We aimed to investigate the effect of obtaining follow-up blood cultures (FUBC) and persistent growth in FUBC on clinical outcomes in bacteremia cases caused by Escherichia coli and Klebsiella pneumoniae in the geriatric age group.
MethodsPatients over 65 years of age with E. coli or K. pneumoniae bacteremia were included in the study. Patients were grouped as those who obtained FUBC and those who did not. Patients with FUBC were grouped as persistent bacteremia or negative FUBC. Demographic, clinical characteristics and outcomes were compared between these groups.
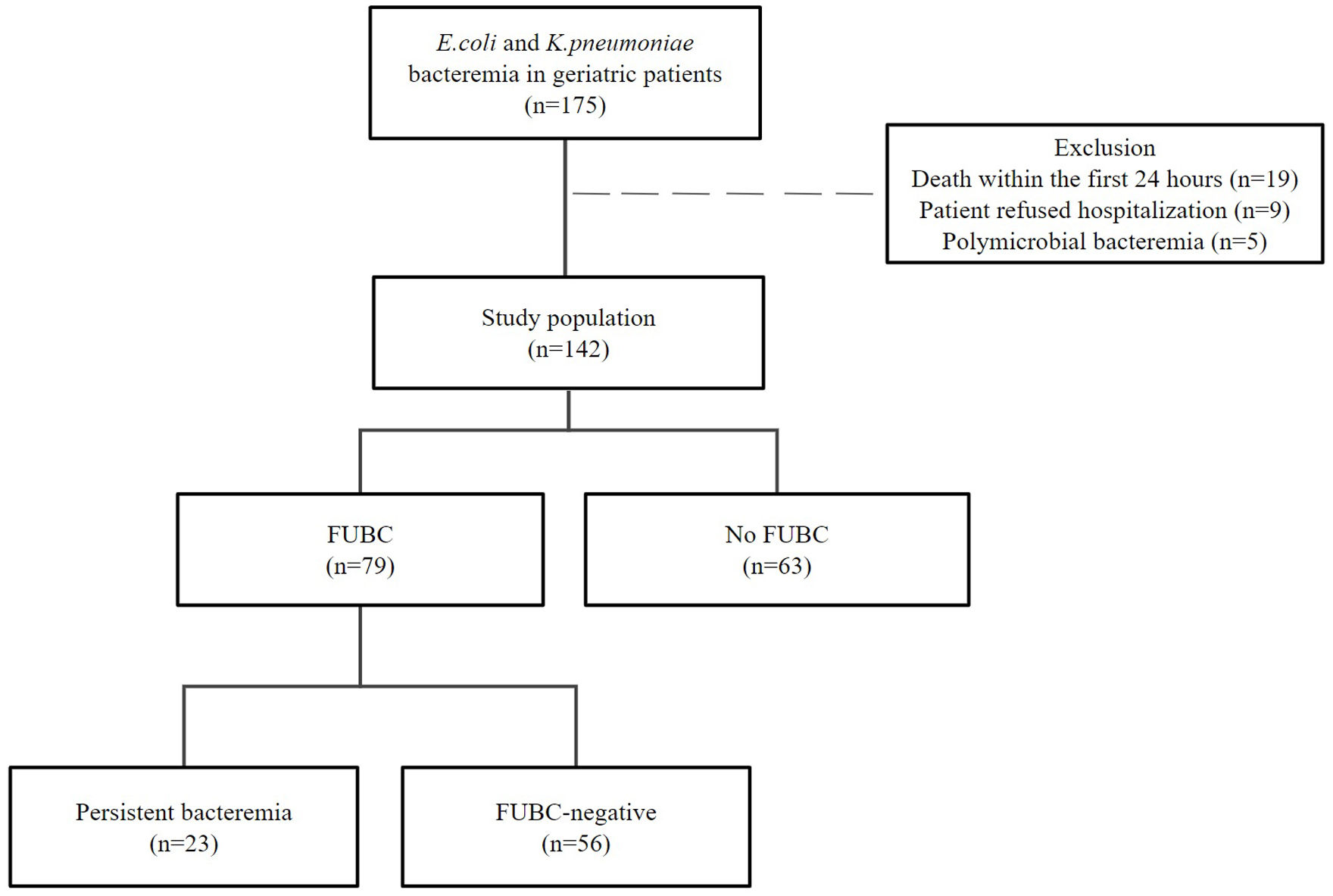
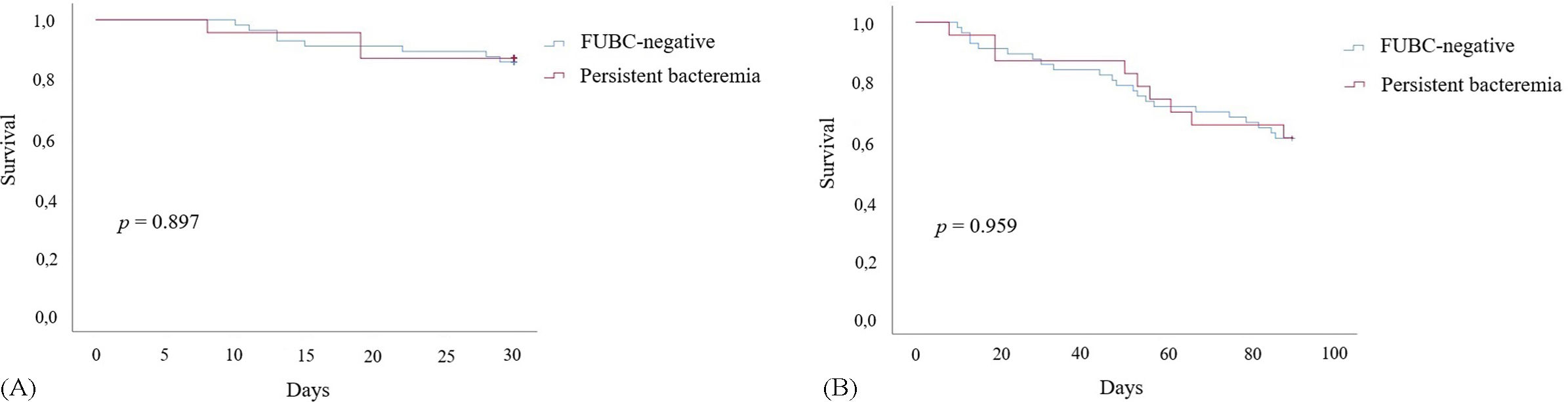
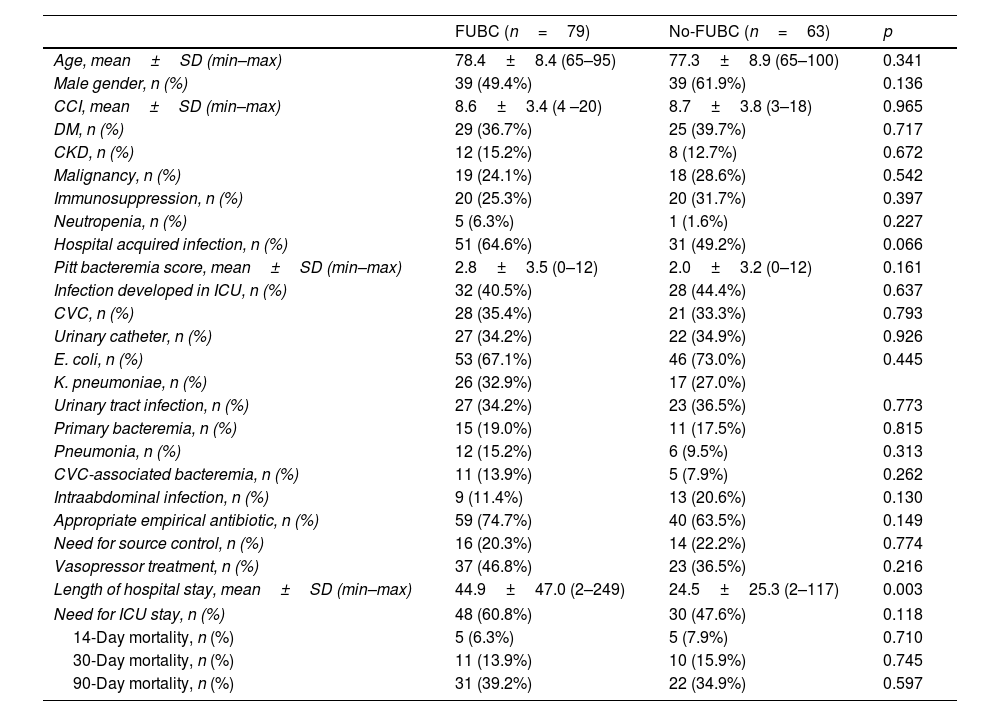
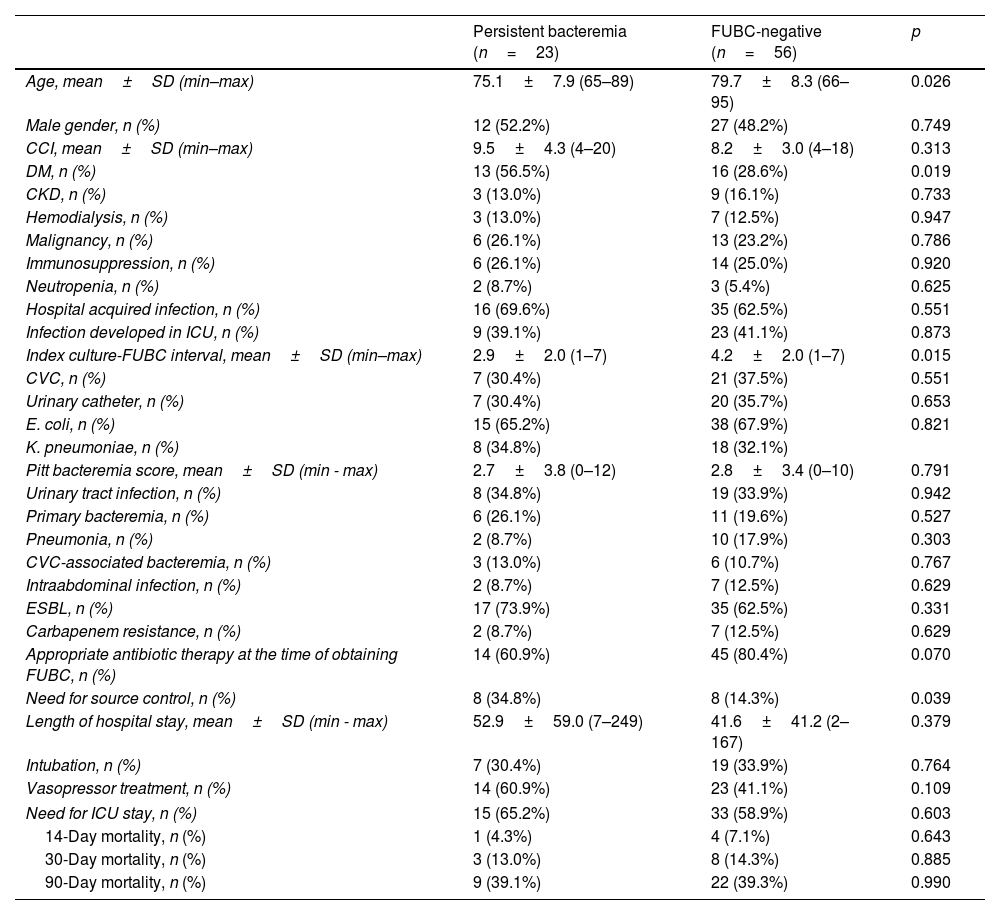
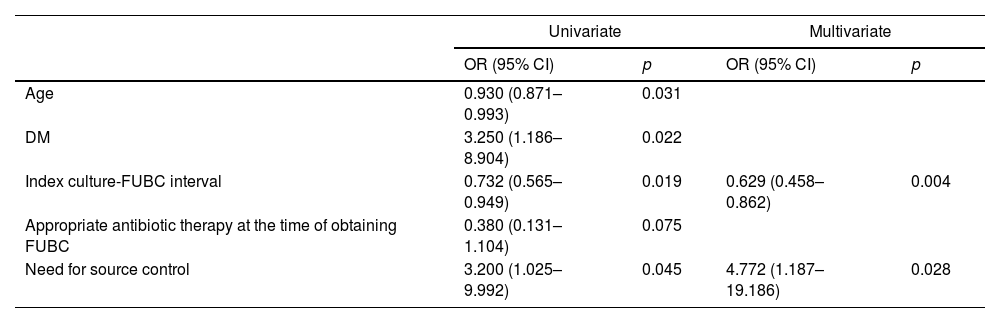
ResultsA total of 142 patients were included. FUBC were obtained in 79 patients (55.6%) and persistent bacteremia was detected in 23 (29.1%). There were no significant differences in demographic data and clinical characteristics between patients with and without FUBC. There was no statistical difference in the outcomes between patients with of without FUBC. Patients with persistent bacteremia had a higher prevalence of diabetes mellitus, and the interval between index blood culture and FUBC was shorter (2.9 days versus 4.2 days). There was no difference between patients with persistent bacteremia and patients with no growth on FUBC in terms of length of hospital stay, need for intensive care unit, intubation, vasopressor therapy, and 14, 30, 90-day all-cause mortality.
ConclusionIn patients over 65 years of age with E. coli or K. pneumoniae bacteremia, neither obtaining FUBC nor persistent bacteremia on FUBC showed any difference in terms of outcomes such as mortality and need for admission to an intensive care unit.
Investigamos el efecto de la obtención de hemocultivos de seguimiento (FUBC) y el crecimiento persistente en FUBC sobre los resultados clínicos en bacteriemia causada por E. coli y K. pneumoniae en pacientes geriátricos.
MétodosSe incluyeron pacientes mayores de 65 años con bacteriemia por E. coli o K. pneumoniae. Se agruparon en aquellos a los que se les realizaron FUBC y a los que no. A los que se les realizaron FUBC se clasificaron según si presentaban bacteriemia persistente o si los FUBC fueron negativos. Se compararon las características demográficas, clínicas y los resultados entre estos grupos.
ResultadosSe incluyeron 142 pacientes. Se realizaron FUBC en 79 (55.6%) y se detectó bacteriemia persistente en 23 de ellos (29.1%). No se encontraron diferencias significativas en los datos demográficos y las características clínicas entre los pacientes con y sin FUBC. Tampoco hubo diferencias significativas en los resultados entre los pacientes a los que se les realizaron FUBC y aquellos que no. Los pacientes con bacteriemia persistente presentaron una mayor prevalencia de diabetes mellitus, y el intervalo entre el hemocultivo índice y el FUBC fue más corto (2,9 días frente a 4,2 días). No se observaron diferencias entre los pacientes con bacteriemia persistente y aquellos sin crecimiento en los FUBC en términos de duración de estancia hospitalaria, necesidad de cuidados intensivos, intubación, terapia vasopresora y mortalidad por todas las causas a los 14, 30 y 90 días.
ConclusiónEn pacientes mayores de 65 años con bacteriemia por E. coli o K. pneumoniae, ni la obtención de FUBC ni la bacteriemia persistente en los FUBC mostraron ninguna diferencia en términos de mortalidad y necesidad de cuidados intensivos.
Artículo
Socio de la Sociedad Española de Enfermedades Infecciosas y Microbiología Clínica

Para acceder a la revista
Es necesario que lo haga desde la zona privada de la web de la SEIMC, clique aquí