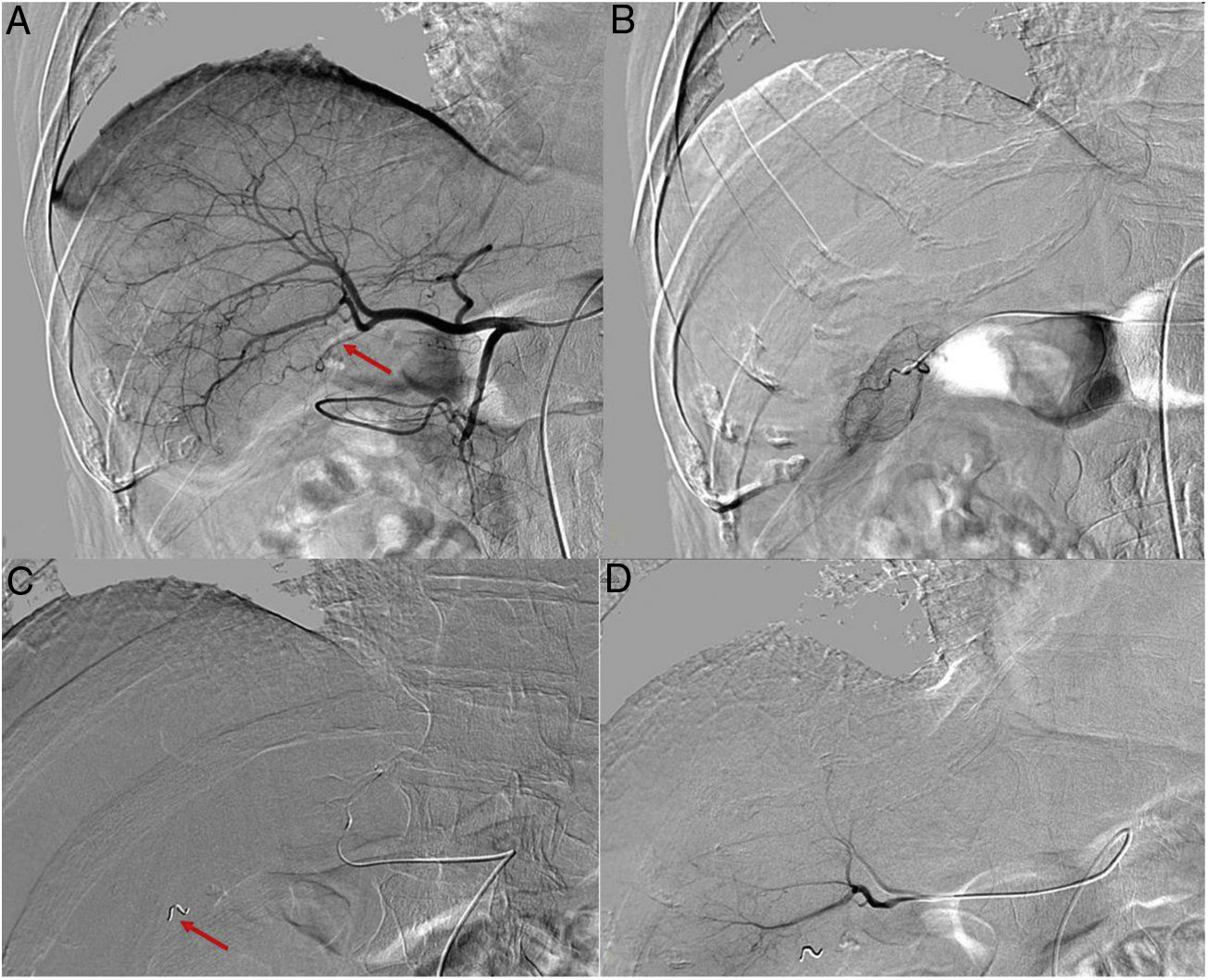
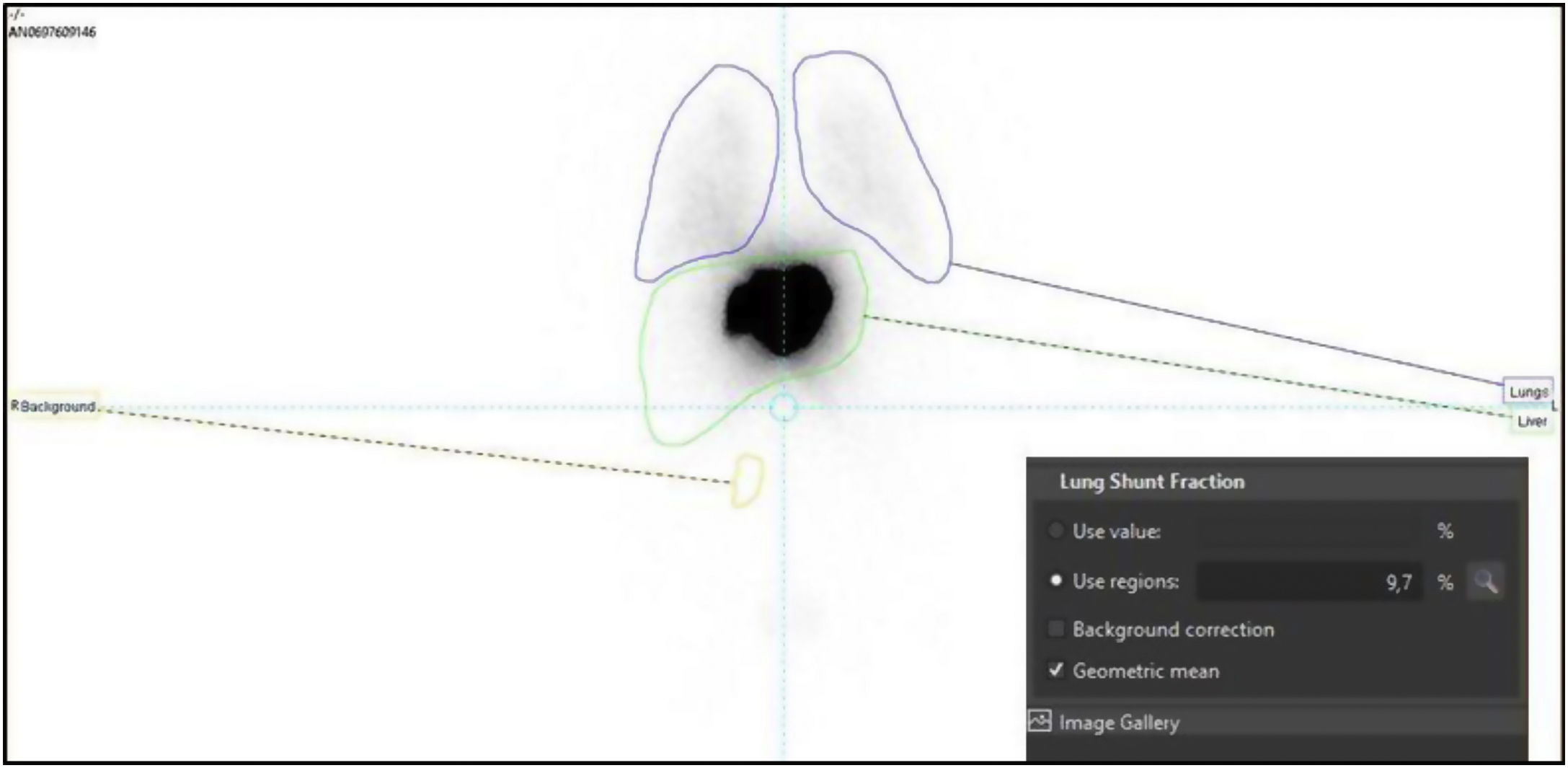
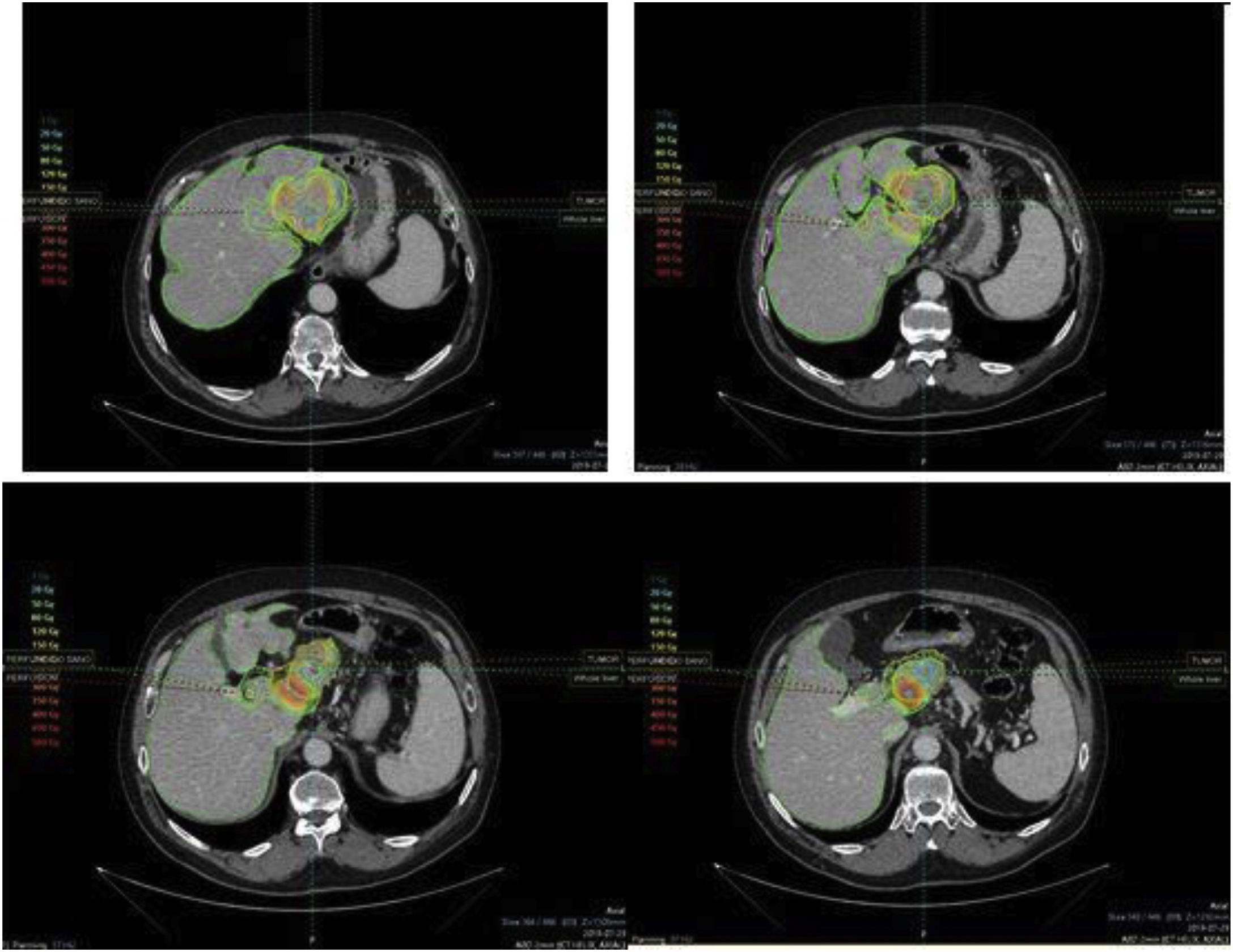
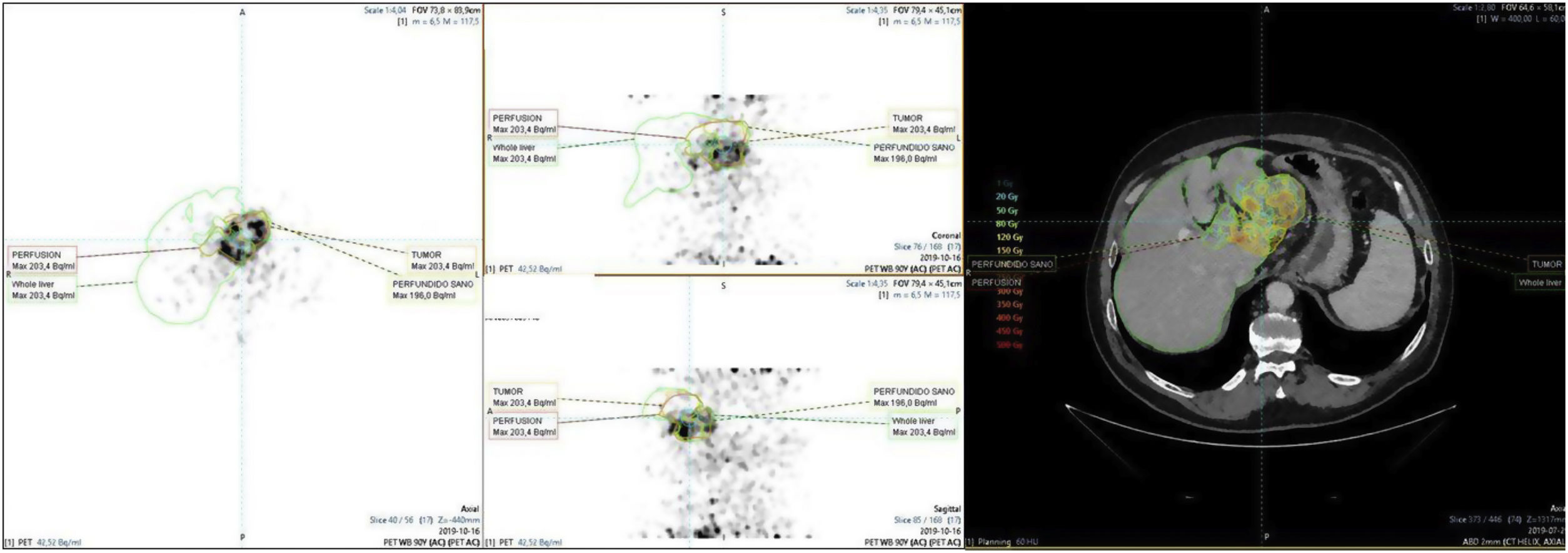
To determine the therapeutic effectiveness and safety of transarterial radioembolization (TARE) with Yttrium-90 in patients with colorectal cancer (CRC) liver metastases and to evaluate the prognostic value of different biomarkers.
Material and methodsThis prospective longitudinal study enrolled consecutive patients with CRC liver metastases treated with TARE between November 2015 and june 2020. The therapeutic response at three and six months (RECIST1.1 criteria) and the relationship of biomarkers with therapeutic response, by calculating objective tumor response rates (ORR) and disease control (DCR), and overall survival (OS) and progression-free (PFS).
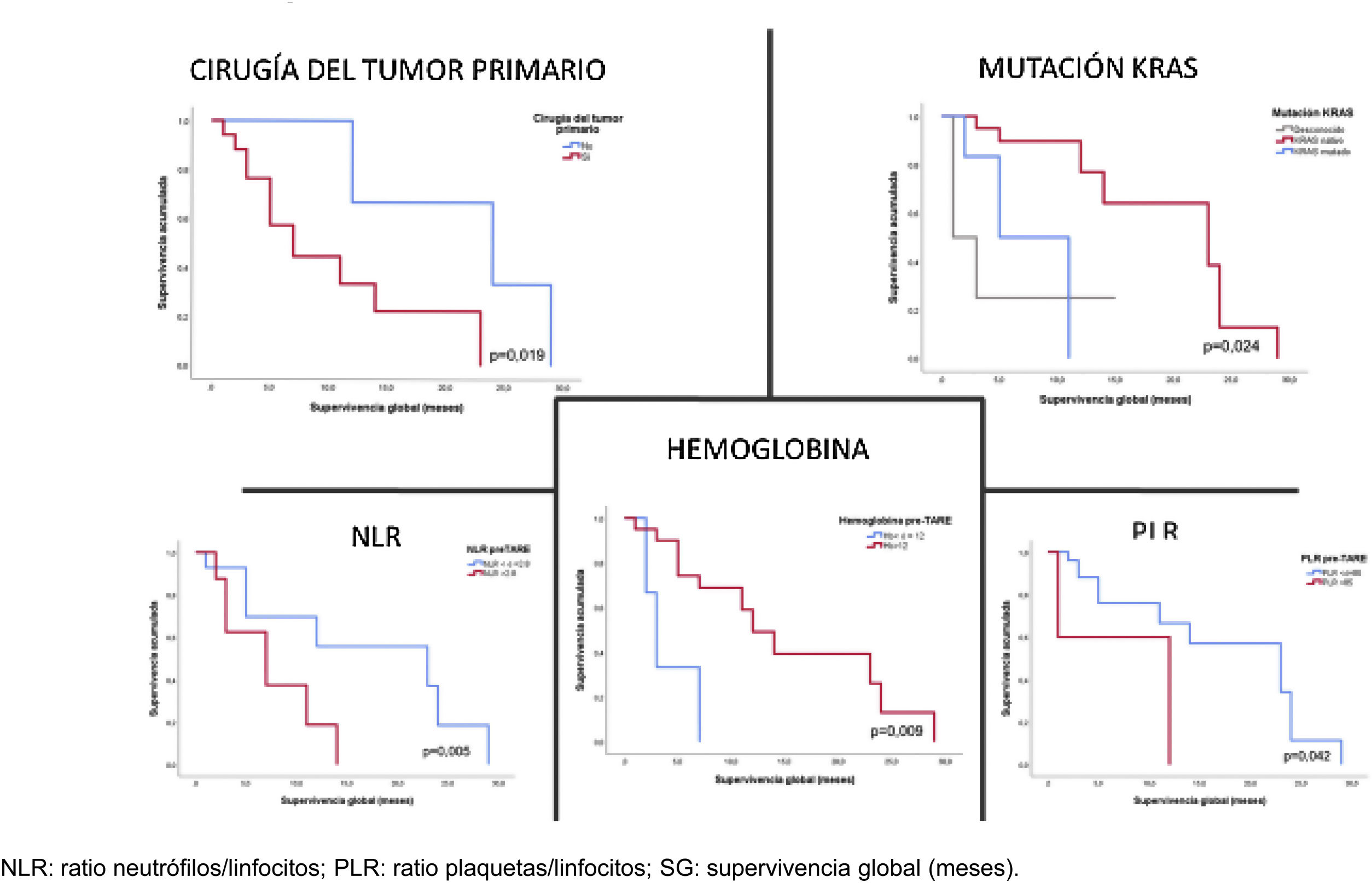
ResultsThirty TAREs were performed in 23 patients (mean age, 61.61 ± 9.13 years; 56.5% male). At three months, the objective response rate (ORR) was 16.7% and the disease control rate (DCR) 53.3%. At six months, the disease progressed in 80%. The ORR and DCR were significantly associated with age at diagnosis (P = 0.047), previous bevacizumab treatment (P = 0.008), pre-TARE haemoglobin (P = 0.008), NLR (P = 0.040), pre-TARE albumin (P = 0.012), pre-TARE ALT (P = 0.023) and tumour-absorbed dose > 115 Gy (P = 0.033). Median overall survival (OS) was 12 months (95% CI, 4.75–19.25 months) and median progression-free survival (PFS) 3 months (95% CI, 2.41–3.59). OS was significantly associated with primary tumour resection (P = 0.019), KRAS mutation (HR: 5.15; P = 0.024), pre-TARE haemoglobin (HR: 0.50; p = 0.009), pre-TARE NLR (HR: 1.65; P = 0.005) and PLR (HR: 1.01; P = 0.042).
ConclusionTARE prognosis and therapeutic response were predicted by different biomarkers, ranging from biochemical parameters to tumour dosimetrics.
Analizar la eficacia terapéutica, seguridad y valor pronóstico de diferentes biomarcadores de la radioembolización transarterial con esferas de itrio-90 (TARE) en pacientes con metástasis hepáticas de cáncer colorrectal.
Material y métodosEstudio prospectivo que incluye los pacientes con metástasis hepáticas de cancer colorrectal tratados con TARE entre noviembre de 2015 y junio de 2020. Se analizó la respuesta terapéutica (3 y 6 meses, criterios RECIST v1.1) mediante el cálculo de las tasas de respuesta tumoral objetiva (ORR) y de control de la enfermedad (DCR), así como la asociación de los biomarcadores con la respuesta terapéutica y la supervivencia global (SG) y libre de progresión (SLP).
ResultadosTreinta TARE en 23 pacientes (edad media 61,61 ± 9, 13 años; 56,5% varones). La ORR a los 3 meses fue del 16,7% y el DCR del 53,3%. A los 6 meses progresaron el 80% de los pacientes. La ORR y DCR se asociaron con la edad (p = 0,047), tratamiento con bevacizumab (p = 0,008), hemoglobina (p = 0,008), NLR (p = 0,040), albúmina (p = 0,012) y GPT (p = 0,023) previas a la TARE, y la dosis absorbida tumoral estimada > 115 Gy (p = 0,033). La mediana de SG fue de 12 meses (IC 95%: 4,75–19,25 meses) y de SLP 3 meses (IC 95%: 2,41-3,59 meses). La SG se asoció con la cirugía del tumor primario (p = 0,019), mutación KRAS (p = 0,024), hemoglobina (p = 0,009), NLR (p = 0,005) y PLR (p = 0,042) previos a la TARE.
ConclusiónLos biomarcadores con capacidad para predecir el pronóstico y respuesta terapéutica a la TARE incluyen desde parámetros bioquímicos a factores relacionados con la dosimetría tumoral estimada.
Article
If you experience access problems, you can contact the SEMNIM Technical Secretariat by email at secretaria.tecnica@semnim.es or by phone at +34 619 594 780.

Revista Española de Medicina Nuclear e Imagen Molecular (English Edition)