients did not complied with the latex avoidance instructions: one of them came to the dentist and did not notified and he did not have any allergic reaction with the contact with latex gloves. The level of specific IgE antibodies to latex of this patient was greater at the end of the study. The other two patients had latex contact at home, having urticaria and angioaedema. This two sensitised children became allergic during the follow up; the values of SPT and specific-IgE increased in this two patients. The other 3 sensitised children complied the latex avoidance; none of them had significant changes in their diagnostic tests.
Specific-IgE to latex was significantly higher at the end of the study in those patients who had contact with latex (specific-IgE median 0.54 Ku/l) compared to those who completely avoid latex (specific-IgE median 4.86 Ku/l) (p = 0.007, Mann-Whitney U test). No differences were found in SPT values.
DISCUSSION
Children with spina bifida and other urogenital disorders requiring multiple operations, are one of the most important risk group to natural rubber latex allergy 9,10.
In our study surgery in early childhood increased the risk for having clinical latex allergy instead of being sensitised (one positive test and no symptoms with latex). We did not find differences between allergic and sensitised in age, sex, respiratory allergy, atopic dermatitis, food allergy. Atopic dermatitis has been described several times as a risk factor to latex allergy; but we did not find evidence of this maybe because of the small sample.
Several recommendations for preventive measures had been published in the last years 21-23. Some studies had evaluated their effectiveness.
In two studies realised in a healthcare workers population with latex hypersensitivity, a decrease of IgE to latex levels after changing latex gloves in the hospital environment was found 24,25.
There are other studies that evaluate not only primary but secondary prevention too in multioperated children. Cremer et al 26, did not find a decrease of specific IgE in children with spina bifida despite of avoidance instructions.
The authors Ylitalo y Turjanmaa 27, in 2000, included in their study patients with spina bifida and patients without previous surgeries. They described the difficulty for children to completely avoid latex and concluded that secondary prevention did not decreased e the levels of the diagnostic tests. An explanation could be the latex contacts at home. They recommend paying more attention to home environment.
Nieto et al 28, two years later, concluded that primary prevention measures in children with spina bifida decreases the prevalence of latex allergy.
The same year another study including patients with spina bifida and other neurologic defects concluded that secondary prevention is effective and that more stress must be put on the avoidance of latex in the medical setting than out 29.
In our study the hospital was a safety free-latex environment, in fact 5 children had undergone surgery without any allergic problem. The specific-IgE levels of those patients did not change significantly. Despite the avoidance instructions, 11 of the patients (64 %) had contact with latex at home, and in all specific-IgE levels increased, so this latex exposure in home environment could be responsible of maintaining the latex hypersensitivity in those children, as it was mentioned in previous studies 27.
About the other patients, SPT and IgE antibodies did not change significantly during the study, neither between allergic and sensitised patients. Ylitalo et al, in their study of the year 2000, did not find differences in those tests between symptomatic and asymptomatic patients too. More studies are needed to clarify the role of the prick test and the specific IgE in the follow up of children with latex IgE allergy.
After the diagnosis the main avoidance instructions point out the importance of avoid latex exposure in medical setting, because an important allergic group is multioperated patients and several times they need continuous medical assistance. In our paper only one patient belong to this group, so in our patients home environment became the most important exposure place to prevent.
In conclusion, the efficacy of primary prevention has been demonstrated in previous studies, and must be used as the first step in risk population. About secondary prevention, we must care about medical environment because latex is a very important allergen at the hospital. According to our study we must put more stress on home and school setting, in order to reduce those NRL contacts, that could maintain latex hypersensitivity in some patients.
GRATEFULNESS
This study has been founded by Maphre grant (1999), a Spanish Allergy and Clinical Immunology Society grant (SEAIC, 2000), and a grant from the Comunidad de Madrid (08.7/0001/1999.1).
Correspondence:
M.ª Carmen Diéguez Pastor
Laboratorio de Alergia (planta 2.ª derecha)
Hospital Ramón y Cajal.
Ctra. de Colmenar, Km 9.100. 28034 Madrid
Teléfono: 913 368 341; 654 392 450