To analyze the epidemiological characteristics of heart failure (HF) and estimate the burden of the disease on the health service by means of real world data (RWD).
Patients and methodsAll patients discharged from any Basque Health Service hospital after a first admission for HF between 2011 and 2015.
Data sourcesDatabases of our health service.
Outcomes30- and 365-day admissions, potentially avoidable hospitalizations (PAHs), mortality.
Statistical analysisDescriptive statistics, age-standardized event rates.
ResultsThe cohort was composed of 15,109 patients (mean age 79.8±10.1 years). At discharge patients had a median of 8 chronic conditions. 36% of them had had hospitalizations and 83% had visited a specialist (42% of them a cardiologist) during the previous year. Mortality was 24.5% within 365 days after discharge.
Within 30 days after discharge, there were 2608 unplanned admissions, 49% for non-cardiovascular disease (CVD), 36% for HF and 15% for a CVD other than HF. 34% were classified as PAH. In the first 365 days after discharge, there were 14,559 hospitalizations, 54% for non-CVD reasons, 32% for HF and 13% for a CVD other than HF. Overall, 35% were PAHs.
Conclusion(1) People admitted for HF are old, and they have multimorbidity and high rates of admissions due to non-CVD reasons and PAHs after discharge. These finding suggest the need of strengthening continuity of care and managing comorbidities. (2) Besides, most people admitted for HF have previous contacts with the Healthcare system, which suggests opportunities for prevention before disease worsening.
Analizar las características epidemiológicas de la insuficiencia cardiaca (IC), y calcular la carga de la enfermedad para el servicio sanitario, utilizando los datos del mundo real (DMR).
Pacientes y métodosTodos los pacientes que recibieron el alta hospitalaria en el Sistema Sanitario del País Vasco, tras el primer ingreso por IC, entre 2011 y 2015.
Fuentes de datosBases de datos de nuestro servicio sanitario. Resultados: ingresos de 30 y 365 días, hospitalizaciones potencialmente evitables (HPE), mortalidad.
Análisis estadísticoEstadísticas descriptivas, tasas de episodios estandarizados por edad.
ResultadosLa cohorte se compuso de 15.109 pacientes (edad media: 79,8±10,1 años). Al alta, los pacientes tenían una media de 8 situaciones crónicas. El 36% de ellos habían sido hospitalizados y el 83% habían visitado a un especialista (el 42% a un cardiólogo) durante el año anterior. La mortalidad fue del 24,5%, dentro de los 365 días posteriores al alta.
Dentro del plazo de los 30 días posteriores al alta, se produjeron 2.608 ingresos no planificados: el 49% por enfermedades no cardiovasculares, el 36% por IC y el 15% por enfermedades cardiovasculares (ECV) diferentes a IC. El 34% fueron clasificados como HPE. En los primeros 365 días posteriores al alta, se produjeron 14.559 hospitalizaciones: el 54% por enfermedades no cardiovasculares, el 32% por IC y el 13% por una ECV diferente a IC. En general, el 35% fueron HPE.
Conclusión(1) Las personas ingresadas por IC son mayores, tienen multi-morbilidades y altas tasas de ingresos debidos a motivos no cardiovasculares y HPE tras el alta. Estos hallazgos sugieren la necesidad de reforzar la continuidad de la atención y gestionar las comorbilidades, y (2) Además, muchas personas ingresadas por IC han tenido un contacto anterior con el sistema sanitario, lo cual es sugerente de oportunidades de prevención antes del empeoramiento de la enfermedad.
Data collected during daily practice and other so-called real world data (RWD), that is, gathered outside formal research-related or academic activities, should serve to generate complementary evidence about effectiveness and safety provided by clinical trials.1
Specifically, data from clinical trials are not able to answer all relevant scientific questions and have limited external validity, due to their underrepresentation of some subpopulations, especially some of the most vulnerable groups.2
The main priorities of society include accelerating the knowledge creation and transfer, as well as improving the effectiveness, quality and efficacy of healthcare provision. To make progress in these areas, it is becoming ever more relevant to exploit the data generated during healthcare provision to generate evidence through daily practice, that is, we need to promote an ‘evidence-generating medicine’ approach.3
On the other hand, recent years have seen the digitalization of numerous healthcare management processes and systems. In the case of the Basque Country, the single electronic health record system is fully deployed and 97% of prescriptions are issued digitally. Paradoxically, the health system has yet to show itself able to transform this enormous amount of information into knowledge, and use this knowledge to improve the quality of healthcare. This approach is one of the cornerstones of the concept of a ‘learning health system’, that is, an ideal system in which “science, informatics, incentives and culture are aligned for continuous improvement and innovation, with best practices seamlessly embedded in the delivery process and new knowledge captured as an integral by-product of the delivery experience”.4
Heart failure is a complex clinical syndrome characterized by an inability of the heart to pump enough blood to fully meet the body's metabolic demands. The prevalence of heart failure is increasing due to population aging and longer survival of patients with heart disease. Compared to heart failure prevalence rates of 2% in other European countries and the USA, Spanish studies point to figures around 5%, though the difference is probably partly attributable to methodological flaws in the research.5 Heart failure is already the leading cause of hospitalization in over-65-year-olds and accounts for 3% of all hospitalizations and 2% of healthcare expenditure in developed countries.6 These characteristics -complexity, frequency and severity- make HF an ideal model for learning to manage real world data and draw lessons to be applied in the detailed analysis of any other conditions.
The aim of this article is to describe a cohort created with all patients diagnosed with HF in the Basque Country between January 1, 2011 and December 31, 2015. Although the cohort is dynamic and new cases will be added for 2016 onwards, in this study, we set out to describe the main clinical and sociodemographic characteristics and the history of health service use of the 16,109 people included to date. Based on this cohort, our objectives were to analyze the epidemiological characteristics of the disease by means of RWD, as well as estimate the burden of this disease on the health service at 1 year after a first admission for HF.
Material and methodsStudy population and settingThe Basque Health Service (Osakidetza) is a public organization that provides universal healthcare free at the point of delivery to the population of the Basque Country (approximately 2.15 million). Osakidetza is divided into 13 healthcare regions each covered by an integrated healthcare organization (IHO). Each IHO manages primary and hospital-based care (with at least one referral hospital) for its catchment population, these ranging from 10,800 to 380,000.
The study population is composed of people over 40 years of age discharged after a first admission for HF (principal diagnosis: ICD-9 codes 398.91, 402.01, 402.11, 402.91, 404.01, 404.03, 404.11, 404.13, 404.91, 404.93, and 428.xx) from any of the hospitals of the Basque health network between January 1, 2011 and December 1, 2015. Individuals who died during hospitalization were excluded.
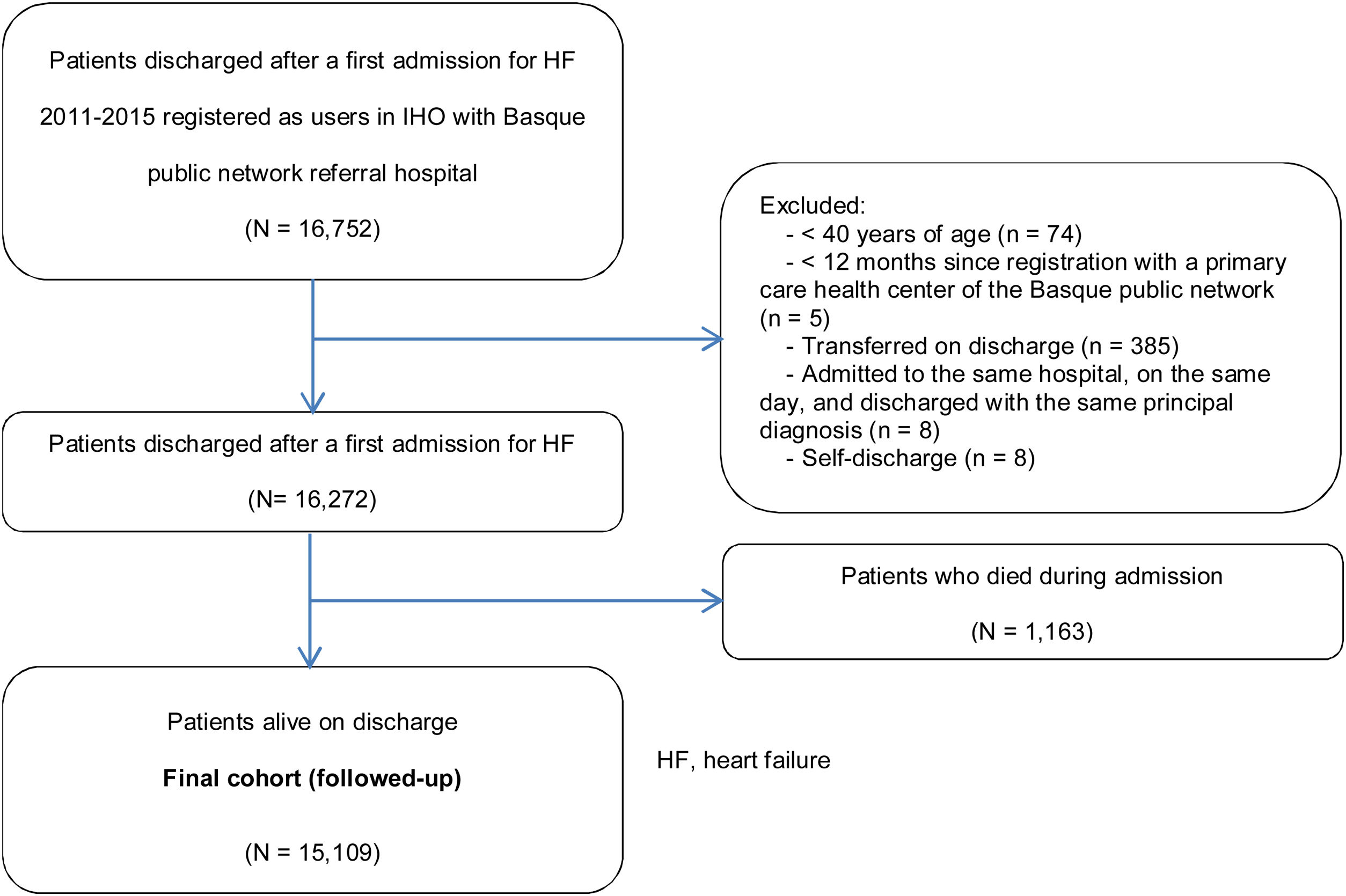
Case identificationThe final follow-up cohort was obtained through the following steps (Fig. 1). We first identified all the hospitalizations for HF between January 1, 1997 and December 31, 2015 (information necessary to estimate the prevalence and incidence of HF). Subsequently, we selected patients whose first admission for HF (index discharge) happened between 2011 and 2015 and who were registered users in an IHO whose referral hospital was part of the Basque public network (i.e., excluding patients whose referral hospital was a private clinic). Exclusion criteria were: (a) age under 40 years old; (b) assignment to the IHO for less than 12 months; (c) transfer on discharge (identified based on the discharge report mentioning transfer to another hospital or admission to a different hospital the same day or the day after discharge); (d) admission to the same hospital on the same day as discharge with the same principal diagnosis; and (e) self-discharge. From this population, the patients who were alive on discharge comprise the final follow-up cohort (n=15,109). In addition, we excluded any cases in which the recorded date of death preceded the date of discharge (i.e., with inconsistent data suggestive of errors in data entry).
Data sourcesData were obtained through the Osakidetza Business Intelligence platform, a web-based visual analytics tool, which enables us to exploit and analyze data from the data warehouse that stores all the structured data considered relevant from the administrative information databases and electronic health records of the Basque Health Service. Hence, we have information available on all healthcare settings: primary care, hospital care (inpatient wards, day hospital, hospital at home, outpatient clinics, emergency services), and laboratory, histopathology and pharmacy services, among others. In this way, the platform gathers most of the information recorded in each encounter between an individual and the Basque public health system and links it with each patient using a pseudo-anonymized identifier. All the information is stored in an unlinked manner, ensuring that researchers do not have access to personal data of the individuals in the cohort. This research has been approved by the Clinical Research Ethics Committee of the Basque Country.
Patient characteristics and study variablesBaseline characteristicsAge, sex, comorbidity, use of health services in the 12 months prior to the index discharge, healthcare region of residence and calendar year
Comorbidity: two different aggregate measures of morbidity based on the ICD-9 diagnoses extracted from both the primary care health and the hospital discharge records:
- -
Charlson comorbidity index and its components of morbidity as adapted by Deyo et al.7
- -
Modified chronic condition classification system developed by Magnan et al.8
This algorithm is based on the Clinical Classifications Software and the Chronic Condition Indicator, both developed by the Healthcare Cost and Utilization Project,9 and identifies 69 clinically relevant chronic conditions. The modified algorithm identifies 71 chronic conditions (Table A1).
Previous use of healthcare servicesHospital discharges, emergency department attendances without hospitalization and specialized and primary care consultations in the 12 months prior to the index discharge.
Adverse events and disease burden on the health serviceMortality, unplanned admissions, potentially avoidable hospitalizations (PAHs) within 30 days and 1 year after the index discharge and length of stay. We distinguish admissions for the HF itself, from those for another cardiovascular disease and those for reasons unrelated to cardiovascular disease. All PAHs are defined and grouped according to the criteria published by Caminal et al.,10 except for PAHs due to HF, for which a more specific definition from the medical practice variations project was chosen (Atlas de Variaciones en la Práctica Médica).11 Both studies offer validated PAH criteria to Spanish context and are widely used.12,13
Statistical analysisBaseline characteristics: These are expressed as percentages for categorical data, medians and interquartile ranges for continuous, non-normally distributed variables and mean and standard deviations (SDs) for continuous normally-distributed variables.
Prevalence and incidence of the disease: Prevalence rates were calculated for men and women and by IHO as of January 1, 2016. The numerator is all live patients ≥40 years of age with at least one admission for HF since 1997 and the denominator the Basque population ≥40 years of age after excluding individuals living in geographical areas whose main referral hospital is not part of the Basque public network of hospitals.
The annual cumulative incidence for men and women was calculated by dividing the number of first discharges after admissions for HF per year in patients ≥40 years of age divided by the Basque population ≥40 years of age at the beginning of each year. The age-standardized incidence rates were calculated for 5-year periods using the direct method and taking the total population of the Basque Country as of January 1, 2015 as a reference.
As a measure of variation between IHOs, we used the systematic component of variance (SCV), which measures variation in the deviation between the observed and expected rates, expressed as a percentage of the expected rate. The greater the SCV, the greater systematic variation (variation not likely due to chance).14
Disease burden on the health service: The total rate of unplanned admissions, rate of PAHs and length of hospital stay were calculated for each sex. Patterns in mortality by sex were explored using survival curves and hazard functions. To assess differences by calendar year, negative binomial models adjusted for age and level of comorbidity (Charlson index) were constructed for the rates of 30- and 365-day admissions and logistic regression models for mortality rates.
All statistical analyses were conducted using R version 3.3.2.
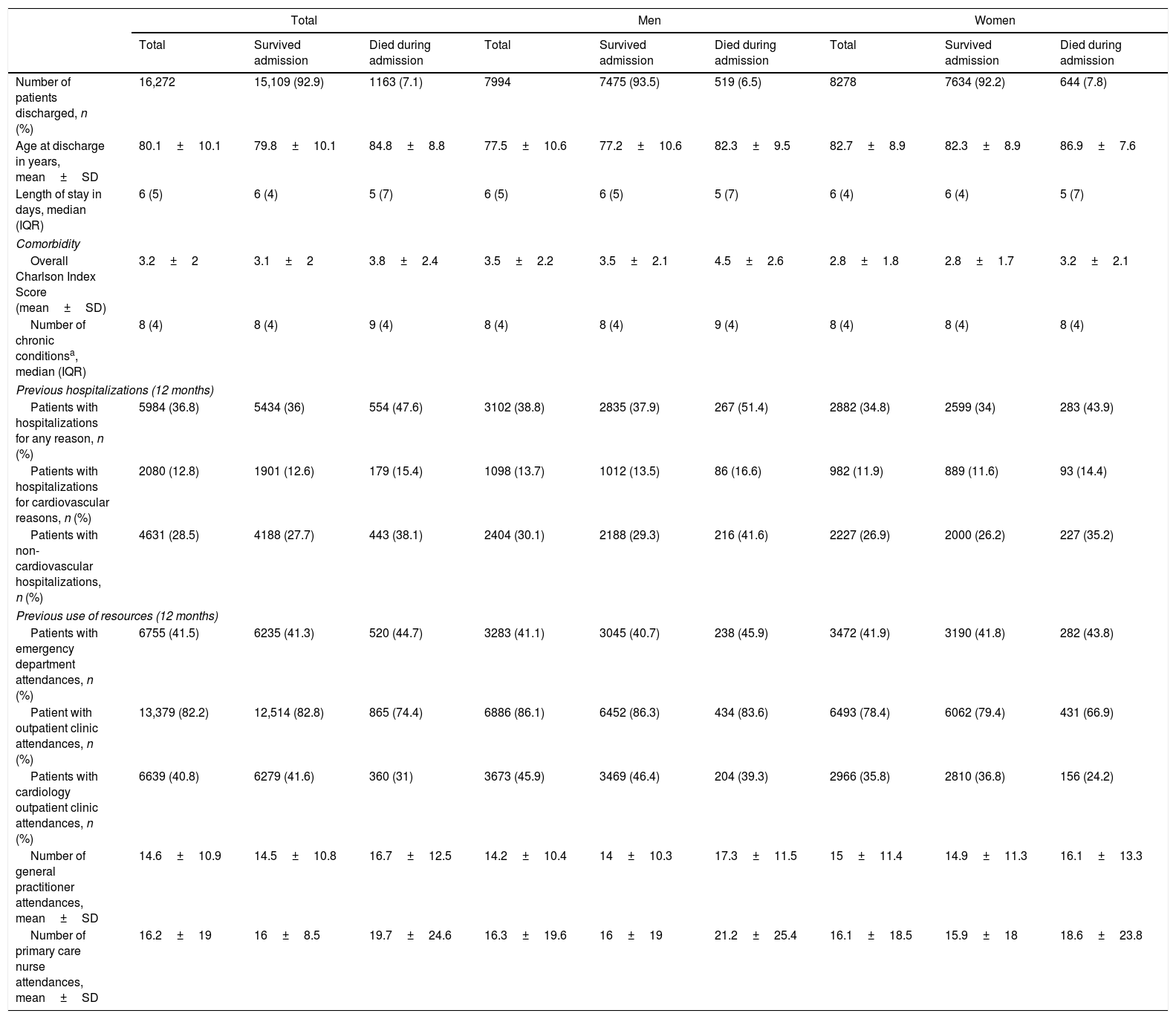
ResultsBaseline characteristics of the cohortA total of 16,272 people were discharged after a first admission for HF during the study period, of whom 1163 died during hospitalization. Those who died during the index admission were older than those who survived (84.8 years; SD=8.8 vs. 79.8 years; SD=10.1). In both men and women, the probability of dying increased with age and with comorbidity status measured by the Charlson index: 5.5% if 1–2; 7.4% if 3–4; and 10.3% if 5 or greater. Overall, crude mortality during the index admission was greater among women. Those who died during admission had more hospitalizations in the previous 12 months (Table 1).
Baseline characteristics of patients discharged after a first admission for heart failure between 2011 and 2015.
| Total | Men | Women | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | Survived admission | Died during admission | Total | Survived admission | Died during admission | Total | Survived admission | Died during admission | |
| Number of patients discharged, n (%) | 16,272 | 15,109 (92.9) | 1163 (7.1) | 7994 | 7475 (93.5) | 519 (6.5) | 8278 | 7634 (92.2) | 644 (7.8) |
| Age at discharge in years, mean±SD | 80.1±10.1 | 79.8±10.1 | 84.8±8.8 | 77.5±10.6 | 77.2±10.6 | 82.3±9.5 | 82.7±8.9 | 82.3±8.9 | 86.9±7.6 |
| Length of stay in days, median (IQR) | 6 (5) | 6 (4) | 5 (7) | 6 (5) | 6 (5) | 5 (7) | 6 (4) | 6 (4) | 5 (7) |
| Comorbidity | |||||||||
| Overall Charlson Index Score (mean±SD) | 3.2±2 | 3.1±2 | 3.8±2.4 | 3.5±2.2 | 3.5±2.1 | 4.5±2.6 | 2.8±1.8 | 2.8±1.7 | 3.2±2.1 |
| Number of chronic conditionsa, median (IQR) | 8 (4) | 8 (4) | 9 (4) | 8 (4) | 8 (4) | 9 (4) | 8 (4) | 8 (4) | 8 (4) |
| Previous hospitalizations (12 months) | |||||||||
| Patients with hospitalizations for any reason, n (%) | 5984 (36.8) | 5434 (36) | 554 (47.6) | 3102 (38.8) | 2835 (37.9) | 267 (51.4) | 2882 (34.8) | 2599 (34) | 283 (43.9) |
| Patients with hospitalizations for cardiovascular reasons, n (%) | 2080 (12.8) | 1901 (12.6) | 179 (15.4) | 1098 (13.7) | 1012 (13.5) | 86 (16.6) | 982 (11.9) | 889 (11.6) | 93 (14.4) |
| Patients with non-cardiovascular hospitalizations, n (%) | 4631 (28.5) | 4188 (27.7) | 443 (38.1) | 2404 (30.1) | 2188 (29.3) | 216 (41.6) | 2227 (26.9) | 2000 (26.2) | 227 (35.2) |
| Previous use of resources (12 months) | |||||||||
| Patients with emergency department attendances, n (%) | 6755 (41.5) | 6235 (41.3) | 520 (44.7) | 3283 (41.1) | 3045 (40.7) | 238 (45.9) | 3472 (41.9) | 3190 (41.8) | 282 (43.8) |
| Patient with outpatient clinic attendances, n (%) | 13,379 (82.2) | 12,514 (82.8) | 865 (74.4) | 6886 (86.1) | 6452 (86.3) | 434 (83.6) | 6493 (78.4) | 6062 (79.4) | 431 (66.9) |
| Patients with cardiology outpatient clinic attendances, n (%) | 6639 (40.8) | 6279 (41.6) | 360 (31) | 3673 (45.9) | 3469 (46.4) | 204 (39.3) | 2966 (35.8) | 2810 (36.8) | 156 (24.2) |
| Number of general practitioner attendances, mean±SD | 14.6±10.9 | 14.5±10.8 | 16.7±12.5 | 14.2±10.4 | 14±10.3 | 17.3±11.5 | 15±11.4 | 14.9±11.3 | 16.1±13.3 |
| Number of primary care nurse attendances, mean±SD | 16.2±19 | 16±8.5 | 19.7±24.6 | 16.3±19.6 | 16±19 | 21.2±25.4 | 16.1±18.5 | 15.9±18 | 18.6±23.8 |
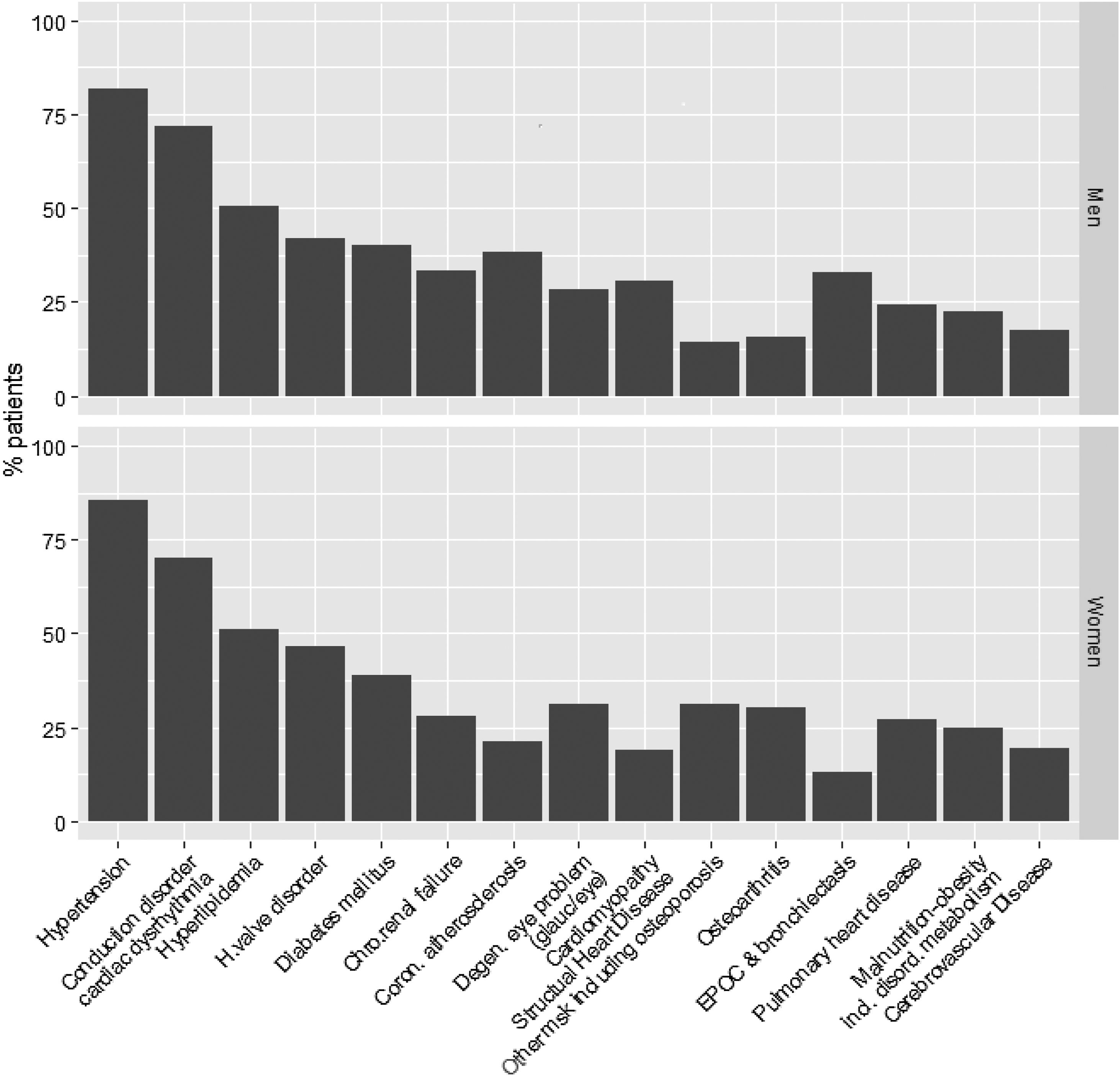
Among the 15,109 patients included in the cohort, health care provider attended a similar number of men and women (men: n=7475 patients, 49.5%; mean age=77.2±10.6 years vs women: n=7634 patients, 50.5%; mean age=82.3±8.9 years). At the index discharge, the patients of our cohort had a median of 8 different chronic conditions. The most common conditions were hypertension (12,355 patients, 81.8%), conduction disorder or cardiac dysrhythmia (10,333 patients, 68.4%) and hyperlipidemia (7199, 47.6%) (Fig. 2; Table A2). We also observed sustained increases in the mean level of comorbidity measured using the overall Charlson index (from 3±1.9 in 2011 to 3.2±2 in 2015; p<0.001) and age at the first discharge after admission for HF (79.1±10.4 in 2011 to 80.4±9.9 in 2015; p<0.001) (Table A3).
A total of 5434 patients in the cohort (36%) had at least one hospitalization in the year prior to the index discharge. Of these 1901 (12.6%) had been admitted for a cardiovascular reason with a principal diagnosis other than HF. Moreover, 6235 (41.3%) had attended an emergency department of the Basque health network at least once and 12,514 (82.8%) had had consultations with specialists, half of these (6279, 41.6%) with a cardiologist. Regarding the use of primary care services, the 15,109 individuals in the cohort attended their health center a mean of 14.5±10.8 times for general practitioner appointments and 16±18.5 times for nursing appointments (Table 1).
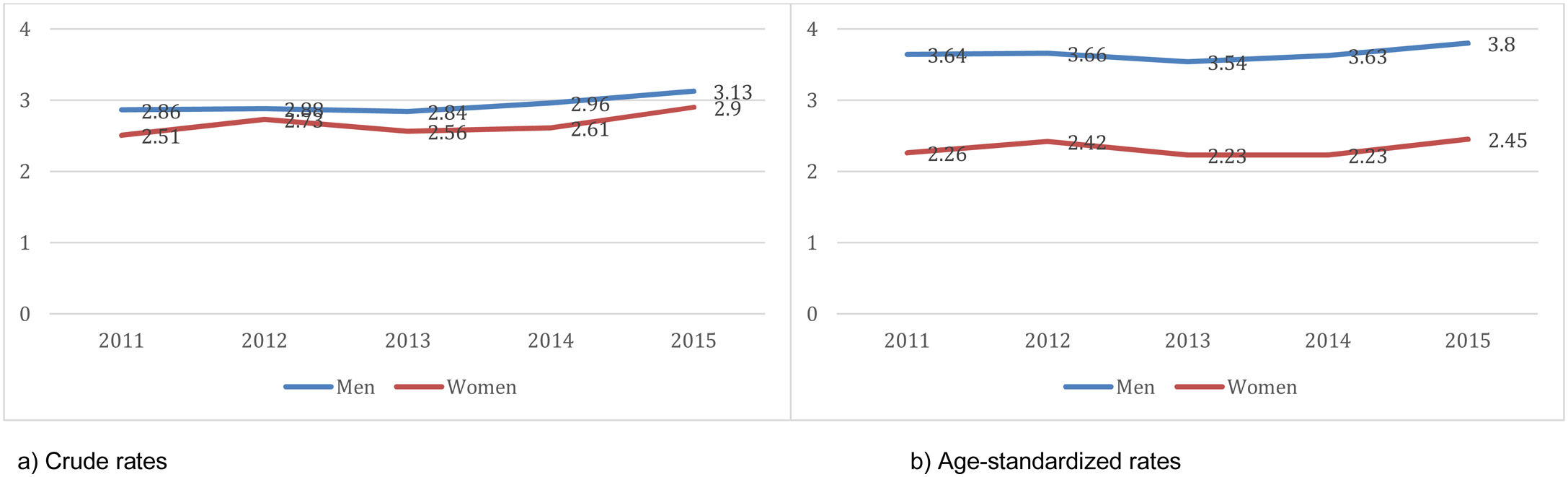
Disease burden based on electronic health recordsAs of December 31, 2015, the number of persons with the disease registered in the information system of the Basque Health System was 15,087, yielding a prevalence of 1.22% among over-40-year-olds (1.28% in men vs 1.26% in women). The prevalence in the different IHOs ranged between 0.78% and 1.33% in women and between 0.93% and 1.43% in men. The age-standardized incidence of first discharges after admission for HF ranged between 2.23 and 2.45 per 1000 women per year in women and between 3.54 and 3.8 per 1000 men per year, in the first years analyzed (Fig. 3). We did not observe any trend (either increasing or decreasing. Ptrend>0.05 in all analysis). Across the geographical area studied, there was little variation in the new discharges after admission for HF (SCV<0.1 in all the years studied, in both men and women; Table A4).
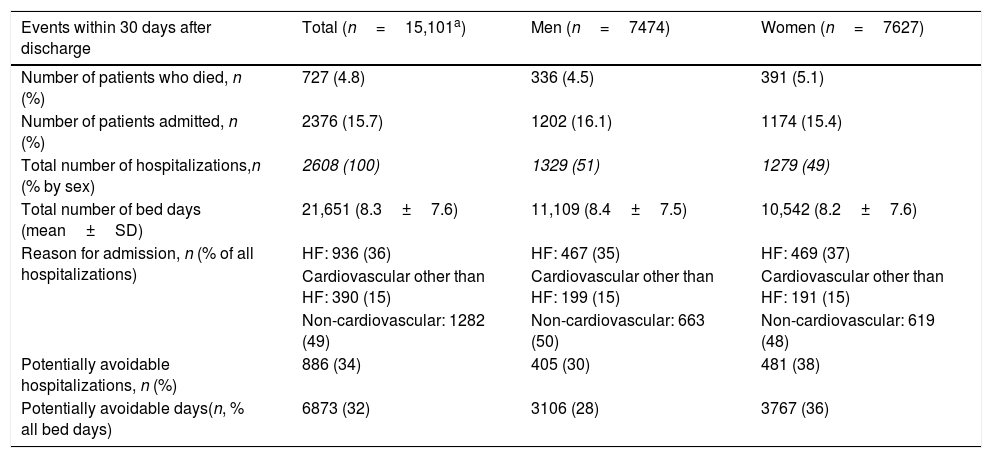
A total of 336 men (4.5%) and 391 women (5.1%) died within 30 days after the index discharge. In this period, 1202 men (16.1%) and 1174 women (15.4%) had new unplanned admissions, some of them more than once, yielding a total of 1329 admissions in men and 1279 in women and resulting in 21,651 cumulative bed days, with mean hospital stays in days of 8.4±7.5 in men and 8.2±7.6 in women. The main reason for unplanned hospitalization as documented at discharge was non-cardiovascular in 1282 cases (49%), HF itself in 936 (36%), and cardiovascular conditions other than HF in 390 (15%). Of these hospitalizations, 30% in men and 38% in women were considered to be potentially avoidable according to the criteria used in this study (6873 potentially avoidable days combining both sexes; Table 2).
Frequencies and percentages of patients who died or had unplanned or potentially avoidable hospitalizations within 30 and 365 days after the first admission for heart failure and lengths of hospital stay.
| Events within 30 days after discharge | Total (n=15,101a) | Men (n=7474) | Women (n=7627) |
|---|---|---|---|
| Number of patients who died, n (%) | 727 (4.8) | 336 (4.5) | 391 (5.1) |
| Number of patients admitted, n (%) | 2376 (15.7) | 1202 (16.1) | 1174 (15.4) |
| Total number of hospitalizations,n (% by sex) | 2608 (100) | 1329 (51) | 1279 (49) |
| Total number of bed days (mean±SD) | 21,651 (8.3±7.6) | 11,109 (8.4±7.5) | 10,542 (8.2±7.6) |
| Reason for admission, n (% of all hospitalizations) | HF: 936 (36) | HF: 467 (35) | HF: 469 (37) |
| Cardiovascular other than HF: 390 (15) | Cardiovascular other than HF: 199 (15) | Cardiovascular other than HF: 191 (15) | |
| Non-cardiovascular: 1282 (49) | Non-cardiovascular: 663 (50) | Non-cardiovascular: 619 (48) | |
| Potentially avoidable hospitalizations, n (%) | 886 (34) | 405 (30) | 481 (38) |
| Potentially avoidable days(n, % all bed days) | 6873 (32) | 3106 (28) | 3767 (36) |
| Events within 365 days after discharge | Total (n=15,101a) | Men (n=7474) | Women (n=7627) |
|---|---|---|---|
| Number of patients who died, n (%) | 3695 (24.5) | 1875 (25.1) | 1820 (23.9) |
| Number of patients admitted, n (%) | 7760 (51.4) | 3917 (52.4) | 3843 (50.4) |
| Total number of hospitalizations,n (% by sex) | 14,559 (100) | 7621 (47) | 6938 (53) |
| Total number of bed days (mean±SD) | 116,061 (8±7.4) | 61,284 (8±7.3) | 54,777 (7.9±7.5) |
| Reason for admission, n (% of all hospitalizations) | HF: 4700 (32) | HF: 2369 (31) | HF: 2331 (34) |
| Cardiovascular other than HF: 1945 (13) | Cardiovascular other than HF: 1027 (13) | Cardiovascular other than HF: 918 (13) | |
| Non-cardiovascular: 7914 (54) | Non-cardiovascular: 4225 (55) | Non-cardiovascular: 3689 (53) | |
| Potentially avoidable hospitalizations, n (%) | 5088 (35) | 2431 (32) | 2657 (38) |
| Potentially avoidable days(n, % all bed days) | 37,766 (33) | 17,767 (29) | 19,999 (37) |
The main reasons for admission within 30 days after discharge, besides HF itself (936 cases, 418 of which – 45% – were PAHs), were cardiovascular reasons other than HF: dysrhythmia (110 cases, 0% – PAH); coronary atherosclerosis (52 cases, 100% – PAH); and acute cerebrovascular disease (51 cases, 3–5.9% – PAHs). Among non-cardiovascular reasons, the most common were chronic obstructive pulmonary disease (101 cases, 100% – PAHs) and lower respiratory tract infections other than pneumonia (99 cases, 9–9.1% PAHs) (Table A5). No trend (increasing or decreasing) was observed in the incidence of the various events studied over the course of the study period (Table A6).
Within 365 days after the index dischargeA total of 1875 men (25.1%) and 1820 women (23.9%) died within the first year after their first admission for HF. During this time, 3917 men (52.4%) and 3843 women (50.4%) from the initial cohort generated 14,559 unplanned hospitalizations, resulting in 116,061 cumulative bed days (mean hospital stay 8±7.4 days). Among these hospitalizations, the principal diagnosis documented at discharge was HF in 4700 cases (32%), other cardiovascular conditions in 1945 (13%), and other non-cardiovascular conditions in 7914 (54%). Of these hospitalizations, 32% in men and 38% in women were considered to have been potentially avoidable according to the criterion used in this study (37,766 potentially avoidable days of hospital stay in both sexes) (Table 2).
For the entire year after the index discharge, the reasons for admission were very similar to those observed within 30 days after discharge. The rates of PAH were also similar, both overall and by disease group (Table A5). Finally, the median overall survival was 1166 days (95% CI: 1105–1248) in men and 1204 days (95% CI: 1150–1266) in women (Figure B1).
DiscussionMain findings (summary)Based on the data available from hospital electronic health records, the estimated prevalence of HF among over-40-year-olds in the Basque Country is 1.22% as of December 31, 2015. The annual incidence rate has slightly increased during the study period in both women (from 2.51 to 2.9 per 1000) and men (from 2.86 to 3.13 per 1000). This increase seems to be attributable to changes in the population pyramid, as it disappears after adjusting for age.
The absolute number of first discharges after admission for HF was similar in men and women, but the mean age was approximately 5 years older in women than in men (78 vs 83 years). Most of the cohort had a high level of comorbidity, patients having a median of over eight chronic conditions. Four out of five patients had hypertension and one third of the men had chronic obstructive pulmonary disease. Overall, 42% of the patients had had contact with cardiologists in the 12 months prior to the index discharge. Among those who survived the first admission, 5% died within the following 30 days, and 25% died within the first year after discharge. Approximately 15% and 50% of the patients had at least one unplanned hospitalization within 30 and 365 days after the index discharge, respectively, 54% were for non-cardiovascular reasons (49% and 54% within 30 and 365 days, respectively) and 4 out 10 of the hospitalizations were considered to have been potentially avoidable. On the other hand, we have observed an increasing trend in the prevalence of cardiovascular risk factors in individuals diagnosed with HF.
Comparison with other studies using the same or other methodsThe incidence of HF in our study, applying very restrictive criteria, was in the lower range of rates published in the literature, which vary depending on the diagnostic criteria used and the geographical area.15,16 We should note that the methodology used varies substantially between studies. Up to six methods based on signs and symptoms have been described, these yielding very different estimates of the prevalence of the disease.17
The prevalence in our study population was also in the lower range of rates published in Spain, but closer to that reported for most of the developed countries, probably due to methodological limitations of the former.5
Nevertheless, we observed some secular trends in the epidemiology of the disease. As can be inferred from other studies, the incidence of HF remains stable18 or even has decreased19 in recent years. In contrast, the prevalence is increasing undoubtedly due to population aging, but also due to increasing survival thanks to the use of successful evidence-based treatments.20
Our finding of an increase in comorbidities in these patients is consistent with results of other studies, both regarding cardiovascular diseases21 and so-called discordant comorbidities, described in the aforementioned study by Tran et al. and in the Swedish cohort of Vetrano et al.22
Finally, the rates of admission within 30 days and 1 year after discharge are similar to those described for neighboring countries, but notably lower than those found in studies in some other countries such as the USA.23
Notably, the rate of PAH reported in the latter study was higher than ours, which may point to one of the common causes of variations in clinical practice, namely, those associated with differences in the care offered.24 This finding, however, has to be interpreted with caution, as the operational definitions of PAH are substantially different in these studies.
Implications for preventionThe prevention of HF requires the prevention and control of hypertension and other modifiable risk factors for heart disease.
On the one hand, we are dealing with patients with ever more complex conditions: we observed an increase in the prevalence of hypertension, hyperlipidemia and other known risk factors, and the number of associated chronic conditions is increasing, as is the age at presentation. On the other hand, patients have been in contact with clinicians at different levels of care multiple times during the year prior to the first discharge after admission for HF, especially primary care, which means that there are numerous opportunities for the secondary prevention of the disease, for example those contacts could be better used to try to improve the low rates of compliance with guideline-directed pharmacological therapy.25 In patients with a very high cardiovascular risk, the target of maintaining systolic pressure under 120mmHg has shown to reduce the rates of mortality, cardiovascular events and hospitalizations for HF.26
In the current circumstances, with clinicians generating large quantities of data, there is a need for expert processing of the data available, in order to synthesize it and make it genuinely useful for decision making. In relation to this, the use of real world data would greatly facilitate advances toward more individualized care, through the introduction of tools to support clinical decision making.27
Impact on the health systemThe cohort generated 14,559 hospitalizations within the first year after their first discharge resulting in 116,061 bed days. Though the absolute figure is notable, it is even more striking that 35% of all these hospitalizations could, in theory, have been avoided. This should make us reflect on the potential impact of interventions in this area. Measures that have been found to be effective against PAHs include strengthening continuity of care, this having been shown to reduce the rate of PAH by 5% to 8%.28 It has also been suggested that specific intervention plans, such as the current Basque population-based plan, could improve care outcomes.29
Finally, and in contrast to the dominant hypothesis that PAHs are caused by suboptimal management of the disease at the outpatient level, recent studies suggest that PAHs reflect more or less intensive use of the referral hospital, as well as of the care provided in primary care,11 or failure of prevention efforts to reduce the demand for emergency care by chronic patients.30
On the other hand, 5 out of 10 unplanned hospitalizations after the index discharge were for non-cardiovascular reasons (49% and 54% within 30 and 365 days, respectively) while HF accounted for only a third of all unplanned hospitalizations (36% and 32% within 30 and 365 days, respectively). These results are consistent with those reported in other studies and suggest the need for HF care to focus on the integrated patient health burden and better management of comorbidities in HF.31
LimitationsThe definition of disease in this study probably implies exclusion of most cases of less severe HF (NYHA 1–2). Although other types of data relevant to the study of HF, such as ejection fraction or brain natriuretic peptide, are electronically stored, they are not currently available in a structured format and hence were not included in our analysis. Nevertheless, the diagnostic criteria selected, together with the advanced age of patients in the cohort, make us believe that most of them will be chronic patients, in an advanced stage of the disease and with certain level of systolic and diastolic dysfunction.
On the other hand, exclusion criteria could have been a source of heterogeneity. Actually, excluding transfer on discharge could introduce bias in analysis of geographic variation. However, we think that the impact of heterogeneity on main results will be low.
Finally, we did not analyze data related to pharmacy. This could have helped to better describe our population and to detect gaps in use and dose of guideline-directed medical therapy that could partly explain these findings. In fact, according to a recently published article, in the real-world setting, the use of standard-of-care therapies, both before and after the onset of worsening HF, is low, with high unmet medical needs, suggesting that patients with worsening HF require novel treatment strategies as well as greater optimization of existing guideline-directed therapy.32 In that sense, we are working on a project that will integrate drugs prescription and dispensation trajectories into our cohort, and that could bring new insights to the real-world use of standard-of-care therapies and consequences in patients with HF.
Conclusions(1) People admitted for HF are old, have multimorbidity, and high rates of admissions due to non-CVD reasons and PAHs after discharge. These findings suggest the need of strengthening continuity of care and managing comorbidities. (2) Besides, most people admitted for HF have previous contacts with the Healthcare system identifying opportunities for prevention before disease worsening.
Author contributionsFelipe Aizpuru: conception and design of the study; interpretation of data; drafting of the manuscript. Eduardo Millán: acquisition, analysis and interpretation of data; revision of the manuscript. Inés Garmendia: acquisition, analysis of data; revision of the manuscript. Maider Mateos: acquisition and analysis of data; revision of the manuscript. Julián Librero: design of the study; revision of the manuscript.
All the authors approved the final version to be published and agreed to be accountable for all aspects of the work.
FundingThe present study has been financed by the Spanish Government; Carlos III Health Institute, grant number PI13/00912 and Basque Government, grant number 2014111055.
Conflict of interestNone of the authors report conflicts of interest in this work.
We thank Ideas Need Communicating Language Services, Bilbao, who translated and reviewed the manuscript; the Informatics and Health Information Services of the Directorate of the Basque Health Service (Osakidetza), for their help identifying and extracting the data for the study; to Carlos Sola, Deputy Director of the Healthcare division of Osakidetza, for his continued support; the members of the Group of Studies for a Learning Health System: Aitziber Etxagibel, Ricardo Samper, Mikel Ogueta (Osakidetza-HealthCare directorate); Nekane Jaio, María Armendariz (Osakidetza-Barrualde Health Integrated Organization); María Robledo, Naiara Parraza, Antxon Apiñaniz (Osakidetza-Araba Health Integrated Organization); Berta Ibáñez, Arkaitz Galbete (NavarraBiomed); Francisco Estupiñán (Instituto Aragonés de Ciencias de la Salud-IACS).