Cardiac myxoma is an important but uncommon cause of stroke in younger patients. Few published case series analyse the frequency and clinical presentation of neurological complications in patients with myxoma.
ObjectiveTo list all neurological complications from cardiac myxoma recorded in our hospital in the past 28 years.
Patients and methodsWe retrospectively reviewed the neurological manifestations of cardiac myxoma in patients treated in our hospital between December 1983 and March 2012.
ResultsOf the 36 patients with cardiac myxoma, 8 (22%) presented neurological manifestations. Half were women and mean age of patients was 52.4±11.6 years. Sudden-onset hemiparesis was the most frequent neurological symptom (63%). Established ischaemic stroke was the most common clinical manifestation (75%), followed by transient ischaemic attack. The most commonly affected territory corresponded to the middle cerebral artery. Myxoma was diagnosed by echocardiography in all cases. Mean myxoma size was 4.1cm and most of the tumours (63%) had a polypoid surface. All tumours were successfully removed by surgery. There were no in-hospital deaths.
ConclusionsCardiac myxomas frequently present with neurological symptoms, especially ischaemic events (established stroke or transient ischaemic attack), in younger patients with no cardiovascular risk factors. The anterior circulation is more frequently affected, especially the middle cerebral artery. Echocardiography can facilitate prompt diagnosis and early treatment of the lesion.
El mixoma cardiaco es una causa poco frecuente pero importante de infarto cerebral en pacientes jóvenes. Existen pocas series de pacientes que analicen la frecuencia de las manifestaciones neurológicas en pacientes con mixoma y su presentación clínica.
ObjetivoConocer las complicaciones neurológicas del mixoma cardiaco en nuestro hospital durante los últimos 28 años.
Pacientes y métodosRevisión retrospectiva de las manifestaciones neurológicas de 36 pacientes operados de mixoma cardiaco con confirmación patológica en nuestro centro desde diciembre de 1983 hasta marzo del 2012.
ResultadosOcho de los 36 pacientes con mixomas cardiacos (22%) intervenidos en nuestro centro presentaron clínica neurológica. El 50% eran mujeres y la edad media±desviación estándar de 52,4±11,6 años. El síntoma neurológico más frecuente fue la hemiparesia de aparición brusca (63%). El ictus isquémico establecido fue la manifestación clínica más frecuente (75%), seguido del accidente isquémico transitorio. El territorio más afectado fue el de la arteria cerebral media. En todos los casos se alcanzó el diagnóstico del tumor mediante ecocardiografía. El tamaño medio del mixoma fue de 4,12cm. La mayoría (63%) presentaba una superficie polipoide. Todos los tumores fueron resecados quirúrgicamente con éxito. No hubo muertes hospitalarias.
ConclusionesLos mixomas cardíacos comienzan frecuentemente con manifestaciones neurológicas, en particular como eventos isquémicos (AIT o ictus establecidos) en pacientes jóvenes y sin factores de riesgo cardiovascular. El territorio anterior, en especial la arteria cerebral media, suele estar más frecuentemente afectado. La ecocardiografía puede facilitar el diagnóstico y permitir un tratamiento precoz de la lesión.
Cardiac myxoma is the most frequent primary cardiac tumour, accounting for as many as 80% of all cardiac tumours described in surgical series.1 It is generally believed that they arise from multipotent mesenchymal cells of the endocardium.2 Although up to 10% of all patients may be asymptomatic,3 most patients present one of the symptoms in the classic triad: intracardiac obstruction (50%–70%), peripheral or central systemic embolism (16%–45%) and constitutional symptoms (fever, asthenia, and weight loss) in as many as 50%.3
Neurological manifestations of cardiac myxomas are frequently due to cerebrovascular complications, which occur in 25% to 45% of all cases and may appear as the initial symptom.4 The most frequent stroke presentation was ischaemic stroke due to emboli from the myxoma. Strokes may be recurrent if the tumour is not diagnosed and treated. Presentation as haemorrhagic stroke is infrequent, and this tends to be associated with the formation of aneurysms in the cerebral circulation.
This series describes neurological manifestations in patients with cardiac myxomas which we observed in our hospital over the last 28 years. Here, we analyse morphology and ultrasound data from myxomas and review literature on this topic.
Patients and methodsWe reviewed anatomical pathology reports dating from December 1983 to March 2012 and found 36 patients with an anatomical pathology diagnosis of cardiac myxoma (mean age±standard deviation=56±13 years, age range 26–80 years, 64% women). These clinical histories were revised in detail and researchers recorded presence or absence of neurological conditions associated with the myxoma. In each of these cases, we recorded the patient's cardiovascular risk factors, drug therapy prior to admission, neurological symptoms, neuroimaging tests performed, diagnosis, myxoma size and type, and the patient's clinical and functional outcome. The last parameter was obtained by estimating mRS at 6 months. Statistical testing was completed using SPSS software version 17.0 (IBM Corporation, Armonk, New York, USA).
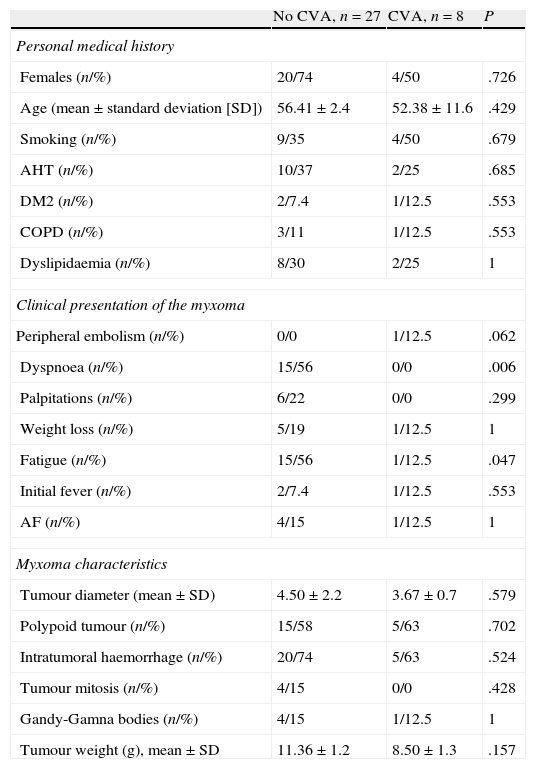
Results (Tables 1 and 2)Personal historyOf the 36 patients with myxoma, 8 (22%) presented neurological manifestations. Of these patients, mean age was 52.4±11.6 years (range, 26–64), and 50% were women. Two patients (25%) had no cerebrovascular risk factors and the remaining 75% presented one or more known risk factors. Within this group of 8, 4 were smokers (50%), 3 had hypertension (37.5%), 3 had type 2 diabetes (37.5%), 2 had dyslipidaemia (25%), 2 were obese (25%), 1 had chronic obstructive pulmonary disease (12.5%) and 1 had atrial fibrillation (12.5%). There were no significant differences between medical histories in the patient group with myxoma and neurological manifestations and the other patients (myxoma without neurological manifestations).
Baseline clinical characteristics.
| No CVA, n=27 | CVA, n=8 | P | |
| Personal medical history | |||
| Females (n/%) | 20/74 | 4/50 | .726 |
| Age (mean±standard deviation [SD]) | 56.41±2.4 | 52.38±11.6 | .429 |
| Smoking (n/%) | 9/35 | 4/50 | .679 |
| AHT (n/%) | 10/37 | 2/25 | .685 |
| DM2 (n/%) | 2/7.4 | 1/12.5 | .553 |
| COPD (n/%) | 3/11 | 1/12.5 | .553 |
| Dyslipidaemia (n/%) | 8/30 | 2/25 | 1 |
| Clinical presentation of the myxoma | |||
| Peripheral embolism (n/%) | 0/0 | 1/12.5 | .062 |
| Dyspnoea (n/%) | 15/56 | 0/0 | .006 |
| Palpitations (n/%) | 6/22 | 0/0 | .299 |
| Weight loss (n/%) | 5/19 | 1/12.5 | 1 |
| Fatigue (n/%) | 15/56 | 1/12.5 | .047 |
| Initial fever (n/%) | 2/7.4 | 1/12.5 | .553 |
| AF (n/%) | 4/15 | 1/12.5 | 1 |
| Myxoma characteristics | |||
| Tumour diameter (mean±SD) | 4.50±2.2 | 3.67±0.7 | .579 |
| Polypoid tumour (n/%) | 15/58 | 5/63 | .702 |
| Intratumoral haemorrhage (n/%) | 20/74 | 5/63 | .524 |
| Tumour mitosis (n/%) | 4/15 | 0/0 | .428 |
| Gandy-Gamna bodies (n/%) | 4/15 | 1/12.5 | 1 |
| Tumour weight (g), mean±SD | 11.36±1.2 | 8.50±1.3 | .157 |
Risk factors and neurological manifestations among patients with myxoma.
| Patient | Sex | Age | CVRF | Neurological syndrome | Neuroimaging study | Diagnosis | Time elapsed between stroke and diagnosing the myxoma | mRS at 6 months |
| 1 | M | 54 | Smoker, high cholesterol, obesity | Loss of vision in right temporal visual fields, bilateral frontal headache, nausea | CT: left occipital ischaemic infarct | Ischaemic stroke in left PCA territory | Simultaneous | 2 |
| 2 | M | 26 | No | Right-sided weakness | CT: left frontoparietal infarct | Ischaemic stroke in left MCA territory | Simultaneous | 4 |
| 3 | F | 56 | Lacunar stroke | Self-limiting motor aphasia | Not completed | TIA in left MCA territory | 4 years | 0 |
| 4 | M | 58 | AHT, NIDDM, smoker | Disorientation in time and space, left arm paraesthesia, left-sided hemiparesis | CT: right parietal infarct | Ischaemic stroke in right MCA territory | Simultaneous | 2 |
| 5 | F | 52 | NIDDM, dyslipidaemia | Vertigo, left-sided paraesthesia | CT: normalMRI: ischaemic lesions in vertebrobasilar territory | Minor ischaemic stroke in the vertebrobasilar territory | Simultaneous | 0 |
| 6 | M | 60 | Dyslipidaemia, smoking, alcohol abuse, obesity | Vertigo and self-limiting bilateral loss of vision | Not completed | TIA in vertebrobasilar territory | Simultaneous | 0 |
| 7 | F | 49 | AHT, smoker | Mixed aphasia and right-sided hemiparesis | CT: left temporoparietal infarct | Ischaemic stroke in left MCA territory | Simultaneous | 4 |
| 8 | F | 64 | AHT, DM, smoker, alcohol abuse | Left-sided hemiparesis | CT: right parietal infarct | Ischaemic stroke in right MCA territory | Simultaneous | 3 |
MCA: middle cerebral artery; PCA: posterior cerebral artery; TIA: transient ischaemic attack; DM: diabetes mellitus; NIDDM: non-insulin-dependent diabetes mellitus; CVRF: cardiovascular risk factors; AHT: arterial hypertension; mRS: modified Rankin Scale; MRI: magnetic resonance imaging; CT: computed tomography.
Patients with myxoma and no neurological symptoms more commonly presented dyspnoea and fatigue than patients with neurological manifestations (56, 0%; 56, 12.5%; P=.006 and .047, respectively). For all other classic manifestations (peripheral embolism, palpitations, weight loss, fever, AF), there were no differences between groups.
Anatomical pathologyAll patients received a confirmed diagnosis of left atrial myxoma by means of an anatomical pathology study of surgically extracted samples.
Mean tumour weight was 8.5±1.3g. The maximum tumour diameter ranged from 3 to 5cm, with a mean diameter of 4.12cm. Five cases (63%) displayed polypoid morphology. None of the myxomas had surface thrombi.
Microscopic examination found that 5 myxomas presented intratumoral haemorrhage (63%). There was one case of Gandy-Gamna bodies (12.5%), and none of the samples displayed tumour mitosis.
The study found no significant differences between patients with myxoma and neurological manifestations and patients with myxoma only.
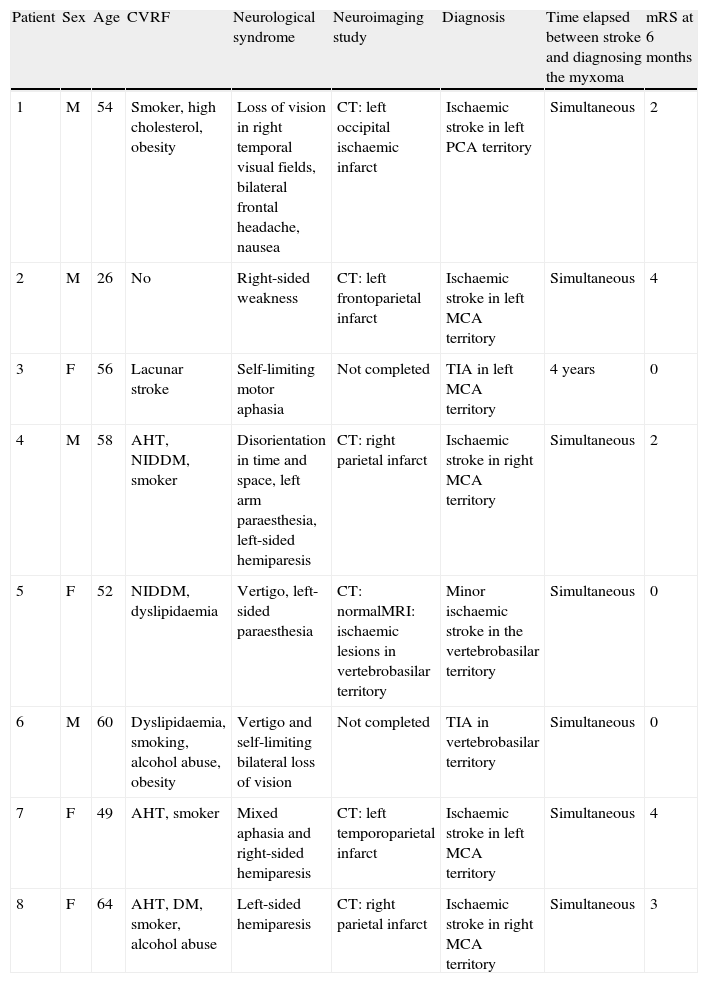
Clinical presentation of neurological symptomsIn 7 of 8 patients (87.5%) with atrial myxoma and neurological symptoms, these symptoms appeared as the initial manifestation of the lesion. The most common neurological symptom was sudden-onset unilateral motor deficit, which occurred in 5 patients (63%). Two patients (25%) presented headache, 2 (25%) decreased visual field, 1 (12.5%) language disorder, 1 (12.5%) loss of consciousness, and 1 (12.5%) experienced an episode of dizziness and blurred vision.
The remaining patient (patient 3) reported non-specific initial symptoms (fever, arthralgia, and headache); a few years before, he had suffered an infarct in the left caudate nucleus that resulted in dysarthria. It was attributed to small-vessel disease even though the patient was not hypertensive and had no other cerebrovascular risk factors.
NeuroimagingNeuroimaging studies were available for 6 of the 8 patients (75%: simple CT in 5 cases, simple CT plus MRI in 1). There were no records of neuroimaging studies for patients 3 and 6, probably because the syndrome was self-limiting.
In all 6 patients with neuroimaging studies, imaging results correspond to ischaemic stroke in an acute or subacute stage. There was no evidence of haemorrhagic stroke or findings that would point to other entities. The infarct locations in patients with neuroimaging studies were the anterior circulation in 4 patients (66%) and the posterior circulation in 2 (33%). The most frequently affected territory was that of the middle cerebral artery (4 cases, 2 on each side), followed by the posterior cerebral artery (1 case), and 1 case of lacunar stroke of the perforating branches of the basilar artery. In the 2 patients with TIA, clinical signs pointed to affectation of the middle cerebral artery (MCA) in 1 case, and to the basilar artery in the other.
EchocardiogramAll patients were diagnosed by transthoracic echocardiogram, and all myxomas were located in the left atrium. In 3 patients (37%), the tumour prolapsed towards the mitral valve during diastole, but without obstructing that valve. Ventricular function was preserved in all cases (greater than 60%). Transthoracic echocardiography showed left atrial myxomas of varying sizes; mean diameter was 3.67±0.7cm (range, 3–4cm).
TreatmentThe 6 patients with stroke and the 2 with TIA were treated with single anti-platelet therapy, beginning after the brain CT was performed (for established stroke), or after symptoms had resolved (for cases of TIA). Once the atrial myxoma had been diagnosed by ultrasound, antiplatelet drugs were replaced with anticoagulants in 5 patients (63%), while anticoagulants were associated with antiplatelet drugs in the other 3 patients.
In all cases, the left atrial myxoma was successfully removed. Mean time between the onset of neurological symptoms and the surgical procedure was 16.3 days (range, 1–30 days) in all cases except patient 3 who had suffered a TIA 4 years before being diagnosed with myxoma. The mean myocardial ischaemia time during surgery was 37±16min, with a mean extracorporeal circulation time of 74±24min. Half of the myxomas were resected through the left atrium and the rest through the right atrium by passing through the interatrial septum. Mean hospitalisation time in the intensive care unit was 2.38±0.5 days, and patients remained in hospital after the surgery a mean of 16.6±7.3 days. No in-hospital deaths were recorded. Upon discharge, 3 patients were taking anticoagulants (1 had atrial fibrillation); another also had antiplatelet drugs, and 2 were taking antiplatelet drugs only. One patient (12.5%, patient 2) required permanent implantation of a pacemaker in the early postoperative period, and 2 patients (25%) had atrial arrhythmias during the same period.
Outcomes and follow-upPatients with stroke or TIA were referred to outpatient neurology departments upon discharge, and they were examined in those departments at the 6 month mark. Estimated mRS scores at 6 months were 0 in 3 cases (Patients 3, 5, and 6); 4 in 2 cases (patients 2 and 7); 2 in 2 cases (patients 1 and 4); and 3 in the last case (patient 8).
These 8 patients have also been monitored by a cardiovascular surgery department over periods ranging from 1.5 to 29 years. During this time, one patient died of breast cancer 5 years after the myxoma surgery. Two more patients also developed tumours in other locations (breast cancer, peripheral neuroma). One patient experienced atrial arrhythmia during the follow-up period. There were no recorded cases of remitting cardiac myxoma in these patients.
None of the 8 patients presented cardiological symptoms, and none had further neurological complications subsequent to being diagnosed and surgically treated for atrial myxoma.
DiscussionThis descriptive study includes the neurological manifestations recorded in a total of 3 patients who underwent surgery for atrial myxoma in our hospital between December 1983 and March 2012. Although myxomatous emboli are a well-known and relatively frequent cause of stroke in young patients, most of the published descriptions correspond to isolated clinical cases. In the 2 largest series, Pinede et al. and Lee et al. provide their respective analyses of 23 and 11 patients with myxoma and cerebral infarct.3,5 After searching different electronic medical databases, we recovered only 1 such series published in Spain, describing 9 cases recorded in Hospital Vall d’Hebron.6
The frequency of neurological manifestations was 22% in our patient series, and this percentage is similar to that in other published studies.4,5 Females made up 50% of the group, unlike in other prior series7 and our total myxoma population which both show clear female predominance (63%). Mean age of patients with atrial myxoma and neurological symptoms was lower than that in the general series (52.4±11.6 years vs 56±13 years). This tendency is also present in earlier series, indicating that potentially emboligenic myxomas show earlier clinical manifestations than other types, due to the presence of embolic events.
In our series, established stroke was the most frequent manifestation of atrial myxoma (75% of all cases), vs 25% in TIA. These data differ from the largest Spanish series published to date, which indicated TIA as the predominant neurological manifestation of atrial myxomas.6 However, other large series show percentages similar to ours here. This fact may be related to the different pathological characteristics displayed by myxomas. Pathological samples in our series showed a predominance of polypoid myxomas, which are more prone to embolism.3,5 There were no cases of thrombus, which allows us to hypothesise that a large percentage of the embolic events resulted from the fracturing of small tumour fragments that would not have been prone to spontaneous lysis. The series cited above6 did not specify the morphological and pathological characteristics of myxomas in patients with neurological symptoms, and it therefore cannot be compared to our study.
Regarding topography of the neurological lesion, 5 of the 8 cases affected the anterior territory, and the remaining 3, the posterior territory. All cases except 2 involved medium-calibre blood vessels, specifically the middle cerebral artery (5 cases) and the posterior cerebral artery (1 case). The 2 remaining cases corresponded to symptoms indicative of perforating branch involvement in the posterior territory. In the most representative earlier series,5,6 the anterior/posterior circulation ratios were 7:4 and 6:3 respectively. Other less common cerebrovascular myxoma presentations, such as progressive dementia, retinal infarcts, or multiple embolisms, were not recorded in our patients.
In all cases in our series, neurological manifestations (TIA or stroke in this case) presented as the initial symptom of myxoma. In 7 of 8 cases, the tumour was diagnosed during the patient's hospital stay, and surgically removed within 30 days of the event. In the case of Patient 3, there was a significant delay between onset of neurological symptoms and the diagnosis and treatment of atrial myxoma (4 years). All cases were diagnosed using echocardiography, which attests to the need for including this technique in routine examinations following a stroke of unknown origin. None of the patients experienced myxoma recurrence, and 30-day surgical mortality was non-existent. These data support the importance of early diagnosis and treatment of this condition before new embolic or cardiac complications appear. Likewise, in the modern era of thrombolytic treatment,8,9 identifying strokes secondary to this condition may help us understand the specific response in this patient subgroup.
ConclusionsCardiac myxomas frequently appear in conjunction with neurological manifestations, especially ischaemic events (TIA or established stroke) in young patients with no cardiovascular risk factors. The anterior territory, and the middle cerebral artery in particular, are the most frequently affected areas. Echocardiography may facilitate diagnosis and enable early treatment of the myxoma.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Pérez Andreu J, Parrilla G, Arribas JM, García-Villalba B, Lucas JJ, Garcia Navarro M, et al. Manifestaciones neurológicas de los mixomas cardiacos. Experiencia en un centro de referencia. Neurología. 2013;28:529–534.