We report the case of a male patient presenting with hyperammonemia after liver transplantation that was successfully reduced after treatment with carglumic acid and l-arginine. Carglumic acid can be a useful tool to reduce high ammonia levels that frequently appear after liver transplantation.
Case reportOur patient is a 60-year-old male who received a first liver transplant to remove a hepatocellular carcinoma due to a virus B infection. He received the liver of an 80-year-old donor who had a double venous system and poor blood flow, which hampered the operation. In the first 48h he had a graft dysfunction secondary to ischemia. While waiting an urgent retransplantation procedure, he began to manifest encephalopathic symptoms. His ammonia levels were found to be 155μmol/L (normal <50μmol/L), but no measures were undertaken. He then received parenteral nutrition containing 8g of nitrogen per day. Given his progressive neurological impairment, 12h later ammonia levels were measured again and had reached 199μmol/L. The inborn errors of metabolism specialist was contacted, exogenous protein nutrition was stopped and pharmacological treatment was initiated with 5g of arginine and 2g of carglumic acid administered via a nasogastric tube. In the follow-up blood test performed 3h later, ammonia levels had decreased to 122μmol/L, thus ruling out the need for dialysis or other drugs. A second course of 3g of arginine and 1g of carglumic acid achieved a further reduction of ammonia levels to 98μmol/L 6h after the initiation of treatment. The patient regained consciousness. In the following hours he received his second liver transplant. His ammonia levels after surgery were 78μmol//L and it was decided to maintain treatment with 3g of arginine every 8h until 48h later, by which time the patient had begun normal nutrition and had normal ammonia levels.
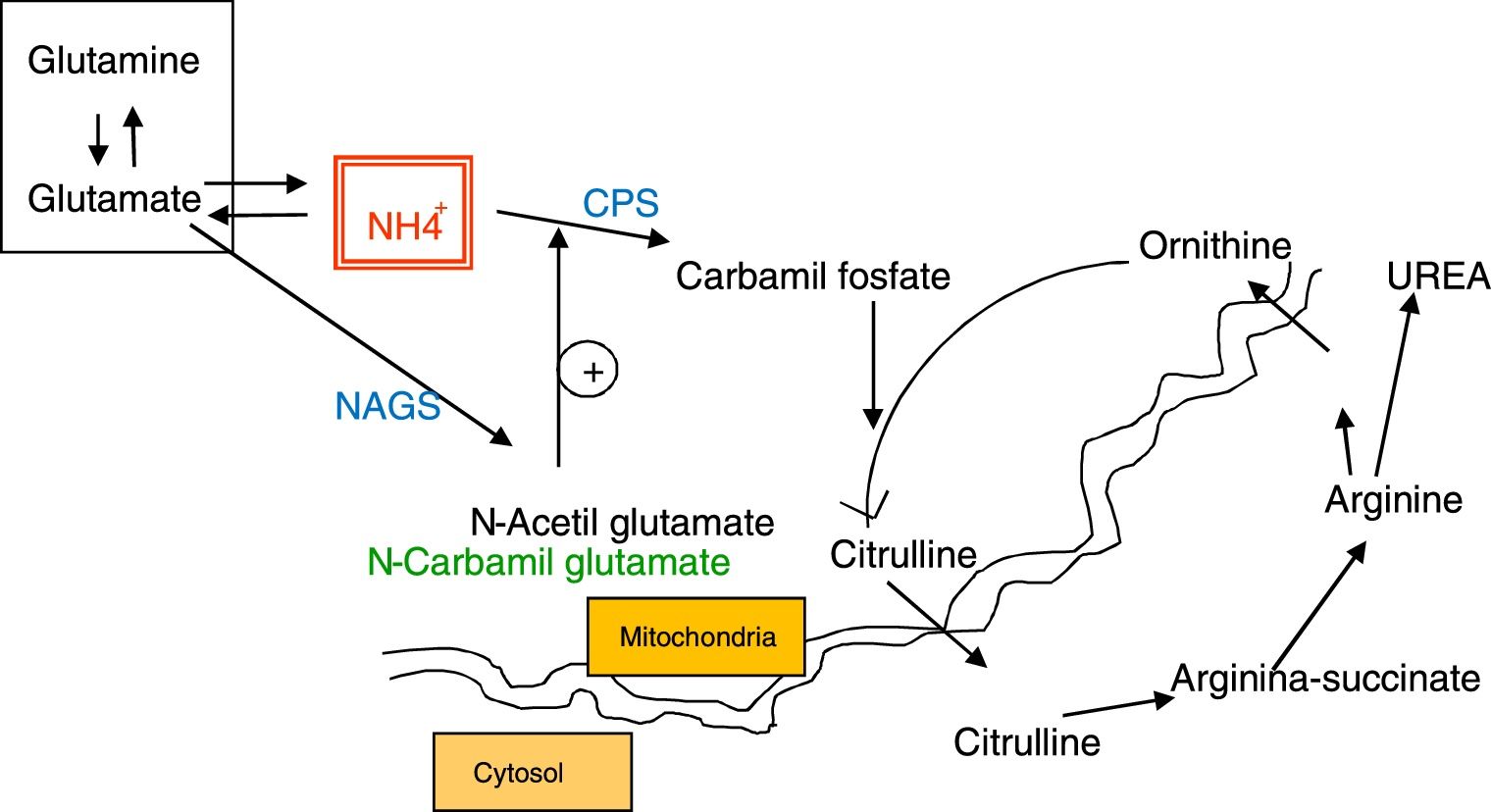
DiscussionFollowing liver transplantation, hyperammonemia is a frequent complication and it is due to the inability to eliminate ammonia through ureagenesis during the time the patient has no functional liver before, during and in the early post-implantation stages. After transplantation, the urea cycle is less effective because of a defect in glutamate synthetase activity1,2 but we believe it is also due to a secondary N-acetyl glutamate synthase (NAGS) deficiency as a result of possible acetyl-CoA depletion. A transplanted liver is invariably subjected to ischemic injury due to the transient lack of blood perfusion during transportation 3 and, as in our case, due to surgical complications. Ischemia depletes hepatocyte stores of ATP4 since, in the absence of aerobic respiration, mitochondrial oxidation of the enzymatic cofactors NADH and FADH2 does not take place. Lack of respiration also prevents the generation of acetylCoA from aerobic glycolysis or from beta-oxidation (since acylCoa dehydrogenase is FAD dependent), which is likely to promote an overall acetylCoA depletion in hepatocytes. As acetylCoA is one of the two substrates of NAGS, its deficiency induces a lack of NAGS activity in hepatocytes, resulting in reduced levels of N-acetyl glutamate (NAG) and therefore a reduced activity of the initiating enzyme of the urea cycle, carbamoyl phosphate synthase (CPS) (see Fig. 1).
All these factors can contribute to impairing the ability of the graft to eliminate the excessive ammonia accumulated. In these patients, hyperammonemia is not usually very high, but it can sometimes be a major complication as it induces cerebral edema and leads to neurological symptoms that can leave sequelae. Measures against cerebral edema and dialysis may be necessary treatments at this stage. Ammonia levels should be measured routinely after liver transplantation, and treatment should be started as soon as possible. Although the posttransplantation hyperammonemia episode can hardly be avoided, its duration and intensity could be minimized contributing to actívate the urea cycle in the newly transplanted liver. Patients could benefit from the intake of arginine and carglumic acid (currently commercialized as Carbaglu®), a structural analog of NAG that can bind to and activate the CPS enzyme.5,6
Conflict of interestThere is no conflict of interest at work.