Idiopathic toe walking, a differential diagnosis for neurological and orthopaedic disorders, has been associated with neurodevelopmental alterations. Neurodevelopmental assessment at early ages using specific tests may improve management and follow-up of these patients. The aim of our study is to analyse the neurodevelopmental characteristics of preschool idiopathic toe-walkers (ITW) by comparing them to a control group.
MethodOur descriptive cross-sectional study compared possible risk factors, neurodevelopmental characteristics, and scores on the Child Neuropsychological Maturity Questionnaire (CUMANIN) between a group of 56 ITWs aged 3-6 and a control group including 40 children.
ResultsThe proportion of males was significantly higher in the ITW group (P=.008). The percentage of patients with a family history (P=.000) and biological risk factors during the perinatal period (P=.032) was also higher in this group. According to the parents’ reports, motor coordination in ITWs was significantly poorer (59%; P=.009). ITWs scored significantly lower on CUMANIN subscales of psychomotricity (P=.001) and memory (P=.001), as well as in verbal development (P=.000), non-verbal development (P=.026), and overall development (P=.004). Foot preference was less marked in the ITW group (P=.047).
ConclusionsThe neurodevelopmental characteristics of our sample suggest that idiopathic toe walking is a marker of neurodevelopmental impairment. However, further studies are necessary to confirm these findings.
La marcha de puntillas idiopática, considerada un diagnóstico de exclusión de alteraciones neurológicas y ortopédicas, se ha relacionado con alteraciones en diversas áreas del desarrollo neuromadurativo. La evaluación del neurodesarrollo en edades tempranas mediante un test específico podría mejorar el seguimiento y el abordaje terapéutico de estos niños. El objetivo de este trabajo es analizar las características neuromadurativas de los andadores de puntillas (AP) de edad preescolar en relación con un grupo control (GC).
MétodoMediante un estudio descriptivo transversal se han comparado los posibles factores de riesgo, las características del neurodesarrollo y los resultados del Cuestionario de Madurez Neuropsicológica Infantil (CUMANIN) de un grupo de 56 AP de entre 3 y 6 años con un GC de 40 niños.
ResultadosEntre los AP había un mayor porcentaje de varones (p=0,008) y este grupo presentaba un porcentaje significativamente mayor de antecedentes familiares (p=0,000) y de factores de riesgo biológico durante el período perinatal (p=0,032). Los padres manifestaron que los AP mostraban una torpeza motriz significativamente superior (59%; p=0,009). Los AP obtuvieron en el CUMANIN puntuaciones significativamente inferiores en las escalas de psicomotricidad (p=0,001) y memoria (p=0,001), y en el desarrollo verbal (p=0,000), no verbal (p=0,026) y total (p=0,004), además de una menor definición de la lateralidad podálica (p=0,047).
ConclusionesLos AP de la muestra estudiada presentan características neuromadurativas específicas que apoyan la consideración de la marcha de puntillas como marcador de problemas del desarrollo, aunque son necesarios más estudios.
Children who display a symmetrical toe-toe gait pattern from the time they start walking are called toe walkers.1–6 These children may, however, achieve initial heel strike when asked to do so or when they concentrate on gait.6–8 Idiopathic toe walking seems to be voluntarily modifiable when there is no significant retraction of the gastrocnemius–soleus complex. Retraction is affected by emotional and cognitive factors.6,7,9,10
Although a diagnosis of idiopathic toe walking is currently considered to rule out neurological and orthopaedic disorders,2,4,6,11 this condition has been associated with alterations in different areas of child development in several studies.3,5,6,9,12,13 Furthermore, idiopathic toe walking has also been associated with different complications occurring during pregnancy, delivery, and the postpartum period2,4,14 and 10% to 88% of cases have a family history of the condition. This suggests central nervous system (CNS) involvement.15,16
Therefore, presence of toe walking points to the need for a thorough assessment of neurodevelopmental disorders.1,2,17–19 The unique features of toe walkers have led researchers to consider idiopathic toe walking as a syndrome, given that it reflects delayed neuromotor acquisition and maturation aggravated by secondary retraction of the triceps surae.17
Following this line of reasoning, several researchers have hypothesised that idiopathic toe walking is a motor expression of minimal cerebral dysfunction (MCD).2,5,6 Some studies including series of toe walkers have detected alterations in motor control, visual perception, spatial perception, and language.2,18,19 Accardo et al.19 evaluated 163 children attending an ambulatory paediatric clinic and found lower language quotients in toe walkers than in children with normal gait patterns, especially in the group of preschool children. Likewise, Shulman et al.2 reported a high frequency of attention deficits, hypo- and hyperactivity, articulation and language organisation errors, deficits in fine and gross motor control, and visuomotor problems in a series of 13 toe walkers. In 2010, Williams et al.15 suggested that toe walking was associated with sensory processing dysfunction, defined as an alteration in “the neurological process that organises sensation from one's own body and from the environment and makes it possible to use the body effectively within the environment”. In a subsequent study by the same authors, including children aged 4-8, toe walkers displayed lower sensory integration coefficients and poorer performance on the Bruininks–Oseretsky Test of Motor Proficiency.20
Given the lack of studies on idiopathic toe walking in Spanish populations, and considering that early assessment of developmental characteristics in toe walkers may optimise toe walking assessment, follow-up, and treatment, we decided to use a test specifically developed in Spain to evaluate the presence of difficulties typically displayed by toe walkers at early ages. The purpose of our study is to analyse the neurodevelopmental characteristics of preschool toe walkers and compare them to a control group.
Patients and methodsOur cross-sectional descriptive study was approved by the clinical research ethics committee of the School of Nursing, Physiotherapy, and Podology at Universidad Complutense de Madrid (register number F[EFP]-001/2010). The study was publicised and its characteristics explained in simple language. We included children aged 3-6 who attended the physiotherapy unit at the podology clinic of Universidad Complutense de Madrid between February and July 2010.
After parents signed informed consent forms, we gathered the medical histories of all children and performed a clinical assessment to select the sample. We then included all children aged between 36 and 83 months who were able to cooperate with the assessment protocol. Exclusion criteria were as follows: neurological lesions; genetic syndromes; psychiatric alterations; developmental disorders; congenital or acquired locomotor system disorders; equinus foot secondary to surgery, trauma, or viral infection; unilateral gait alterations; having received or being under treatment for toe walking at the time of inclusion; and any alterations that may prevent children from understanding visual or verbal instructions or advise against inclusion in the study.
We finally selected 96 of the 151 children evaluated during the study period. In the second stage, participants were classified as either toe walkers or controls. The toe walking group included those children who had previously been diagnosed with toe walking by a paediatrician, traumatologist, or neurologist, and who displayed a preference for toe walking. The control group included those children spontaneously attaining a heel-toe gait. Our sample included 56 toe walkers and 40 controls (Fig. 1). Most of the participants were boys.
We interviewed the participants’ parents or guardians to gather data about age, sex, potential risk factors for toe walking, neurodevelopmental milestones, and most frequent clinical manifestations4,11–14,21:
- –
Family history of idiopathic toe walking (parents, uncles or aunts, siblings, cousins).
- –
Biological risk, presence of neurological and sensory, visual, or auditory risk factors.
- –
Birth weight, characteristics of delivery, and complications.
- –
Age when children achieved bowel and bladder control (daytime and night-time), started crawling and walking, and uttered their first words.
- –
Crawling: we recorded whether the child had crawled at any time during development and gathered data on crawling patterns.
- –
Use of baby walkers or any other devices facilitating walking during the early stages.
- –
Clumsiness, parents’ perception that their child fell frequently or was clumsy or uncoordinated compared to his or her peers; frequency of out-of-school physical exercise.
Neurodevelopment was assessed with the CUMANIN questionnaire (Cuestionario de Madurez Neuropsicológica Infantil), a screening tool validated in Spain which evaluates different areas of a child's neurological maturation considering his or her chronological age, providing comprehensive information of a child's development (cognition, perception, motor function, language, and laterality).22–25 This test has been validated in several countries26–28 and has been proven useful in evaluating development in children with and without developmental alterations.29–35
Children were assessed in a quiet, spacious room equipped with all materials necessary for the assessment. Tests were conducted in strict order, following the instructions for each test and adhering to the criteria for correcting test responses included in the questionnaire itself.22 Each of the tests were presented as a challenge to evaluate the children's abilities and limitations in completing increasingly complex tasks, that is, their voluntary adaptive behaviour.36 We administered the tests corresponding to each of the 8 main scales (psychomotor function; articulatory, expressive, and comprehensive language; spatial structuring; visual perception; memory; and rhythm), as well as 2 supplementary tests (verbal fluency and attention); results were expressed as percentiles. Furthermore, we estimated overall verbal development (based on the scales assessing language articulation, expression, and comprehension) and non-verbal development (scales assessing psychomotor function, spatial structuring, visual perception, memory, and rhythm), as well as overall development (combined raw scores on the 8 main scales). We also recorded the hand or foot used by each child to evaluate visual, manual, and foot laterality, and homogeneity or heterogeneity in hand/foot use.22 After tests were corrected, results were discussed with parents; all parents received a report of their child's test results.
Statistical analysis was conducted using SPSS version 19.0 for Windows. The first analysis addressed the characteristics and regularity of the data set; qualitative variables were expressed as frequencies and quantitative variables as means±SD. Groups were compared using the t test for quantitative variables and the chi-squared test for qualitative variables. Statistical significance was set at P<.05 for 95% confidence intervals.
ResultsMean age in our sample was 54.7±12.165 months in the toe walking group and 56.55±14.229 months in the control group. No statistically significant differences were found between groups (P=.495). Boys accounted for 69.6% of toe walkers and 57.7% of controls; there were significantly more boys among toe walkers (P=.008).
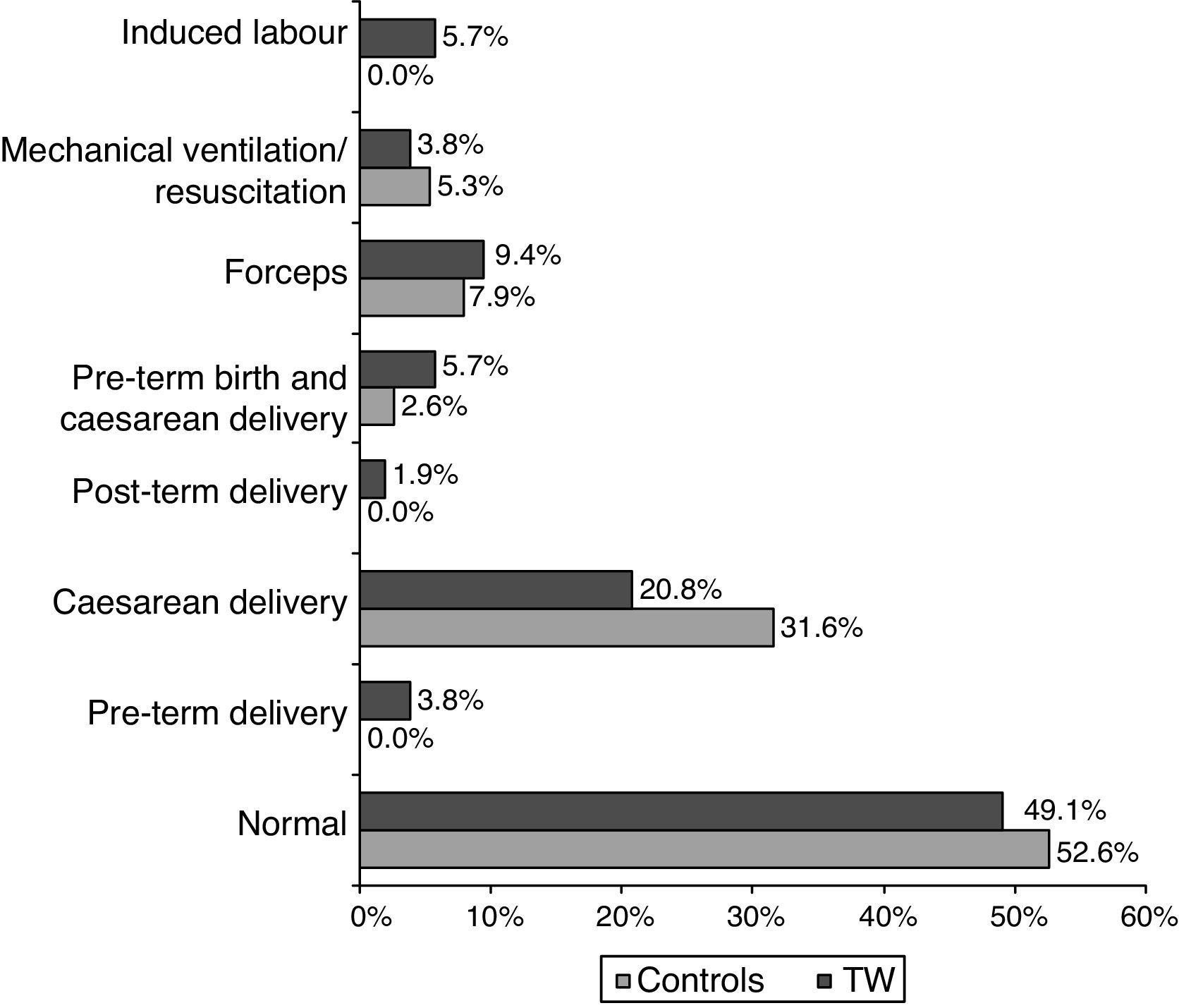
Regarding the initial interview on potential risk factors for toe walking, the only significant intergroup differences were found in family history of toe walking and presence of biological risk factors during the postpartum period (Table 1). We also analysed the circumstances of delivery. Although no statistically significant differences were found between toe walkers and controls, we observed a high frequency of caesarean deliveries and other risk factors associated with delivery in both groups (Fig. 2). Furthermore, we queried parents about the way in which their children started to move around. Although crawling was the most frequent form of locomotion in both groups, dragging and bottom scooting were found in 11.1% and 4.4% of toe walkers, respectively, with no statistically significant differences between groups (P=.432). The percentage of parents or guardians who perceived motor difficulties in their children was significantly higher in the toe-walking group (59%) than in the control group (28%; P=.009). Only 7.1% of the toe walkers did no physical exercise out of school; no significant intergroup differences were found (P=.088).
Family and personal history of toe walking, and other factors potentially associated with toe walking.
| Variable (unit) | Group | Mean | SD | P-value |
|---|---|---|---|---|
| Family history of toe walking (%) | Controls | 0 | .000* | |
| TW | 17 | |||
| Presence of biological risk factor | Controls | 0 | .032* | |
| TW | 11.3 | |||
| Low or very low birth weight | Controls | 13.2 | .057 | |
| TW | 3.8 | |||
| Age of bladder control, daytime (months) | Controls | 28.26 | 5.413 | .431 |
| TW | 27.23 | 6.712 | ||
| Age of bladder control, night-time (months) | Controls | 32.86 | 12.825 | .938 |
| TW | 33.1 | 8.949 | ||
| Age of bowel control (months) | Controls | 27.08 | 9.626 | .793 |
| TW | 27.53 | 6.707 | ||
| Age of crawling onset (months) | Controls | 8.04 | 2.875 | .486 |
| TW | 8.51 | 2.475 | ||
| Age of walking onset (months) | Controls | 12.62 | 2.135 | .262 |
| TW | 13.11 | 2.061 | ||
| Age at first word utterance (months) TW | Controls | 19.54 | 7.377 | .082 |
| 16.88 | 6.707 | |||
| Locomotion by crawling (%) | Controls | 71.8 | .969 | |
| TW | 71.4 | |||
| Walking experience (months) | Controls | 43.82 | 15.398 | .501 |
| TW | 41.80 | 12.541 | ||
| Use of baby walker (%) | Controls | 18.4 | .347 | |
| TW | 26.8 |
TW: toe walkers; SD: standard deviation.
CUMANIN tests demonstrated statistically significant differences between toe walkers and controls in the scales assessing psychomotor function and memory, and in verbal, non-verbal, and overall development, with toe walkers scoring lower than controls (Tables 2 and 3). Regarding visual, manual, and foot laterality, statistically significant intergroup differences were found only in foot laterality (Fig. 3). As for ocular and manual dominance, 59.6% (n=33) of toe walkers displayed right-eye dominance (P=.057) and 92.6% (n=52) showed a preference for the right hand (P=.342), with no significant intergroup differences.
Mean values of the percentiles obtained for each of the CUMANIN subscales.
| Variable | Group | Mean | SD | P-value |
|---|---|---|---|---|
| Psychomotor function | Controls | 71.50 | 15.772 | .001* |
| TW | 29.81 | 24.946 | ||
| Articulation | Controls | 60.00 | 13.578 | .556 |
| TW | 46.11 | 23.243 | ||
| Expression | Controls | 67.35 | 14.756 | .838 |
| TW | 61.21 | 29.619 | ||
| Comprehension | Controls | 65.00 | 13.149 | .822 |
| TW | 59.02 | 26.238 | ||
| Spatial structuring | Controls | 52.78 | 10.062 | .158 |
| TW | 61.88 | 29.313 | ||
| Visual perception | Controls | 70.75 | 16.192 | .193 |
| TW | 55.04 | 26.036 | ||
| Memory | Controls | 91.25 | 8.303 | .001* |
| TW | 68.21 | 23.902 | ||
| Rhythm | Controls | 49.75 | 6.869 | .151 |
| TW | 42.46 | 27.533 | ||
| Verbal fluency | Controls | 97.00 | 20.435 | .475 |
| TW | 78.00 | 26.162 | ||
| Attention | Controls | 52.67 | 18.656 | .644 |
| TW | 37.19 | 20.951 |
TW: toe walkers; SD: standard deviation.
Mean values of percentiles obtained for the different levels of development evaluated with the CUMANIN and development quotient, in absolute values.
| Variable | Group | Mean | SD | P-value |
|---|---|---|---|---|
| Verbal development | Controls | 79.54 | 15.002 | .000* |
| TW | 41.54 | 21.795 | ||
| Non-verbal development | Controls | 81.70 | 16.185 | .026* |
| TW | 51.92 | 28.560 | ||
| Overall development | Controls | 77.44 | 19.575 | .004* |
| TW | 43.13 | 24.692 | ||
| Development quotient | Controls | 124.00 | 21.354 | .085 |
| TW | 97.12 | 11.853 |
TW: toe walkers; SD: standard deviation.
Our study included a large sample of paediatric patients (56 toe walkers and 40 controls, Fig. 1), considering the limited age range of the sample and the prevalence of idiopathic toe walking.15,19,37 The minimum age for participation in our study was set at 3 years: at this age, toe walking may begin to be considered abnormal, leading to recommendations for medical assessment.3,38 We recruited children of up to 6 years of age, since we aimed to detect alterations at early ages which may be associated with poor school performance.25,39 These strict exclusion criteria ensured that the toe walkers included in our study had no developmental alterations that may explain specific neurodevelopmental features. Some reasons for exclusion were pes planovalgus, in-toeing gait, and such neurodevelopmental alterations as intrauterine growth restriction or cognitive alterations.
Interviewing parents or guardians was useful not only for identifying toe walkers and confirming that the included patients did not meet the exclusion criteria, but also for gathering the patients’ medical histories and recording data on associated risk factors, neurodevelopmental milestones, and the most relevant clinical manifestations.2,21,37,40 Interviews were also helpful in providing parents with information about their children.2,4,5,11–14,21
Regarding the characteristics of our sample, both groups had similar mean ages. Although there were significantly more boys in the toe walking group (P=.008), both groups included more boys than girls; the percentage of boys in our study is in line with the literature (50% to 80%).4,9,12–14
The clinical interview gathered data on risk factors for toe walking. One such factor is a family history of toe walking (Table 1), which was present in 17% of toe walkers in our sample (P=.000), a low percentage compared to the rates reported by other authors (30% to 60%).4,11–14,21 The great variability of these rates may reflect the impact of a combination of genetic and environmental factors contributing to this syndrome of the CNS.16 Biological risk factors presenting during the postpartum period, which have been addressed by several authors and may be potential markers of previous CNS damage,2,4 were found in 11.3% of toe walkers and in none of the controls (P=.032). Unlike the data reported by other authors,14 our study found no association between low or very low birth weight and idiopathic toe walking (P=.057). No significant intergroup differences were found in terms of presence of other risk factors frequently associated with toe walking, such as pre-term birth or complications during delivery.41 However, our rates differ from those reported by other studies on toe walking (Fig. 2), with higher rates of caesarean deliveries and lower rates of pre-term births.4,11,14 Furthermore, some children displayed several risk factors, which represents a greater risk of deficiencies.22
Interviews provided data on age of achievement of different neurodevelopmental milestones (sphincter control, crawling, walking, uttering words), which reflect behavioural factors associated with CNS maturation. According to Shulman et al.,2 toe walkers show delays in achieving some of these milestones; in our sample, however, toe walkers and controls achieved these milestones at similar ages (Table 2) and within the age range described in the literature.4,11,14
Baby walkers are not recommended since they have been linked to accidents and alterations in musculoskeletal and neurological development.42 Furthermore, they have been associated with persistent toe walking since they promote a gait pattern similar to that of toe walkers.4,14 In our study, no significant intergroup differences were found in the use of baby walkers (P=.347). Some 26.8% of toe walkers used baby walkers; this percentage is lower than those reported for other paediatric series of toe walkers (over 50%).4,42
Parent-reported signs of poor motor coordination (for example, frequent falls and poorer motor skills than their peers11,14,21) were found in 59% of toe walkers; this percentage was significantly higher than that of controls and higher than the rate reported by other researchers (45% with poor balance).4 Despite having poorer motor skills, which may have led to rejection of physical exercise, toe walkers in our sample did not exercise less frequently than controls (P=.088).21
Regarding neurodevelopmental features, our controls obtained scores within the normal limits for the Spanish population; their scores were similar to those of controls in other studies.23–25 The percentiles of toe walkers were significantly lower than those of controls in the scales assessing psychomotor function and memory, as well as in verbal, non-verbal, and overall development (Tables 2 and 3). These patients showed specific manifestations similar to those reported in other studies.2,19 Toe walkers also scored moderately low in the scale assessing attention and displayed slightly lower-than-average percentiles in articulatory language and rhythm, and below the 60th percentile in language comprehension and visual perception. The deficits displayed by toe walkers in our sample may explain their poorer motor skills and affect crucial areas involved in learning and gait development.29,43–45 These deficits therefore support the hypothesis that toe walking is a manifestation of alterations in the neurocognition processes underlying motor perception and organisation, as well as a marker of developmental problems.1,15
Similar difficulties have been found in children with hyperactivity and MCD (deficits are more severe and tend to affect multiple domains in these children),22 with extremely low birth weight,24 from a poor socioeconomic environment,34 and with school problems.31 The neuropsychological features displayed by these children and toe walkers from our sample result in a heterogeneous profile. In these patients, performance is satisfactory in some areas, which may compensate for their deficits; however, these patients may also benefit from interventions specifically designed for these deficits.22,24,25 This neuropsychological profile points to neurodevelopmental delays and underlines the need for close follow-up and neuropsychological training when difficulties persist or grow. Nevertheless, given that CUMANIN is a screening test, the probable presence of developmental alterations and the need for treatment must be assessed by specialists using specific tools.22
Regarding laterality, which reflects cerebral organisation,23,46 the percentages of children displaying right-hand and right-eye dominance were similar in both groups and fell within the range reported by other authors.23,24 However, a significantly lower number of toe walkers displayed right-foot preference compared to controls (Fig. 3) and age-matched healthy children.23 Although laterality is not completely established at such early ages as those of the children in our sample,46 the fact that the percentage of right-foot preference was lower than that of right-hand preference in toe walkers may indicate delays in functional control inherent to CNS cephalocaudal development in these patients,46 leading to an immature gait pattern. We also studied homogeneity of visual, manual, and foot laterality given that children with normal neuropsychological development have been found to display homogeneous laterality, whereas those with MCD usually show cross laterality or ambidexterity, which involves a greater risk of developing language disorders, learning problems, etc.22 In our sample, 59.6% of toe walkers displayed homogeneous laterality vs 67.5% of controls; differences were not statistically significant (P=.068).
ConclusionsToe walkers in our sample frequently show a family history of toe walking and biological risk factors during the postpartum period. They also scored significantly lower on the neurodevelopmental assessment scales evaluating psychomotor function and memory, and verbal, non-verbal, and overall development. Likewise, they displayed foot laterality less frequently. Our results show that toe walkers have specific neurodevelopmental features, supporting the hypothesis that toe walking is a marker of developmental alterations. Longitudinal studies with larger sample sizes and a multidisciplinary approach are necessary to assess the progression of neuropsychological alterations and establish criteria for treatment indication in toe walkers. Furthermore, the connection between the multiple alterations displayed by toe walkers should be analysed to optimise clinical management in these patients.
FundingThe authors have no financial or personal relationships with other persons or organisations which could create conflicts of interest with regard to this article. This study has received no external funding of any kind.
Conflicts of interestThe authors have no conflicts of interest to declare.
The podology clinic of Universidad Complutense de Madrid provided personnel and material support. Santiago Cano contributed to the statistical analysis. This study would not have been possible without the participation of our patients and their parents.
Please cite this article as: Martín-Casas P, Ballestero-Pérez R, Meneses-Monroy A, Beneit-Montesinos JV, Atín-Arratibel MA, Portellano-Pérez JA. Desarrollo neuromadurativo en andadores de puntillas de edad preescolar. Neurología. 2017;32:446–454.