Bovine alpha herpesvirus 5 (BoHV-5) induces neurological disease in cattle, including tremors, nystagmus, teeth grinding, circling, ataxia, recumbence, paddling, and death2,6,7,9. Nonsuppurative meningoencephalitis is a hallmark of this infection. Like the majority of the alpha herpes viruses, it generates latency in trigeminal ganglia5. The latency-related (LR) gene is the main responsible for the maintenance of latency1,3,4,8. To evaluate the LR function within the viral infection, a recombinant virus was constructed deleting the LR promoter to avoid its expression without affecting overlapping genes in the antisense strand (the immediate early bicp0 gene).
The enhanced green fluorescent protein (EGFP) was used as a reporter to facilitate recombinant virus detection in the cell culture plates. For that purpose, EGFP was PCR-amplified from pEGFP-C1 (Clontech™) and subsequently cloned into a plasmid surrounded by selected 5′ and 3′ recombination arms of BoHV-5. The recombinant plasmid was linearized with ScaI and cotransfected with full-length BoHV-5 DNA previously extracted with DNazol® (Invitrogen™) into CRFK (Crandell Feline Kidney) cells plated in a 24-well plate dish at 90% of cell density. Twenty-four hours later cells were reseeded into a 60mm-plate and covered with 1% methylcellulose medium overlaid after their attachment. EGFP positive plaques were identified under fluorescent microscope, isolated and reseeded in a fresh monolayer. Viral clones were purified six times before being aliquoted and frozen at −80°C. The correct site of recombination and removal of the promoter target was confirmed by sequencing of the recombination ends toward the reporter gene. The recombinant virus was denominated BoHV-5 ΔLR +GFP. To our knowledge, this is the first bovine herpesvirus in which the LR gene has been replaced by EGFP, making this recombinant a powerful tool for in vitro and in vivo studies.
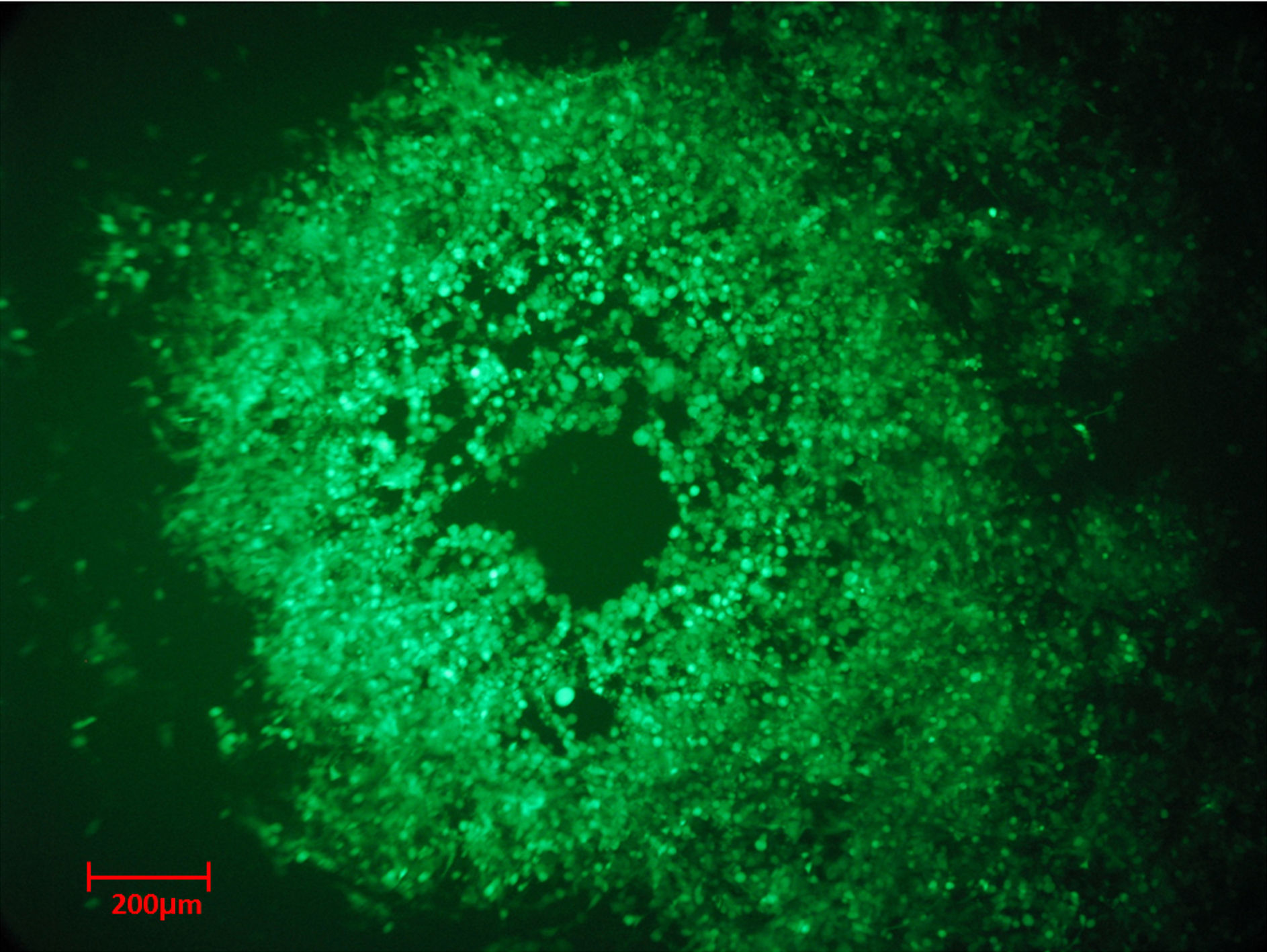
In this section we showed the recombinant BoHV-5 ΔLR +GFP-infecting CRFK cells. Figure 1 shows typical viral plaque morphology. In Figure 2 we used DAPI to contrast the nuclei and to facilitate visualization of cytopathic effects.
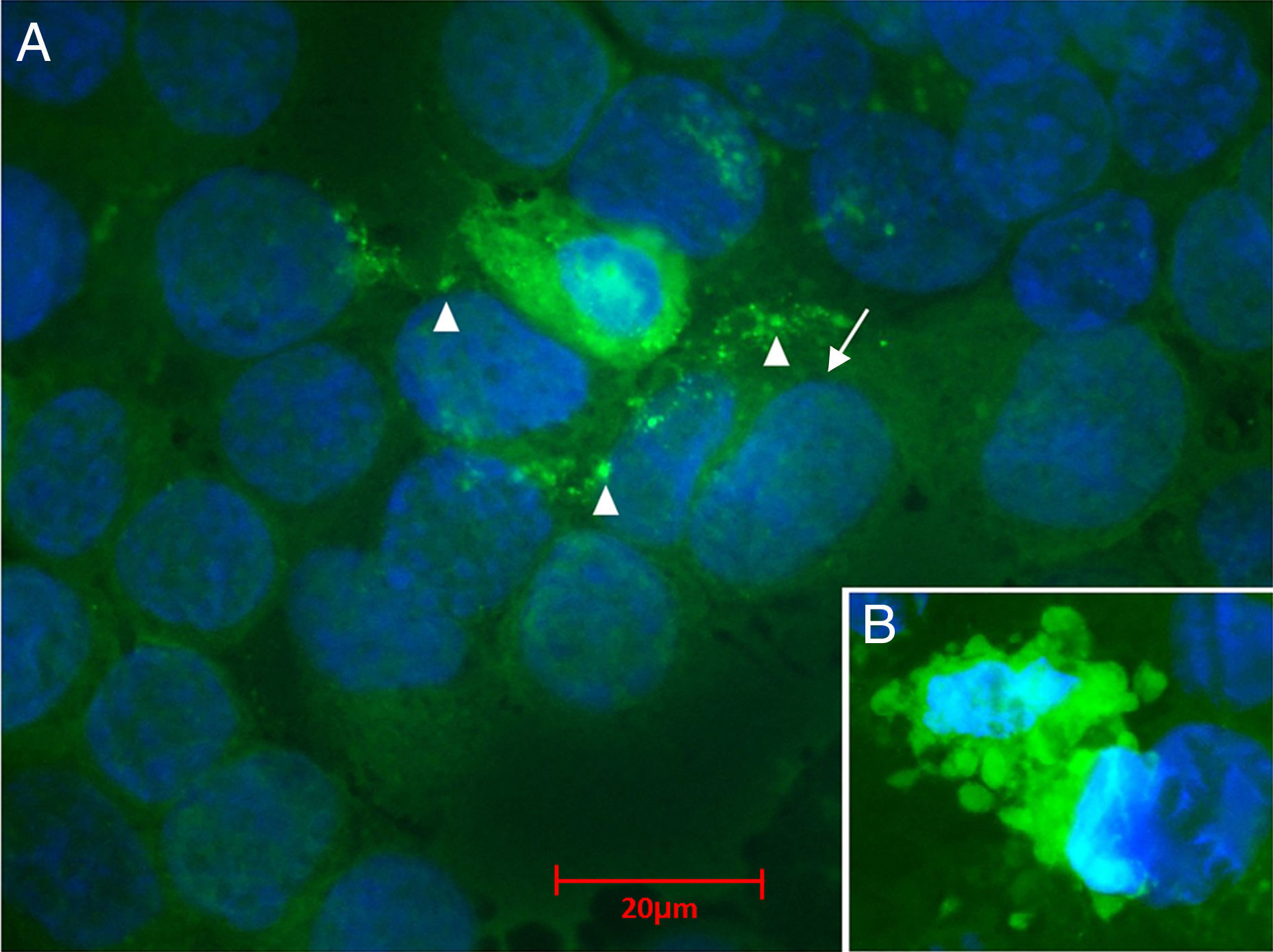
CRFK cells were infected with the BoHV-5 recombinant (ΔLR +GFP) at a MOI of 0.1. Twenty-four hours later they were fixed and nuclear DNA was stained with 4′,6,-diaminodino-2-phenylindole (DAPI) (Invitrogen™). DAPI and GFP expression were monitored under a fluorescent microscope; the obtained images were merged into the same picture using the ImageJ program. (A) Observed expression of GFP in all the cytoplasms of infected cells with the presence of multinucleated cells (arrow), and cytoplasmic inclusion bodies (triangle). (B) Advanced infections showed evidence of apoptosis with cell shrinkage, condensed and pyknotic nuclei and apoptotic bodies.
The authors state that for this investigation no experiments have been performed on humans or animals.
Confidentiality of dataThe authors state that in this article there are no patient data.
Right to privacy and informed consentThe authors state that in this article there are no patient data.
FundingThis research was supported by the Agencia Nacional de Promocion Cientifica y Tecnologica (PICT 2010-0925). CS is recipient of a Fellowship for the conclusion of the PhD degree from the Conicet (Consejo Nacional de Investigaciones Cientificas y Tecnicas).
Conflict of interestThe authors declare that they have no conflicts of interest.
We thank Centro de Virología Animal, Instituto de Ciencia y Tecnología Dr. Cesar Milstein, CONICET, Ciudad de Buenos Aires, Argentina for the use of the fluorescence microscope and Carlos Palacios for his assistance.