Concomitant chemoradiotherapy (CCRT) is widely used in the treatment of patients with stage III non-small lung carcinoma (NSCLC). The early identification of patients with poor prognosis is the premise of personalized treatment for patients. The aim of the study was to evaluate the prognostic value of clinical parameters and primary tumor SUVmax on pre-treatment 18F-FDG PET/CT in patients with stage III NSCLC.
Material and methodsClinical records of 79 stage III-NSCLC patients with pre-treatment 18F-FDG PET/CT imaging, treated with definitive CCRT were retrospectively reviewed. The clinical endpoints in terms of progression-free survival (PFS) and overall survival (OS) were correlated with the median pre-treatment primary tumor SUVmax. Furthermore, other factors influencing patient outcome were analyzed.
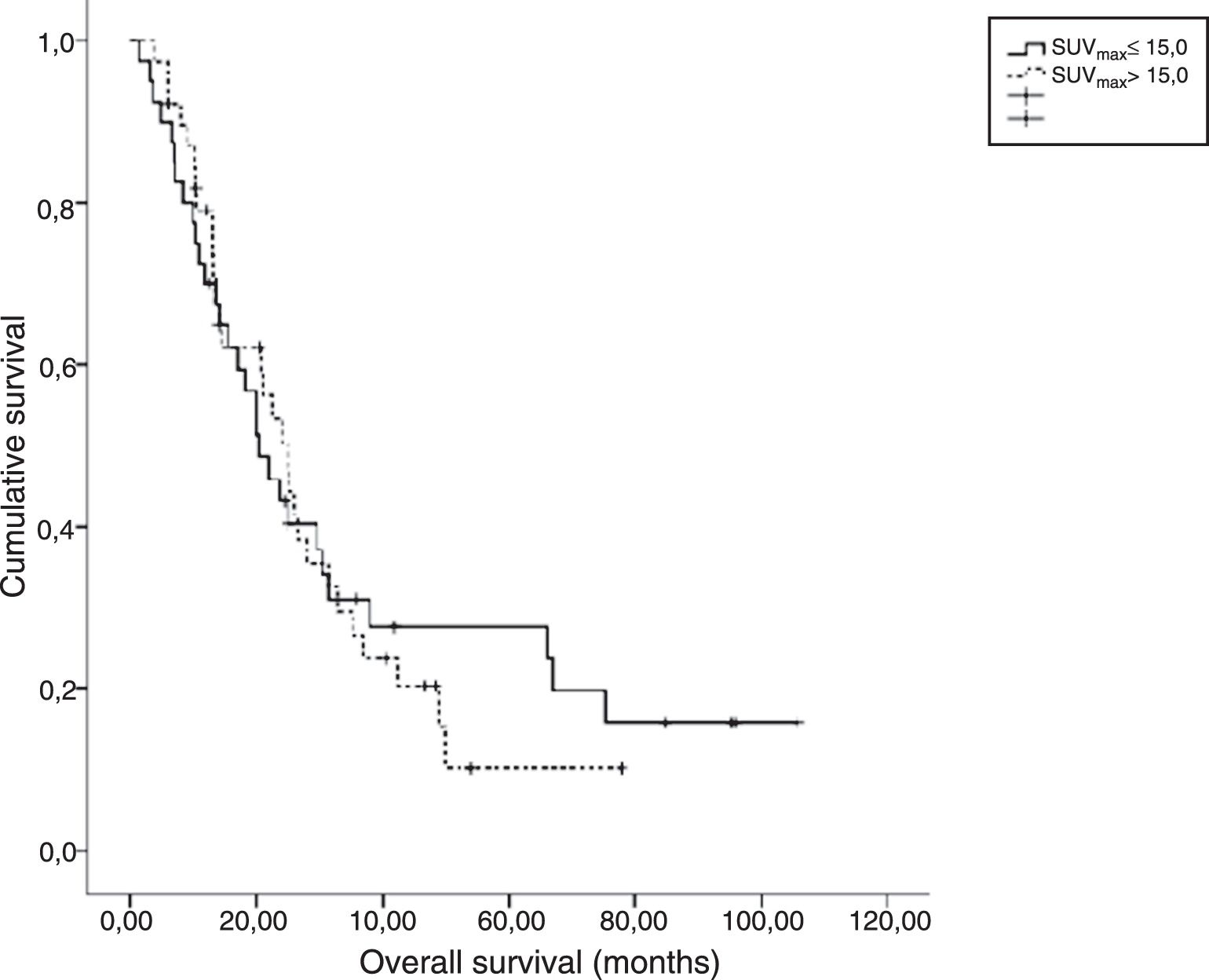
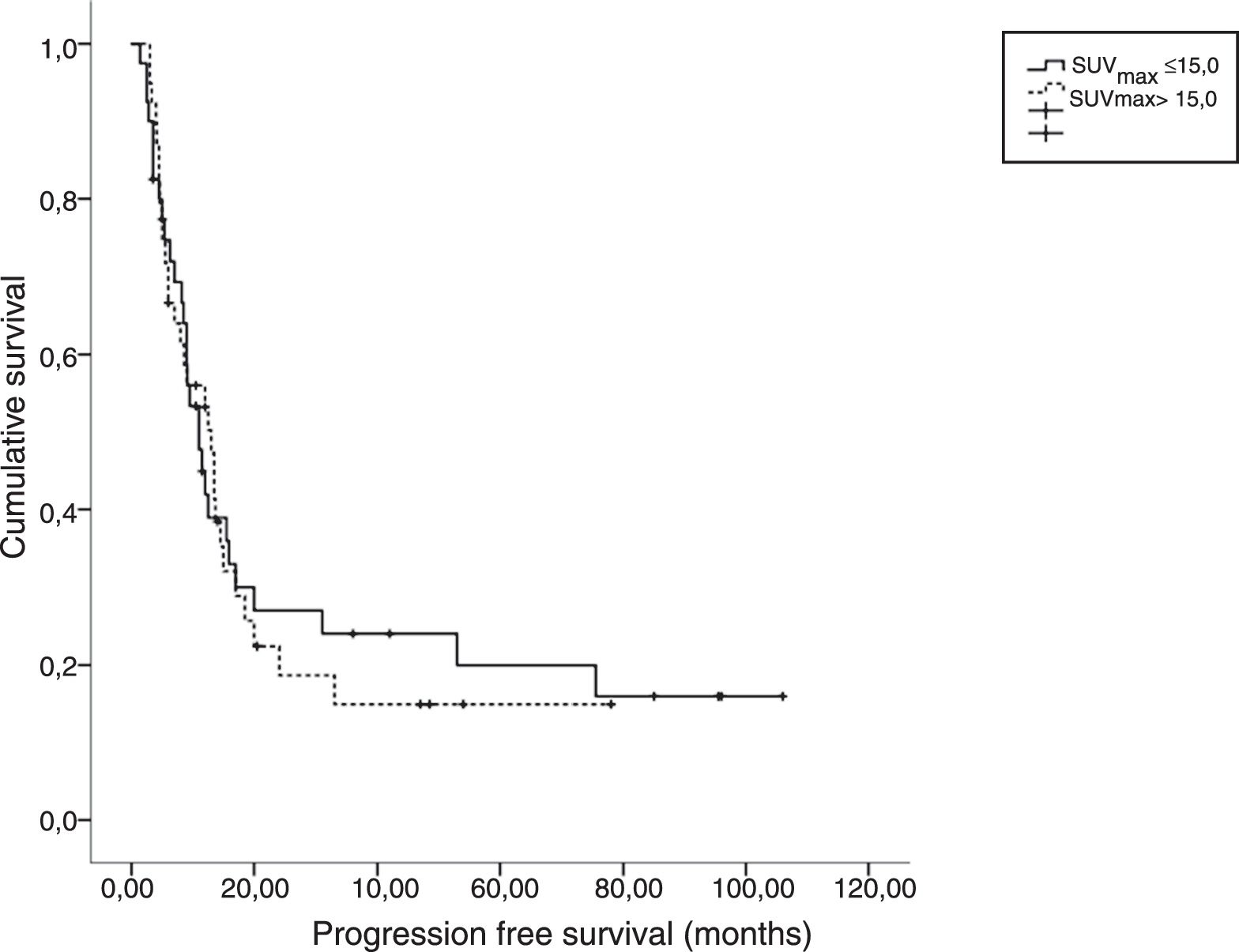
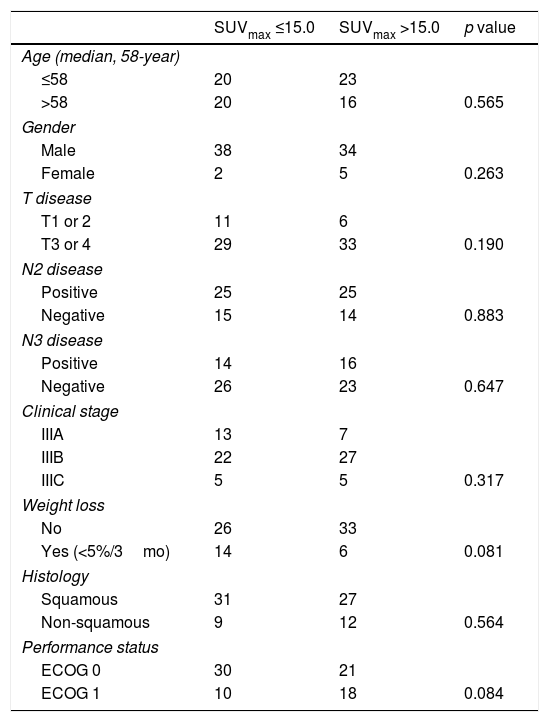
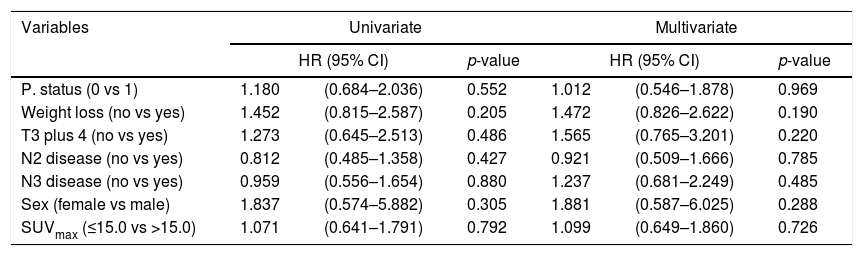
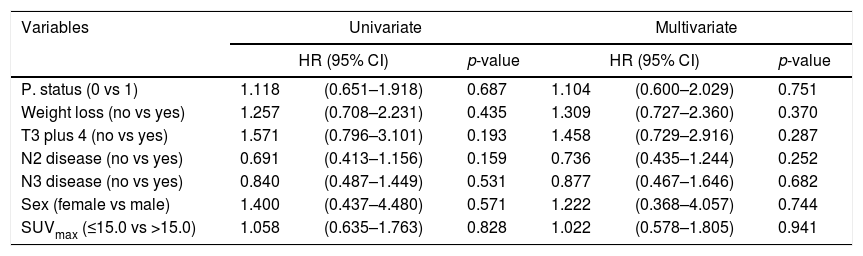
ResultsThe median age of patients was 58 years (range, 45–71) with 72 (91%) males. Squamous cell carcinoma (73%) was the most common histologic type. Performance status was very good (ECOG 0) in 64.5% of patients. Sixty (79%) patients had died at the time of this analysis. Median OS and PFS were 22.5 and 12.0 months, respectively. Patients were dichotomized according to pre-treatment primary tumor SUVmax ≤15.0 vs >15.0. There was no statistically significant difference for OS and PFS in both arms. Multivariate analysis showed that pre-treatment SUVmax was not a significant predictor of OS (HR 1.099, p=0.726) and PFS (HR 1.022, p=0.941).
ConclusionsSUVmax with threshold value of 15.0 on the primary tumor before treatment had no prognostic value in our patient group with stage III NSCLC treated with definitive CCRT.
La quimioradioterapia concomitante (CCRT) se utiliza de forma amplia para tratar a los pacientes con cáncer de pulmón no microcítico (CPCP) en estadio III. La identificación precoz de los pacientes con mal pronóstico constituye la premisa del tratamiento personalizado de los pacientes. El objetivo de este estudio fue evaluar el valor pronóstico de los parámetros clínicos y SUVmax del tumor primario sobre PET/TC con 18F-FDG pre-tratamiento en los pacientes con CPCP en estadio III.
Material y métodosSe revisaron retrospectivamente las historias clínicas de 79 pacientes de CPCP en estadio III con PET/TC con 18F-FDG pre-tratamiento, tratados con CCRT definitiva. Se correlacionaron los criterios de evaluación clínicos en términos de supervivencia libre de progresión (SLP) y supervivencia global (OS) con el valor SUVmax medio del tumor primario pre-tratamiento. Además, también se analizaron otros factores de influencia en el resultado del paciente.
ResultadosLa edad media de los pacientes fue de 58 años (rango, 45-71), con 72 (91%) varones. El carcinoma de células escamosas (73%) fue el tipo histológico más común. El estatus del desempeño fue muy bueno (ECOG 0) en el 64,5% de los pacientes. Sesenta (79%) pacientes habían fallecido en el momento de este análisis. Los valores medios de OS y SLP fueron de 22,5 y 12 meses, respectivamente. Los pacientes se dicotomizaron con arreglo a SUVmax del tumor primario pre-tratamiento ≤15 vs >15. No se encontró diferencia significativa para OS y SLP en ambas ramas. El análisis multivariante reflejó que SUVmax pre-tratamiento no constituía un factor predictivo de OS (HR 1,099, p=0,726) y SLP (HR 1,022, p=0,941).
ConclusionesEl índice SUVmax con un valor límite de 15 con respecto al tumor primario pre-tratamiento no tiene valor pronóstico en nuestro grupo de pacientes de CPCP en estadio III, tratados con CCRT definitiva.
Article
If you experience access problems, you can contact the SEMNIM Technical Secretariat by email at secretaria.tecnica@semnim.es or by phone at +34 619 594 780.

Revista Española de Medicina Nuclear e Imagen Molecular (English Edition)