Rupture of distal biceps has a frequency of 1.2 cases per 100,000 population. Conservative treatment presents a loss in supination of 40% and flexion of 30%, like transfers to the brachialis. Our goal is to review our experience with anatomical reattachment.
Material and methodsCohorts study. We analized an historic cohort, although recent, treated with double approach (Boyd-Anderson) compared to another prospective cohort (treated with single and double approach). Sample size of 80 patients. Mean age of 48.9 ± 5.9 years. We analyzed laterality, time to diagnosis, presurgery time, surgery time, approach, type of reintegration, rehabilitation time, Mayo Elbow Performance Score (MEPS), biomechanical study and complications. Follow up from 2 years to 7 years.
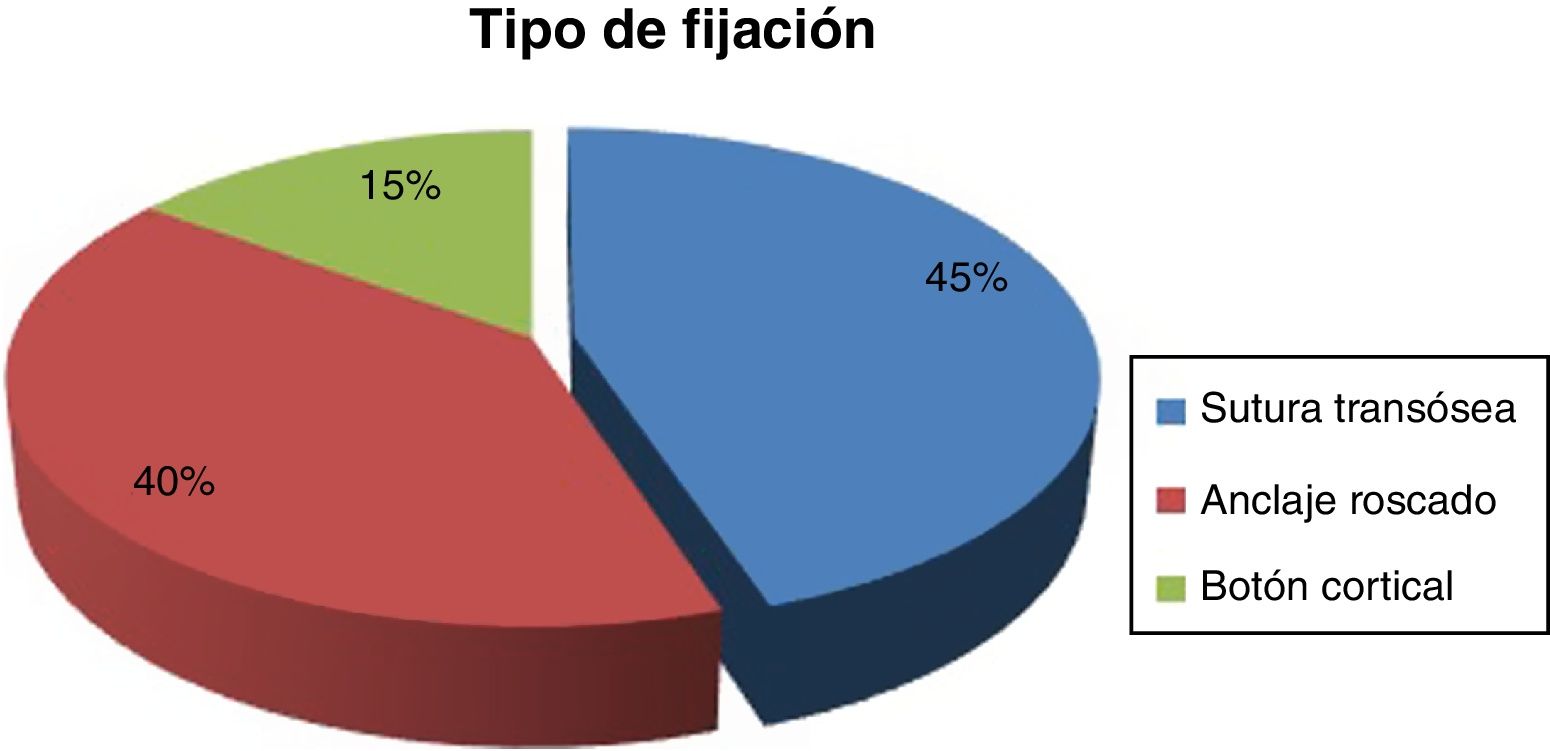
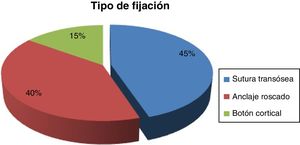
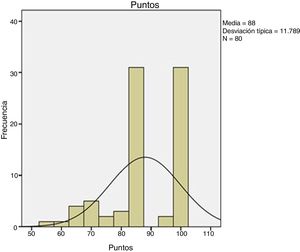
ResultsTime for diagnosis was about 4 days. Preoperative time: 12.2 ± 6.4 days. Surgical time: 61.9 ± 15.7 min. We used the two-way surgical approach (Boyd-Anderson-Morrey) in 78.8% of patients, and the anterior surgical approach (Henry) in 21.2% of cases. Transosseous reinsertion was made in 45% of cases, with screw anchor in 40% and with cortical button in 15%. Rehabilitation time: 91 ± 29.7 days. MEPS: 88 ± 11.7 points. There was loss of flexor strength of 28 ± 16.6%. Complications: 1 case of proximal radioulnar synostosis, 2 radial transient paralysis and 5 cases with residual stiffness. Hypothesis contrast: We reached best results in MEPS with double surgical approach (P = .009), with fewer complications (P = .008). We observed increased pain with cortical buttons (P < .05) and less surgical time if patient was operated before 1 week (P = .03). Relative risk of 0.2 when we compared type of approach with radial nerve lesion risk, considering that double approach had less cases, what it would be consider as a protective factor.
ConclusionsIn our experience, we believe that anatomic reattachment is an effective treatment. Double surgical approach presents best result in MEPS, with fewer complications than with anterior approach.
La rotura de bíceps distal tiene una frecuencia de 1,2 casos por 100.000 habitantes. El tratamiento conservador supone un detrimento supinador del 40% y flexor del 30%, al igual que las transferencias al braquial. Nuestro objetivo es revisar nuestra experiencia con la reinserción anatómica.
Material y métodosEstudio comparativo de cohortes, entre una cohorte histórica, aunque reciente, en la que se aplicó doble abordaje, o de Boyd-Anderson, frente a una cohorte prospectiva donde se aplicó abordaje simple y doble. Tamaño muestral de 80 pacientes. Edad media de 48,9 ± 5,9 años. Analizamos lateralidad, tiempos para diagnóstico, prequirúrgico, quirúrgico, tipo de vía, tipo de reinserción, tiempo de rehabilitación, escala Mayo Elbow Performance Score (MEPS), estudio biomecánico y complicaciones. Seguimiento mínimo en 2 años y máximo en 7 años.
ResultadosTiempo para el diagnóstico, con mediana de 4 días. Tiempo prequirúrgico: 12,2 ± 6,4 días. Tiempo quirúrgico: 61,9 ± 15,7 min. Doble vía (Boyd-Anderson-Morrey) en el 78,8%, vía única (Henry) en el 21,2%. Reinserción transósea (45%), con anclaje roscado (40%) y con botón cortical (15%). Tiempo rehabilitador: 91 ± 29,7 días. MEPS: 88 ± 11,7 puntos. Déficit de fuerza flexora final del 28 ± 16,6%.
Complicaciones1 caso de sinostosis radiocubital proximal, 2 neuroapraxias radiales y 5 casos con rigidez residual. Contraste de hipótesis: mejor resultado MEPS para doble vía (p = 0,009), con menos complicaciones (p = 0,008). Mayor dolor con botones corticales (p < 0,05). Menor tiempo quirúrgico si se operaban antes de una semana (p = 0,03). Riesgo relativo de 0,2 al comparar tipo de abordaje con riesgo de lesión radial, siendo el doble abordaje el que menos lo presentaba, lo que lo configuraría como factor protector frente a esta lesión.
The incidence of ruptures of the distal biceps is 1.2 ruptures/100,000 patients,1 although Kelly et al.2 report a frequency of 2.55 per 100,000 patient-years, or 5.35 cases per 100,000 patient- years. The age groups between the fourth and fifth decades are the most affected. A higher prevalence has been described in weightlifters, smokers (7.5 times more frequent1) and patients under treatment with steroids.
The pathogenesis of these injuries is explained by anatomy. Thus, despite an insertional area of 108 mm,2 with an insertional footprint occupying 63% of the length and 13% of the width of the bicipital tuberosity3–5 and being tendon prepared for stress, repetitive forces, as a flexor and supinator, place it at risk of rupture and a suspected clinical diagnosis will be determined through clinical tests that assess this function.6 The vascular supply, dependent proximally on the brachial artery and distally on the posterior interosseous recurrent artery, leaves an intermediate transition zone of about 21.4 mm,7 which is hypovascular, with a higher risk of rupture. This explains why most ruptures occur 2 cm from the distal insertion.
ObjectivesOur main objective was to analyse our experience with distal biceps reinsertions.
The main assessment variable was the Mayo Elbow Performance Score (MEPS).8
After analysing two surgical procedures for the same condition – the double approach versus the single anterior approach - our fundamental working hypothesis was that bicipital reinsertion using the double approach would provide better clinical results in terms of MEPS.
Our secondary objectives were to confirm potential clinical differences depending on the type of reinsertion or the timing of the intervention, and to analyse differences between the two approaches in terms of complications.
Material and methodsStudy designA comparative cohort study,9 a historical cohort of patients undergoing the double approach, albeit recent, versus a new prospective, experimental cohort in which the single and double approaches were used. In both cases the objective was the same: reinsertion of the bicipital tendon avulsed from its insertion.
PatientsStudy subjectsPatients diagnosed with bicipital tendon rupture by clinical examination and imaging study: ultrasound or magnetic resonance imaging (MRI),10 who underwent tendon reinsertion surgery.
Inclusion criteriaThe following four criteria had to be met:
- 1
Adult patients (between 18 and 65 years) of both sexes.
- 2
Clinical history of suspected bicipital injury, both by a mechanism of sudden counter resistance flexion, and by clinical appearance, with inverted Popeye sign, among others. Clinical examination consistent with the diagnosis.
- 3
Confirmation of diagnosis by ultrasound and/or MRI.
- 4
Acceptance and signing of informed consent from, whereby the patients were informed of the type of treatment and that they were participating in a clinical study, highlighting the minimal risks of superficial infection described in the literature (solved with intraoperative prophylaxis and our centre’s usual protocol: cefazolin, 2 g every 8 h i.v., or clindamycin, 600 mg every 8 h i.v., in the case of allergy, for 24 h) and the risks of synostosis, rigidity and/or neurovascular injury, described in the literature consulted.
Patients were excluded if, although they fulfilled the above four criteria and underwent surgery, it was not possible to directly reinsert the biceps, and they either required grafting or tenodesis to the anterior brachialis.
Statistical methodologySample sizeThe sample size was calculated from data from a historical cohort of 40 patients who underwent the double approach and from an initial pilot sample of 10 patients from the experimental, single-track cohort. To find a clinically relevant difference of 15 u between the means of the preoperative and postoperative MEPS test results, the results of a pilot sample, a population variability of 25 u in the first test, the bilateral nature of the test, an α error of 5% and a 1-β (power) of 80% were considered, resulting in a minimum necessary size of 15 individuals in each of the groups. Glaxo Smith Kline’s CTM ver. 1.1 was used for the calculation.
Finally, 63 patients were recruited in the double approach group and 17 in the single approach group, which slightly exceeds the minimum required sample size.
Thus, we obtained a sample size of 80 patients.
The variables analysed were the following: affiliation data (sex, age, laterality, jobs (high or low demand); time prior to diagnosis, prior to surgery, approach used, type of reinsertion, surgical time, rehabilitation time; MEPS, complications, re-interventions, biomechanical study to determine flexor or extensor deficit, etc.
Follow-up was a minimum of 2 years and a maximum of 7 years.
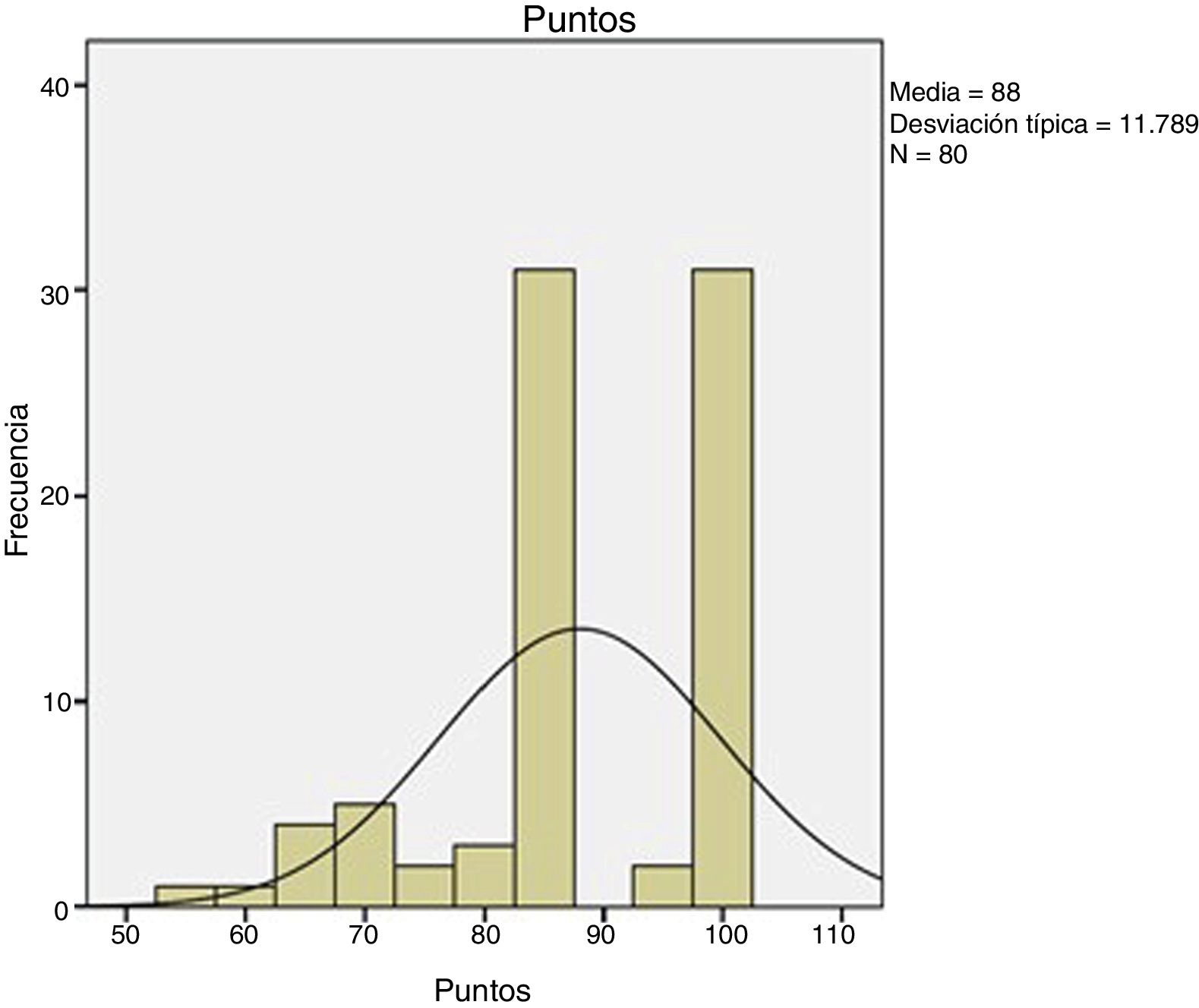
Descriptive analysisFirst, the data was explored to identify extreme values and identify differences between patient subgroups. Quantitative variables were expressed as mean and standard deviation or, if the distributions were asymmetric, median and percentile (P25 and P75), while qualitative variables were expressed with frequency tables and percentages. This analysis was performed for the MEPS test overall and by subgroup.
Inferential analysisWe used SPSS version 19.0 for the statistical study. We performed a normality study using the Kolmogorov-Smirnov test. We carried out a study to assess possible differences between the approaches used, using Student's t-tests (parametric variables), Mann Whitney U-tests (non-parametric) or X2 tests (categorical variables). We also assessed possible differences between types of reinsertion using ANOVA (parametric variables), Kruskall Wallis (non-parametric variables) or contingency tables (categorical variables). With respect to preoperative time (where the sample was divided into two groups: intervention in under 7 days following the trauma or intervention later than 7 days following the trauma), we also used the Student's t-test (for parametric variables), Mann Whitney U test (for non-parametric variables) and the X2 test (for categorical variables).
Analysis using SPSS 19.0 software.
Surgical techniquesThe treatment involves direct reinsertion of the tendon.11 Two fundamental techniques have been described for this:
- •
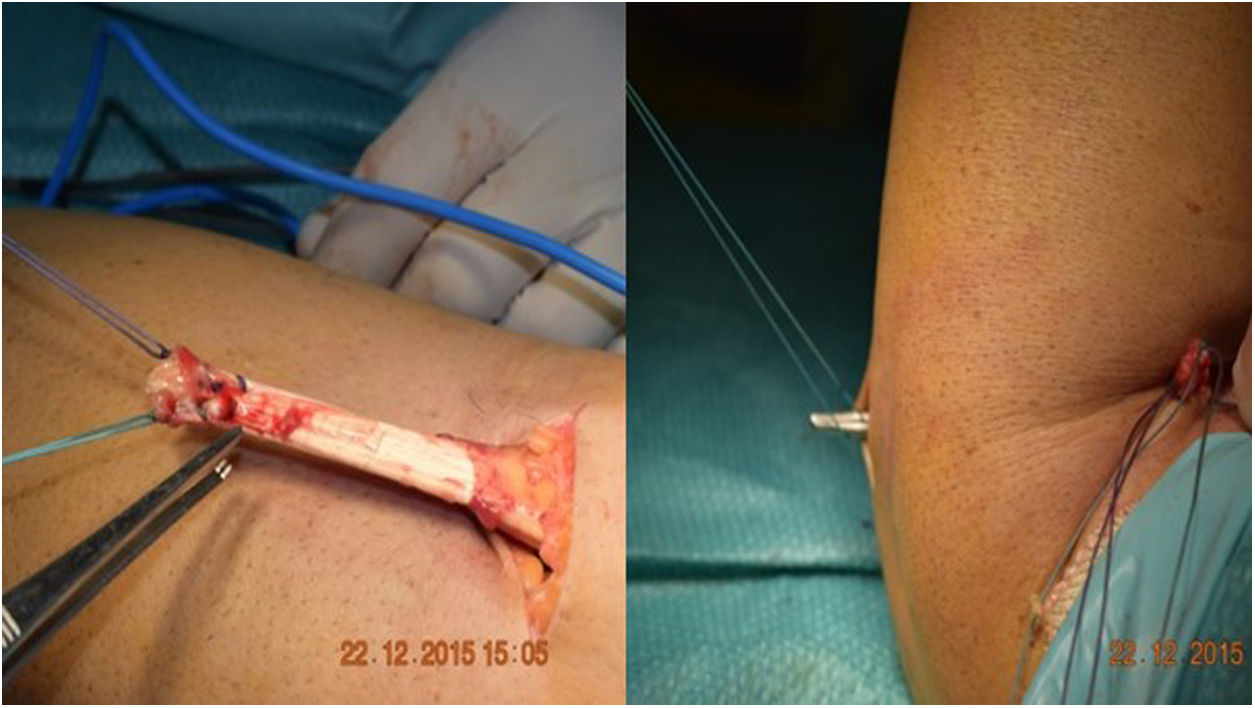
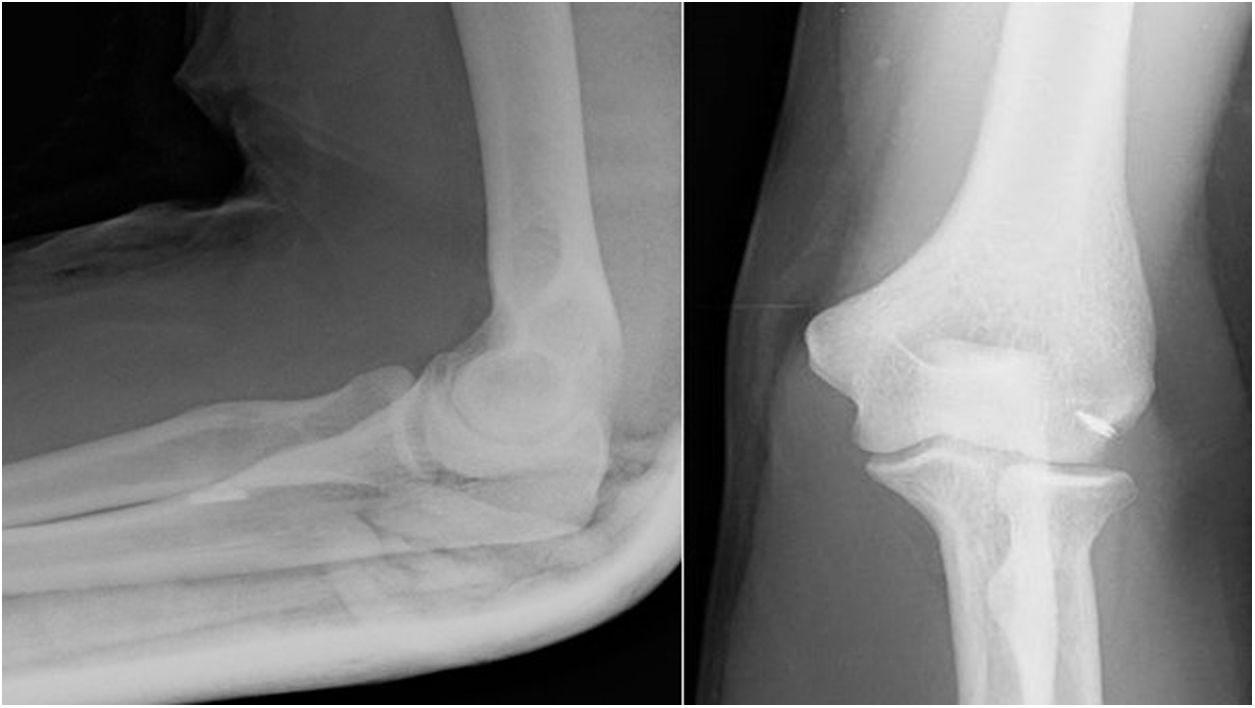
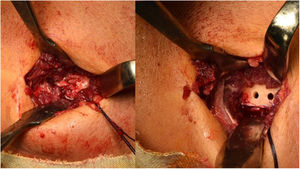
The Boyd-Anderson procedure,12 also used by Morrey,13,14 involves a double incision approach. First, an incision is made proximal to the elbow crease, disinserting the tendon which is prepared with double Krackow sutures. A curved forceps is then passed through the forearm between the ulna and the radius, avoiding contact with the ulnar cortex and with the tip of the forceps directed towards the bicipital tuberosity. A counter-incision is made in this area. After tissue dissection, and avoiding compression with spreaders in the superior region of the tuberosity (so as not to damage the posterior interosseous nerve), the sutured tendon is then transported to the bicipital tuberosity. There, an insertion site is prepared, and three holes drilled for transosseous reinsertion of the tendon (Figs. 1 and 2). Rollo et al.15 have described a technical modification of this double approach in which the tendon is passed through the forearm with the aid of a kind of cannula, which according to the author himself would reduce the risk of radioulnar synostosis, with good results on the MEPS.
Figure 1.Morrey’s modification of the Boyd Anderson approach. (A) Preparation of the tendon. Suture of each of the bundles, Krackow type, with two different threads, to identify each tendon portion. (B) Transfixing forceps exiting through the posterior region of the forearm to serve as transport for suture threads.
- •
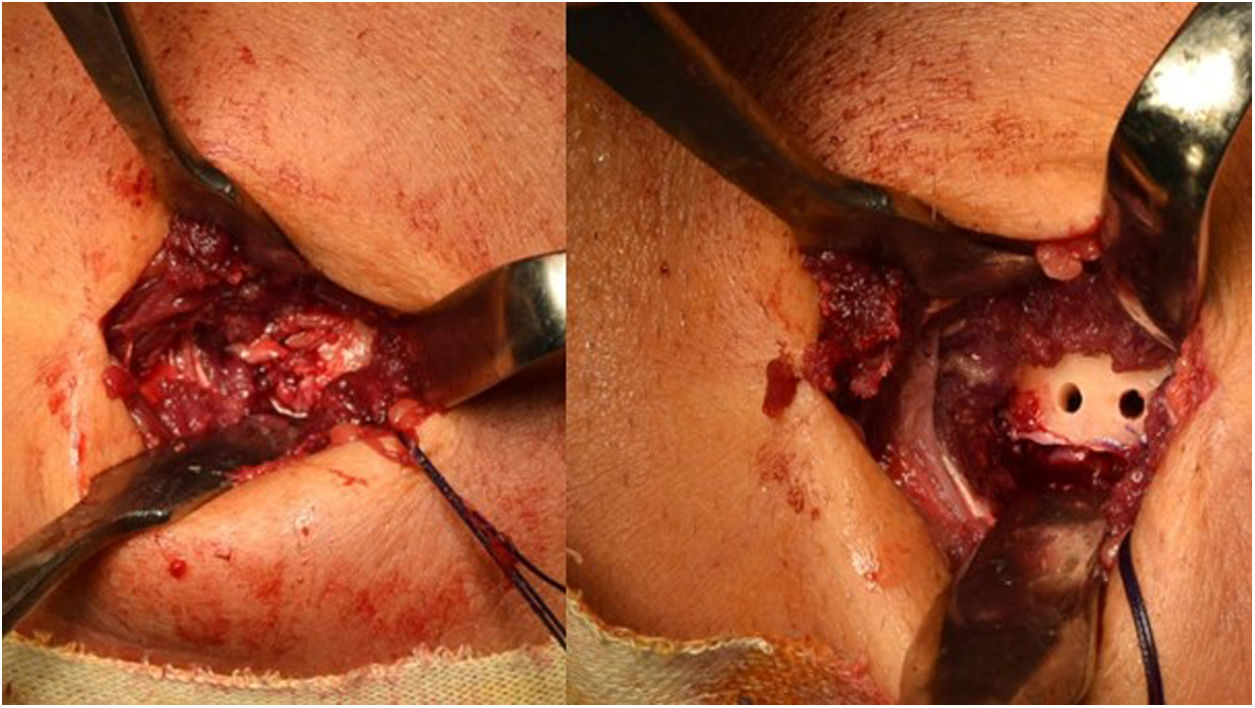
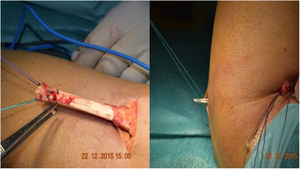
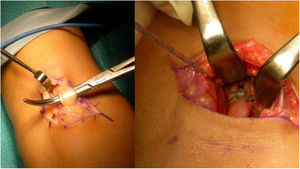
Henry’s route,16 or single anterior route, involves an incision of about 5 cm, longitudinal, starting from the anterior flexion crease of the elbow towards the distal aspect. On reaching the muscular plane, the lateral cutaneous antebrachial nerve should be identified and retracted radially. The bicipital canal should then be accessed, and the proximal end of the tendon located. It is important to find the radial recurrent artery to cauterise or ligate it and avoid the formation of postoperative haematoma. After clearly visualising the insertion area with the arm in full supination, the insertion footprint is curetted, and the anchor placement point is marked. In the case of Ramazzini et al.17 this would be an anchor with cortical fixation (Fig. 3).
We highlight some more technical aspects for correct repair:
Analysing the characteristics of the tendon, the distal biceps requires a force of 204 N to rupture. The insertion is divided into two portions: on the one hand, a short portion, whose insertion measures about 12 mm in length and 7 mm in width, with an area of 60 mm2; on the other, a long portion, whose insertion measures about 9 mm in length and 7 mm in width, with an area of 48 mm.21,18 This concept is useful when it comes to surgical repair, considering that the tendon suture must pick up both tendon bundles. Therefore, in our experience, we perform two Krackow-type sutures, which attempt to pick up both tendon bundles and then facilitate as much as possible the distribution of force through both tendon bundles. On the other hand, the tuberosity is blunt, and it is important to prepare the insertion site, to ensure that pronosupination does not place the suture under excess stress.19
In both cases subsequent immobilisation is required. In our case, for 3 weeks with a brachial splint at 90° flexion and neutral pronosupination, and then gradual rehabilitation is started from the third to the sixth week, protected by orthosis.
The mean age was 48.9 ± 5.9 years in our sample of 80 patients. One hundred percent were male. High-demand work was performed by 86.2% and low-demand work by 13.8%. Of the cases, 67.5% were right-handed and 32.5% were left-handed. Time to diagnosis was a median of 4 days. Of the cases, 98.8% (79) were diagnosed by ultrasound. Only one case was diagnosed exclusively by MRI. MRI was requested in addition to ultrasound to complete the preoperative study in two cases, where the diagnosis was uncertain. We should highlight tests such as Metzman’s test6 in the examination to confirm a suspected diagnosis.
The time to intervention was 12.2 ± 6.4 days, and the surgical time was 61.9 ± 15.7 min. The Boyd-Anderson-Morrey approach was used in 78.8% (63 cases). Henry’s approach was used in 21.2% (17 cases). The type of reinsertion was transosseous (45%), with threaded anchorage (40%) or with cortical button (15%) (Fig. 4).
ResultsThe MEPS score was 88 ± 11.7 (Fig. 5). The rehabilitation time was 91 ± 29.7 days. At the end we found a flexion strength deficit (verified in isokinetic study) of 28%±16.6% and an extension strength deficit of 30.8% ± 17.6%. All cases returned to work.
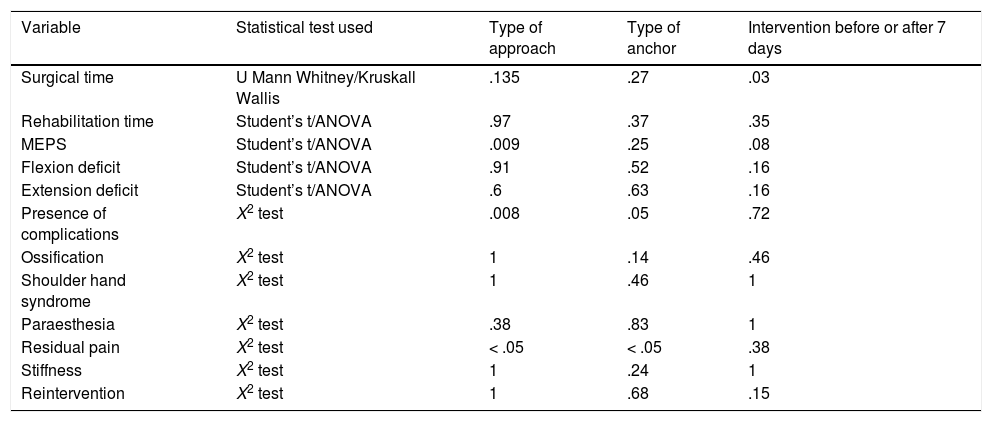
Our comparative study between the different types of approach obtained the following results: there were no differences regarding surgical time (p = .135), rehabilitation treatment time (p = .97), flexor deficit (p = .91) or extensor deficit (p = .6); there were differences in terms of MEPS score, with a p = .009, and this difference was in favour of the Boyd-Anderson approach, with a score of 89.7 ± 10.6, compared to 81.4 ± 13.6 for the anterior approach. In terms of categorical variables, there were only differences in the complication rate (p = .008), which was 11.1% for the Boyd Anderson approach, versus 41.2% for the anterior approach. Similarly, there were differences in residual pain, with a p value <.05, which was 4.8% for the Boyd Anderson approach compared to 41.2% for the anterior approach. There was no difference in the rate of ossification (p = 1), shoulder-hand syndrome (p = 1), paraesthesia (p = .38), stiffness (p = 1) or reoperation rate (p = 1). The relative risk was .2 when comparing the type of approach with the risk of radial injury, the double approach had the least risk of this and therefore constituted a protective factor against this injury.
In terms of type of anchor, we assessed the transosseous suture, threaded anchors, and cortical buttons. In this regard, the results obtained were as follows: there were no differences in all numerical variables, whether in surgical time (p = .27), rehabilitation time (p = .37), MEPS (p = .25), flexion deficit (p = .52) or extension deficit (p = .63); However, there were differences in complications (p = .05), which were slightly more frequent with threaded anchors and residual pain (p < .05) in half the cases treated with cortical buttons (Fig. 6A). There were no differences in terms of ossification (p = .14), shoulder hand syndrome (p = .46), paraesthesia (p = .83), stiffness (p = .24) or reoperation rate (p = .68).
Regarding the timing of the intervention, we only found differences in surgical time (p = .03). Surgical time was 53.6 ± 17.3 min when patients underwent surgery 7 days or less following the trauma, compared to 63.8 ± 14.8 min when they underwent surgery more than a week after the trauma. However, there were no differences in the other variables studied: rehabilitation treatment time (p = .35), MEPS (p = .08), flexor deficit (p = .16), extensor deficit (p = .16), complications (p = .72), ossifications (p = .46), shoulder-hand syndrome (p = 1), paraesthesia (p = 1), residual pain (p = .38), stiffness (p = 1) and reoperation rate (p = .15). The results of these three findings are shown in Table 1.
Inferential analysis.
| Variable | Statistical test used | Type of approach | Type of anchor | Intervention before or after 7 days |
|---|---|---|---|---|
| Surgical time | U Mann Whitney/Kruskall Wallis | .135 | .27 | .03 |
| Rehabilitation time | Student’s t/ANOVA | .97 | .37 | .35 |
| MEPS | Student’s t/ANOVA | .009 | .25 | .08 |
| Flexion deficit | Student’s t/ANOVA | .91 | .52 | .16 |
| Extension deficit | Student’s t/ANOVA | .6 | .63 | .16 |
| Presence of complications | X2 test | .008 | .05 | .72 |
| Ossification | X2 test | 1 | .14 | .46 |
| Shoulder hand syndrome | X2 test | 1 | .46 | 1 |
| Paraesthesia | X2 test | .38 | .83 | 1 |
| Residual pain | X2 test | < .05 | < .05 | .38 |
| Stiffness | X2 test | 1 | .24 | 1 |
| Reintervention | X2 test | 1 | .68 | .15 |
There were differences in terms of MEPS score (p = .009). This difference was in favour of the Boyd-Anderson approach, with a score of 89.7 ± 10.6, compared to 81.4 ± 13.6 for the anterior approach. There were differences in complication rates (p = .008), 11.1% for the Boyd Anderson approach versus 41.2% for the anterior approach. Similarly, there were differences in terms of residual pain (p = .000), which was 4.8% for the Boyd Anderson approach versus 41.2% for the anterior approach. There was a difference in surgical time (p = .03), which was 53.6 ± 17.3 min when the patients were operated 7 days or less following the trauma, versus 63.8 ± 14.8 min when they underwent surgery later than one week following the trauma.
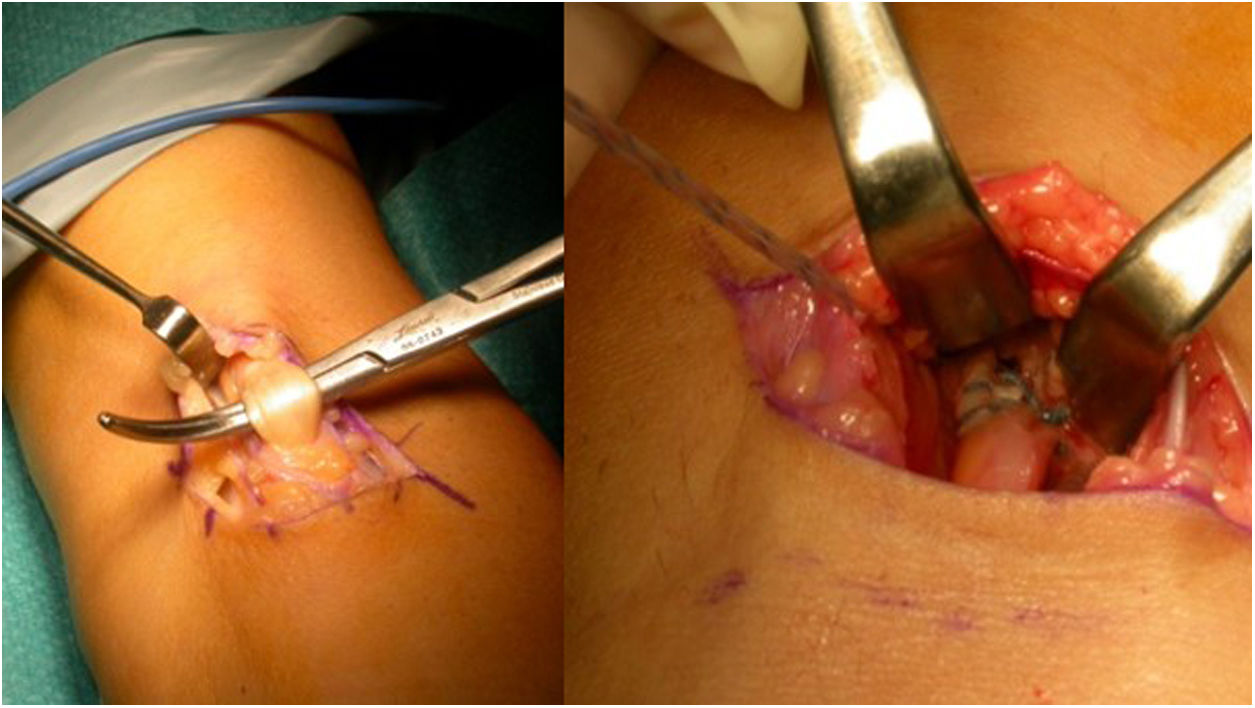
There were complications in 17.5% of cases. Of these, there were 3.8% heterotopic ossifications (3 cases), one with radioulnar synostosis; one case of shoulder-hand syndrome, 2.5% paraesthesias (2 cases), 2.5% radial neuroapraxias (2 cases) and there were no cases of infection. Significant residual pain was reported in 12.5% of the patients, including one case with recurrent residual tendinitis. We found 6.3% cases with stiffness (5 cases). We also found one case of retractable scar, one case of fibrosis, which required arthrolysis, and we found one case with an implant placed in the humeral condyle. We reoperated 5 cases (6.25%): one resuture with Orthoadapt (equine pericardium), two resutures with fascia lata, one with arthrolysis and one resuture after removal of the anchor from its previous insertion, in the humeral condyle (Fig. 6B). This case shows the importance of a correct second posterolateral approach to adequately expose the bicipital tuberosity. This approach should be distal to the area of palpation, when pronosupination of the radial head is performed. The more proximal approach could result in errors, with placement of the implant in the capitellum: in this case the patient had to be reoperated, the implant removed, the approach widened, and the biceps anchored in the bicipital tuberosity. We believe that correct approaches are important, as is an adequate learning curve for the procedure. Another important aspect is to correctly pass through the space between the radius and ulna. Falling outside this space would not lead to the correct insertion point, increasing the risk of concomitant injuries. And in this step, avoid scraping the surface of the ulna with the forceps to reduce the risk of synostosis.
DiscussionConservative treatment of distal bicipital ruptures would be reserved for people with low functional demand, comorbidities, and risk factors. However, it results in decreased strength and flexion and supination resistance. To be specific, it results in a 40%–74%20 loss of supination, a 30%–88%20 decrease in flexion and a 15% decrease in grip strength.7 Anatomical reinsertion results in the fastest recovery with 93.3 points on the MEPS,21 up to 13% greater supination strength and 32% greater supination resistance than if the patient did not undergo surgery. Brachial transfer is another option, but this involves a functional loss in supination of 40% and a 17% loss in flexion. Legg et al.22 reviewed a series of 50 cases of biceps ruptures: 40 treated conservatively and 10 surgically. For this author, orthopaedic treatment resulted in a flexion and supination deficit of 92.4% and 70.65% (p = .01 and p = .004, respectively). The operated patients had better QuickDASH (p = .02) and MEPS scores (p = .014) than the non-operated patients. In the experience of Santos et al.,23 with 40 cases, 16 of which were treated conservatively versus 24 operated, better results were shown for the operated patients in MEPS (p = .001), duration of the procedure (p = .01), return to work (p = .05), pain (p = .003) and decrease in isokinetic strength (p = .005). In this paper we review the experience with 80 cases who underwent tendon reinsertion. We consider that being high-demand workers with this injury, anatomical reinsertion is the best of the treatment options.
Two fundamental procedures have been described: the double approach, or Boyd-Anderson technique,12 which would reproduce 73.4% of the natural bicipital insertion, and the single approach, Henry’s approach,16 which by means of anterior antebrachial access would reproduce 9.7% of the natural bicipital insertion.24 In this study, we used the double approach in 63 cases and the single anterior approach in 17 cases. As described above, in our series there were differences in MEPS score (p = .009) in favour of the Boyd-Anderson approach, with a score of 89.7 ± 10.6 compared to 81.4 ± 13.6 for the anterior approach. There were also differences in complications (p = .008), at 11.1% for the Boyd Anderson approach versus 41.2% for the anterior approach. Of these complications, residual pain (p = .000) was noteworthy, which was 4.8% for the Boyd Anderson approach compared to 41.2% for the anterior approach. Analysing the literature comparing both procedures, we observe contrasting references. Watson et al.3 reviewed 24 articles. In their study of 494 patients, they described a complication rate of 24.5%, 23.9% for the single approach and 25.7% for the double approach, and therefore there were no statistically significant differences (p = .32), unlike our series, where there were more complications with the anterior approach. The rate of antebrachial cutaneous nerve neuroapraxia was 11.6% for the single approach and 5.8% for the double approach (p = .02). They also described heterotopic ossification, which was 3.1% for the single approach versus 7% for the double approach (p = .06). Stiffness was 1.8% for the single approach versus 5.7% for the double (p = .01). Finally, other data they collected were re-rupture (1.8% for the single approach vs. 1.2% for the double approach), infection (1.2% for the single approach vs. 0% for the double approach) or synostosis (0% for the single approach vs. 2.3% for the double approach).
On the other hand, Grewal et al.25 conducted a comparative study of 47 patients undergoing the single versus 44 patients undergoing the double approach. They observed no difference in outcomes on the American Shoulder and Elbow Surgeons Scale (ASES), either in pain (p = .4), function (p = .1) or DASH (p = .3). There was a difference in isometric flexion strength (p = .01) in favour of the double incision treated group. The incidence of neuropraxia was significantly higher for the single approach group (p < .001). For Júnior et al.26 the double approach resulted in a flexion deficit of 9.6% and a supination deficit of 28.9%. Cohen et al.,27 in their review of 33 cases comparing Boyd Anderson with the single approach with Endobutton, found no significant difference in terms of DASH (6.1 vs 5.9 points, p = .69), patient satisfaction (p = .457) and prevalence of complications (39.3% vs 32%, p = .59).
For Giacalone et al.28 the double approach is safe and less aggressive than Henry's approach. However, for Haverstock et al.29 the single approach would not reproduce the correct insertion at the ulnar border of the bicipital tuberosity, nor would it restore the pulley mechanism of the natural bicipital insertion, which the double approach would achieve, allowing an improvement in supination20 and flexion strength (104% compared to 94% for the single approach20). Stockton et al.30 challenged this idea by finding in their series of 37 patients a 20% functional improvement in supination over the supination achieved with the double approach. The correct placement would be at the radial border between 9° and 117°. Hansen et al.,31 in a study of 27 patients, observed that supination strength was significantly lower than that of the healthy arm (p < .05), achieving a DASH of 10.7 points. He observed a decrease in strength (p < .016) in fast concentric supination with rotation angles at 10° (85% ± 20%), 45° (80% ± 22%) and 80° (82% ± 21%) and slow at 45° (83% ± 21%). For other authors, this single approach is very useful. Thus, for Smith and Amirfeyz,32 in their study of 22 patients, early mobilisation did not entail an increased risk of repair failure, discomfort, or wound involvement, with a DASH of 2.7 points and MEPS score of 97.8, although the single transverse incision had the highest risk of associated nerve injury. Similarly, for Pangallo et al.33 the anterior approach, in their study of 18 patients, achieved 80% satisfaction and 80% excellent results on the MEPS scale, with a decrease in supination of only 5%-10%, no re-ruptures and one case of hypoaesthesia. Ramazzini et al.,17 in their series of 79 anterior approach cases, obtained a MEPS score of 95.2, with no difference between placing one or two anchors. Kodde et al.34 also found no difference in supination strength deficit between the two approaches (p = .65). The Mayo Clinic20 would prefer the double approach due to its lower cost, lower rate of neurological complications and greater ability for anatomical reconstruction for these surgeons using this procedure. For Barret et al.35 this double approach with early postoperative mobilisation achieved a strength ratio in flexion up to 94% ± 8% of the normal value and in supination of up to 90.5% ± 12% of the normal value. Even with MEPS scores of 96.3, it was also the preferred indication for Tarallo et al.36 in trauma of less than 2 weeks in young people. Kale et al.37 have recently described a double anterior approach with good outcomes, especially if combined with repair of the bicipital aponeurosis.38
Regarding the anchoring procedure, in our experience there were differences in terms of complications (p = .05), which were slightly more frequent with threaded anchors and residual pain (p < .05), in half of the cases treated with cortical buttons. Recordon et al.,24 who analysed 19 cases treated with the Endobutton® system and 27 cases treated with transosseous sutures, observed that there was no statistically significant difference in MEPS score between the two groups. For Watson et al.3 the complication rate was lower for intraosseous screws (p < .01), and for Grewal et al.25 the rate of complications after cortical button fixation was three times higher than in the transosseous suture cases and two times higher than in the suture anchor cases. Caekebeke et al.,39 who compared two types of anchors, one with Poly-L-lactide (PLLA) (12 cases) versus Poly-EtherEtherKetone (PEEK) anchors (11 cases), in a series of 23 patients, found a VAS for pain that improved to .2 points in the PLLA group and .7 points in the PEEK group, with a DASH of 5.4 points and MEPS score of 98.7, respectively. Kodde et al.,34 in their review of 40 articles involving 1,074 patients, observed that there was no difference between four fixation systems in terms of supination strength deficit (p = .34). Prabhu et al.40 recommended the Endobutton® system, with tenodesis screw, with good results. Witkowski et al.41 used the TightRope® cortical button, with polyethylene sutures, with an MEPS score of 95 ± 10.42 and QuickDASH score of 8.6 ± 18.04. Woodall et al.42 recommended reinsertion with two intramedullary anchors, which would avoid suspensory fixation supported on the contralateral cortex, after bicortical drilling. Lang et al.,43 in their series of 47 patients, recommend transosseous fixation with a double approach, as it would reduce surgical costs and time. Huynh et al.44 reported lower flexion and supination strength, compared to the healthy arm, when cortical buttons and a single approach were used. Finally, Alech-Tournier et al.45 recommend fixation with the ToggleLoc system with ZipLoop, with a strength deficit of 23.9% compared to the other side. Fixation with bone tunnels and double approach is associated with an increased risk of synostosis.
Treatment with Achilles, and semitendinosus, rectus internus or tibialis anterior allografts has been offered for chronic ruptures of more than 4 weeks. From the series by Snir et al.,46 an MEPS was obtained from 18 cases, which increased from 43.1 preoperatively to 94.2 postoperatively (p < .001). On the other hand, for Morrey et al.,47 in very retracted cases, repair at 90° flexion would avoid reconstruction with grafts, even with an MEPS score of 100. For Phadnis, in cases of irreparable ruptures, Achilles allograft48 and fixation with Endobutton fixation resulted in a QuickDASH of 4 points, an MEPS score of 92.9, as well as improvements compared to the preoperative situation, with a p < .001. Alternatives have been described such as the patient's own fibrous lacertus,20,49 triceps,50 hamstring51 or acellular dermis mesh.52 In our series there were three cases of re-rupture requiring subsequent plasty with equine pericardium or cadaver fascia lata.
Finally, the complications of this technique are referred to in the literature. For Ford et al.53 the rate of major complications is 7.5%, and the reintervention rate is 4.5%. The overall complication rate of 7.5%–27%1 is similar to the rate of 17.5% found in our series. Although described earlier in this discussion, two cases (2.5%) of paraesthesia and two cases (2.5%) of radial neuroapraxia were of note in our series. They occurred in patients treated using the single approach. Ward et al.1 described paraesthesia in the lateral cutaneous antebrachial nerve (9.8%), 54 palsy in the posterior interosseous nerve (3.4% in the double approach compared to .8% in the single approach)55 and neurological complications up to 5.6%, primarily affecting the radial and median nerves. Perhaps this risk of injury is due to the distance of less than 11.6 mm between the reamers and the nerve, as proposed by Tat et al..56 To avoid this injury, it is useful to know the safety zone around the bicipital tuberosity. To be specific, 1 cm proximal to the bicipital tuberosity, as described by Becker et al.57 However, Ward et al.1 described a low risk of heterotopic ossification, with radio-ulnar synostosis of 7%, with a 50% risk of recurrence after excision. Ford et al.53 corroborate this direct relationship between the double approach and synostosis, with a statistical significance of p = .0003 and odds ratio of 19. This synostosis, which is 7.2% for Amin et al.,54 could be prevented with indomethacin or, if established, could be treated surgically by interposition of a posterior interosseous flap with antegrade flow, as Martínez et al.58 report. In our experience there was no difference in the rate of ossification or synostosis between the two procedures. On the other hand, Ward et al.1 described a risk of transient paraesthesia. For Garon and Greenberg59 the complication rate was 15%–35%, irrespective of the approach used, although the anterior approach had a higher risk of lateral cutaneous antebrachial nerve involvement. For Ford et al.53 this relationship between the single approach and antebrachial nerve injury is significant (p < .001). For Guerra-Vélez et al.60 the nerve palsy was of the radial nerve, it was transient and in one case, compared to the 12 cases treated using the single approach. The Boyd Anderson approach was also associated with neurological complications, such as radial nerve palsy, which Montiel-Giménez et al.61 reported in 14% of their series, although they subsequently recovered spontaneously. Other complications were tunnel lysis, venous thrombosis,62 mobilisation of the implant, revision in 5.4%, infection in 1.1% and nerve damage in .6%.22,63 To avoid radial nerve injury in the double approach, the arm should be kept in pronation, since in neutral prosupination the posterior interosseous nerve is 4.2 cm from the radiocapitellar joint, 5.6 cm in pronation and 3.2 cm in supination.20 In cases of re-rupture, as mentioned above, repair should require allografts, achieving a DASH score of 4.4 and extension deficit of ―12°.48
The use of endoscopy as an alternative has recently been postulated to assist the technique64,65 or even as a procedure, with various portals. For Bhatia et al.66 portals 1 (parabicipital) and 5 (distal posterior) may be safe to access the distal bicipital insertion. Portals 2 (risk of posterior interosseous nerve injury; p = .003) and 3 (risk of superficial radial nerve injury; p = .036) are not safe for this technique.
LimitationsLimitations of the study designThis is a cohort study in which a historical cohort initially treated using a double approach was considered instead of using a methodology less prone to bias such as a randomised clinical trial. This limitation was suggested by the ethics committee after analysing the benefits observed in the pilot sample of our study and confirming that they were consistent with the results in the literature. The initial sample of 40 patients was a large group when compared to other series in the literature. The committee decided that we should not create a prospective control group without the results of our historical cohort of 40 patients. Thus, the alternative of using a historical cohort was suggested to us. To avoid the potential biases of this design, special emphasis was placed on performing the same surgical procedures in the prospective cohort as in the historical group, i.e., tendon reinsertion only, considering a minimum number of 15 patients in each approach group, with similar numbers between transosseous and anchor reinsertion. Moreover, achieving this objective was not difficult, since the historical cohort was recent, and therefore our department’s surgical protocols had not changed; thus, the surgical methodology used in both cohorts was identical, which solved the main problem usually posed by using historical cohorts. Similarly, the clinical data of the patients who underwent surgery in the prospective cohort was initially collected by one of the co-authors without knowledge of the results obtained in the group who underwent the double approach (historical cohort), which makes it possible to compare the results of the two groups more objectively and to draw or not draw conclusions. We increased the sample slightly to have more cases.
Other limitationsBecause the surgery was performed by a team of orthopaedic surgeons this could lead to different results depending on their familiarity with the technique and the learning curve. Similarly, we consider that having opted for different anchor procedures may be a limitation, which may alter the results obtained. On this point, these anchors were commercially and technologically developed over the follow-up period, which explains why various types of anchors were used, as some of those initially used were not in use in the same commercial presentation by the end of this study, or cortical buttons were tried, as decided by one of the surgeons. We believe that further comparative studies should be conducted between the two types of approach.
AdvantagesWe believe that there was a sufficiently large number of cases and subject matter, for this paper to be of interest. We believe that the clinical assessment with isokinetics, the assessment of various surgical aspects, and the literature review will generate interest in this subject.
ConclusionsReinsertion of the distal biceps aims to restore the flexion and supination strength lost with this injury. In our experience the best clinical results were achieved with the double approach, with a lower complication rate versus the single approach. There were no major differences in our series between the different types of fixation. The Boyd-Anderson approach was less time-consuming than the single approach. This procedure was not without complications, the most representative being ossification and neurapraxia. In our experience, choosing the double approach was a protective factor against nerve injury.
Level of evidenceLevel of evidence II.
FundingThe authors received no funding for this paper.
Conflict of interestsThe authors have no conflict of interest to declare.
We would like to thank Sres. Antonio Muñoz, Francisco Gonzálvez, José Villanueva and Sra. Inmaculada Ruíz, members of our hospital’s operating theatre staff, for the interoperative photographs for this paper.
Please cite this article as: Jiménez-Martín A, Santos-Yubero FJ, Najarro-Cid FJ, Navarro-Martínez S. Reparación de roturas de bíceps distal, experiencia a propósito de 80 casos. Rev Esp Cir Ortop Traumatol. 2021;65:363–373.