Atopic eczema (AE) affects approximately 20% of children in Northern countries. Onset during early infancy is common and is characterised by altered skin barrier, increased water loss and defective lipid layer. Restoration of skin barrier by emollients and/or oil baths is an important part of AE treatment, but its role in preventing xerosis and AE is unknown.
The present pilot study aimed to assess if xerosis, and possibly AE, could be reduced at six months of age by early introduction of frequent oil baths/facial fat cream in infants with dry skin.
MethodsA controlled intervention pilot study included 56 six-week-old infants with xerosis, but not AE. Skin quality score ranging from 0 (normal skin) to 4 (probable AE), was assessed at inclusion, three and six months of age, with skin quality at six months as main outcome. One well baby clinic was recruited for intervention, frequent skin care (oil bath (0.5dl) and facial fat cream, five well baby clinics recruited for observation only.
ResultsThe intervention group (n=24) had more often normal skin (75%) at six months than the observation group (37.5%) (p<0.001), and less often probable AE (4.0 vs. 19.0%, respectively, ns). Oil baths were performed regularly, 2–4 up to 5–7 times/week in the intervention group, vs. fewer oil baths with sparse volume of oil in the observation group. No adverse reactions were reported.
ConclusionRegular oil baths in infants seem to reduce xerosis and may possibly reduce atopic eczema.
Atopic eczema (AE) is a chronic, itching and disabling disease that affects about 20% of children in Northern Europe.1–3 The onset is usually early in life, with 45% of cases reported during the first six months.4 AE is partly determined by genetic factors such as Filaggrin gene mutations,5 but life style and environmental factors may play a role, given the rapidly increasing prevalence during the last decades,6,7 and the higher prevalence in northern countries than for example in Mediterranean countries and in the tropics.6,8
Xerosis (dry skin) is a common feature of AE, and the pathogenic mechanisms involve altered skin barrier in affected as well as apparently normal skin. The abnormalities found in the stratum corneum involve increased water loss through the skin 9 and reduction in total skin surface lipid.10 Normal desquamation of the stratum corneum depends on pH dependent proteases with normal function at low pH.11 Elevated pH of the stratum corneum increases serine protease activity, with secondary generation of inflammatory cytokines and reduced activity of lipid-processing enzymes, resulting in a defective lipid layer.11 A Western lifestyle with excessive use of soap and water and skin care products may change the pH of the skin surface in addition to changing the hydration of the skin, thereby influencing the barrier function of the skin.
In established AE anti-inflammatory treatment, moisturising and lipid replacement therapy are well-known strategies documented in different guidelines.11,12 The increase in AE observed in the recent decades, however,6,7 calls for primary preventive strategies, but none have so far been convincingly successful. The effect of probiotics is still controversial,13 and the effect of emollients for primary prevention of AE is not clear.14 Theoretically the use of oil-baths from the neonatal period, being common skin care for AE, could strengthen the skin barrier and potentially reduce AE development in infants. Prior to testing this strategy, the feasibility of skin care among infants and the potential for reducing AE should be explored.
The primary aim of the present pilot study was to investigate if lipid skin care by regular oil baths and use of fat emollients in infants with dry skin from the age of six weeks could reduce xerosis by six months of age, and secondarily if regular oil baths were feasible.
Subjects and methodsA controlled intervention pilot study recruited six-week-old babies with dry skin in well baby clinics to intervention with skin care or to observation until six months of age.
Six well baby clinics in different parts of the health region of Fredrikstad in the east part of Norway were contacted and agreed to recruit babies. Due to the risk of maternal cross-talk reducing differences in skin care within any well baby clinic, we chose to have interventions in one clinic with observation in the remaining five clinics serving as controls. The largest well baby clinic was chosen for recruiting babies for the intervention, and the remaining five clinics included control children.
SubjectsDuring May to October 2011 the public health nurses successively recruited healthy children with dry skin during the regular six-week visit or at a weighing visit at four to five weeks of age. The inclusion criterion was: Presence of dry skin at the age of four to six weeks. Exclusion criterion: Dry skin with signs of scratching or inflammation.
Skin careParents of the babies in the intervention group were asked to provide a daily oil bath in addition to using a fat emollient (Ceridal®) for the face. The public health nurses provided the families with bath oil (paraffin and emulsifier) monthly, with instructions to use 0.5dl bath oil per day in approximately eight litres of warm, but not hot water, for at least 10min. Parents were encouraged to avoid the use of soaps, as well as woollen clothing next to the skin, common advice for all children with dry skin, not related to the present study.
In the observation group the nurses were encouraged to follow their normal procedure for skin care.
All subjects attended monthly visits, with skin examination at inclusion, three and six months of age, carefully performed by the public health nurses.
Skin assessment and skin scoreInfants were classified into the following mutually exclusive categories: normal skin, dry skin or probable atopic eczema. The skin was regarded normal also in the case of minor localised dry patch in the cheek in wintertime only. A semi-quantitative skin score (0–4) was made for evaluation of the skin, and applied to each child; generalised normal skin (0), patches of dry skin affecting less than 50% of the body (1), dry skin affecting approximately 50% of the body with substantial normal areas in between (2), generalised dry skin (3). Any degree of dry skin with observed scratching was defined as probable eczema (4), as there is no established definition of incident case of AE with recent onset.15
All participating nurses were instructed and assessed by the principal investigator to evaluate normal vs. dry skin, as well as signs of established eczema. The training took place in all well baby clinics.
The primary outcome was primarily absence of dry skin, and secondarily the presence of probable AE at the age of six months.
Feasibility was assessed by the reported number of oil baths per week.
The study was approved by the South-East Regional Medical Ethics Committee, verbal and written information was given to the parents, and written informed consent was obtained prior to inclusion.
Statistical analysesBaseline characteristics are reported as mean with standard deviation (SD) or mean and 95% confidence interval (CI) for continuous variables, whereas categorical are given as counts with percentage (%). For comparisons of semi-quantitative measures (skin score) we applied the unpaired Welch test.16 The statistical significance level was set to five % (p<0.05) and the Statistical Programme for Social Sciences (SPSS inc; Chicago, Ill, USA), version 17 was used for the statistical analysis.
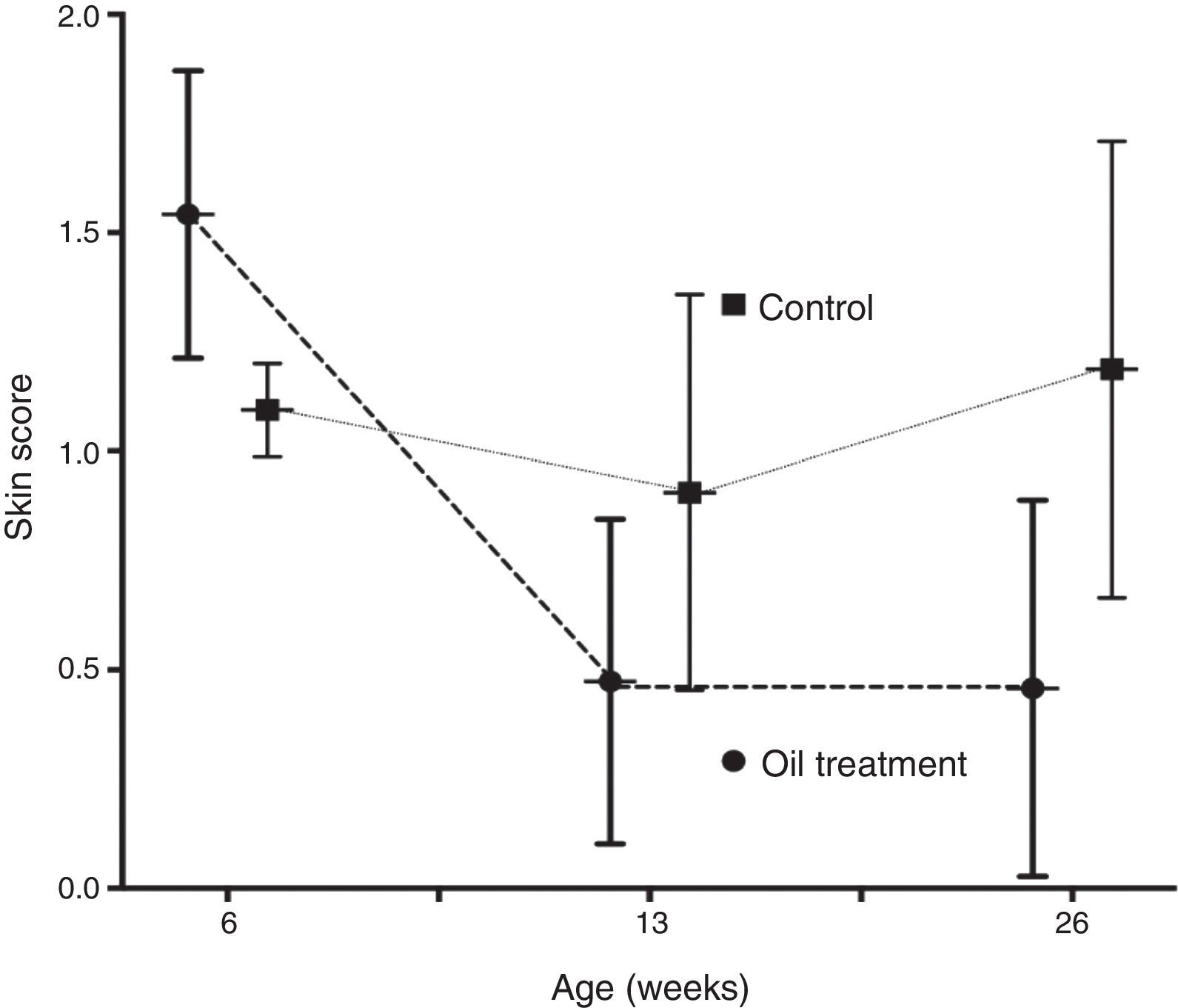
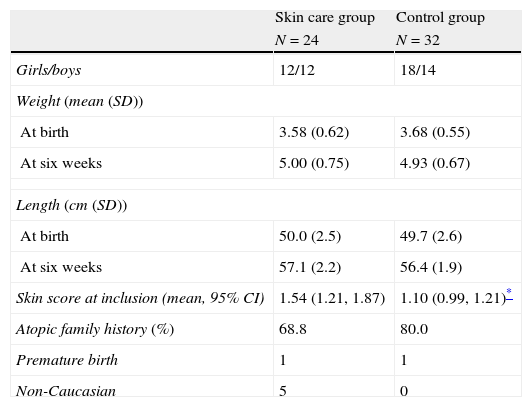
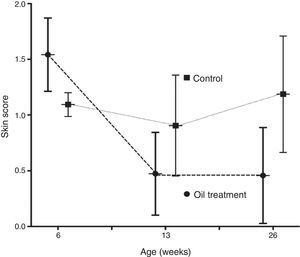
ResultsThe 56 recruited infants (26 boys) had a mean age of 46.5 days on enrolment, with no significant differences in gender, family history of atopy or premature births between the 24 children in the intervention group and the 32 control children. Five children in the intervention group were non-Caucasian; all others were of Caucasian origin (Table 1). All children had dry skin without itching (skin assessment categories 1–3) at inclusion, but with a significantly more widespread xerosis in the skin care group than the observation group. (1.54 vs. 1.10) (p=0.02).
Characteristics of the children in the skin care group vs. control group at six weeks of age. All children had dry skin and none had itching dry skin (probable AE) at inclusion (inclusion and exclusion criteria, respectively).
| Skin care group | Control group | |
| N=24 | N=32 | |
| Girls/boys | 12/12 | 18/14 |
| Weight (mean (SD)) | ||
| At birth | 3.58 (0.62) | 3.68 (0.55) |
| At six weeks | 5.00 (0.75) | 4.93 (0.67) |
| Length (cm (SD)) | ||
| At birth | 50.0 (2.5) | 49.7 (2.6) |
| At six weeks | 57.1 (2.2) | 56.4 (1.9) |
| Skin score at inclusion (mean, 95% CI) | 1.54 (1.21, 1.87) | 1.10 (0.99, 1.21)* |
| Atopic family history (%) | 68.8 | 80.0 |
| Premature birth | 1 | 1 |
| Non-Caucasian | 5 | 0 |
Family history was considered atopic if at least one parent or sibling reported atopic eczema, allergic rhinitis, asthma, or food allergy.
At six months of age the skin care group had significantly more often normal skin (75.0 vs. 37.5%, p=0.01) compared to the control group.
Probable atopic eczema was observed in one child (4%) in the skin care group compared to six infants (19%) in the observation group, (p=0.10, non-significant).
The mean skin score improved significantly from six weeks through three and six months in the intervention group only (p<0.0001 and p<0.0002, respectively). In the observation group the improvement in skin score was non significant (p=0.11 and p=0.12) (Fig. 1).
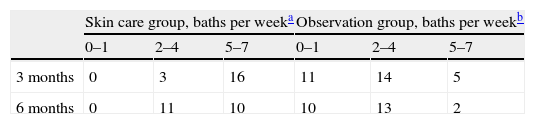
Feasibility of skin careThe skin care group reported significantly more use of oil baths than did the observation group. All children in the skin care group reported at least 2–4 baths per week during the entire study period, at three months 16 out of 24 reported 5–7 baths per week, while at six months 11 out of 24 reported the same.
In the observation group 25 and 21 parents reported use of oil baths at three and six months, respectively, but with oil volume mainly ranging from a few drops to 10ml, 1–7 times per week, mostly 2–4 (Table 2).
No adverse reactions were reported.
DiscussionThe present pilot study demonstrated that regular and frequent oil baths from six weeks of age were associated with significant reduction of dry skin at six months of age, with a further tendency to less probable atopic eczema at six months.
The present study is to our knowledge the first demonstrating reduction of xerosis and achievement of xerosis-free skin in infants by use of frequent oil baths as part of skin care. However, skin care in infants with xerosis is not well documented, although our findings are supported by the advocacy of lipid replacement therapy in the treatment of AE.11 Furthermore, in line with the present pilot study, a recent pilot study on primary prevention of AE using petrolatum-based emollients indicated a probable positive effect on reducing atopic eczema among 20 infants. The study did not include a control group, thus no conclusion could be made.14
Although our pilot study was not a primary prevention study as the inclusion criterion was dry skin at six weeks, it indicated the possibility that oil baths may be beneficial also to prevent atopic eczema development. Due to the limited number of subjects in the pilot design, further studies are needed.
The present study demonstrated feasibility of using regular oil baths, and a reduction in xerosis for a length of time in infants without eczema. However, the study had several limitations. First, the study was a pilot study conducted to assess the potential effect of intervention, and thus not powered to assess efficacy in primary prevention of atopic eczema. Secondly we used no stringent doctor diagnosis of eczema or skin quality according to established criteria. A skin score was made for the purpose of having a semi-quantitative assessment, which could be performed by the nurses in the well baby clinics, meaning that all results and apparent significances have to be considered carefully. In addition, the nurses were not blinded to the intervention and observation, a serious limitation, which could clearly affect the evaluation. Thirdly, we did not measure the amount of oil used and frequency of baths, apart from parental reports during well baby visits. Finally, the parents in the intervention group were encouraged to avoid soap and woollen clothing for the infants, normal recommendations to avoid skin irritation,12 but might be looked upon as an intervention, and not evaluated.
The demographic data show a difference in ethnicity, but as the immigrant families were in the intervention group, this could be regarded as a strength rather than a limitation, as atopic diseases are reported more frequently in immigrants.17
The significant difference at inclusion between the groups according to degree of xerosis could as well be regarded as a strength rather than a limitation, as the intervention group was the one with the most widespread xerosis.
In conclusion, in spite of the limitations, the present pilot study showed that regular skin care, with a focus on oil baths of infants, was feasible, reduced xerosis, and may be a potential means for primary prevention of atopic eczema.
Ethical disclosuresProtection of human subjects and animals in researchThe authors declare that the procedures followed were in accordance with the regulations of the responsible Clinical Research Ethics Committee and in accordance with those of the World Medical Association and the Helsinki Declaration.
Confidentiality of dataThe authors declare that they have followed the protocols of their work centre on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects mentioned in the article. The author for correspondence is in possession of this document.
Participation of the co-authorsProf. Karin L. Carlsen and Prof. Kai Håkon Carlsen participated in the planning of the study, as well as the writing of the manuscript. Dr. Teresa L. Berents participated in planning the skin treatment, skin assessment and writing of the manuscript. Petter Mowinckel assisted with statistical analyses and writing of the manuscript.
Conflict of interestThere is no conflict of interest of any of the authors.
We are very grateful to all the public health nurses in the six well baby clinics in Fredrikstad who helped us with the study, in addition to their daily work. We are also very grateful to Dr. Suzanne Crowley, Oslo University Hospital, for English correction of the manuscript.