The SARS-CoV-2 virus first appeared in China in December 2019, and reached Spain several weeks later. While infection mainly causes respiratory symptoms, cases have been reported since the beginning of the pandemic of symptoms suggesting neurological involvement: headache, anosmia, myalgia, asthenia, insomnia, confusion, bradyphrenia, etc. These symptoms have been reported both in mild cases and in more severe cases of COVID-19 requiring orotracheal intubation and intensive care unit (ICU) admission.
A widely accepted hypothesis is that the virus uses the angiotensin-converting enzyme 2 (ACE2) receptor to enter the cell. As the receptor is expressed in glial cells and neurons in physiological conditions, it is logical that the virus could cause neurological symptoms over the course of the infection.1,2 Reports of neurological symptoms associated with the infection are increasingly frequent, and include cases of Guillain-Barré syndrome, stroke, intraparenchymal haemorrhage, and cerebral thrombosis.3–13
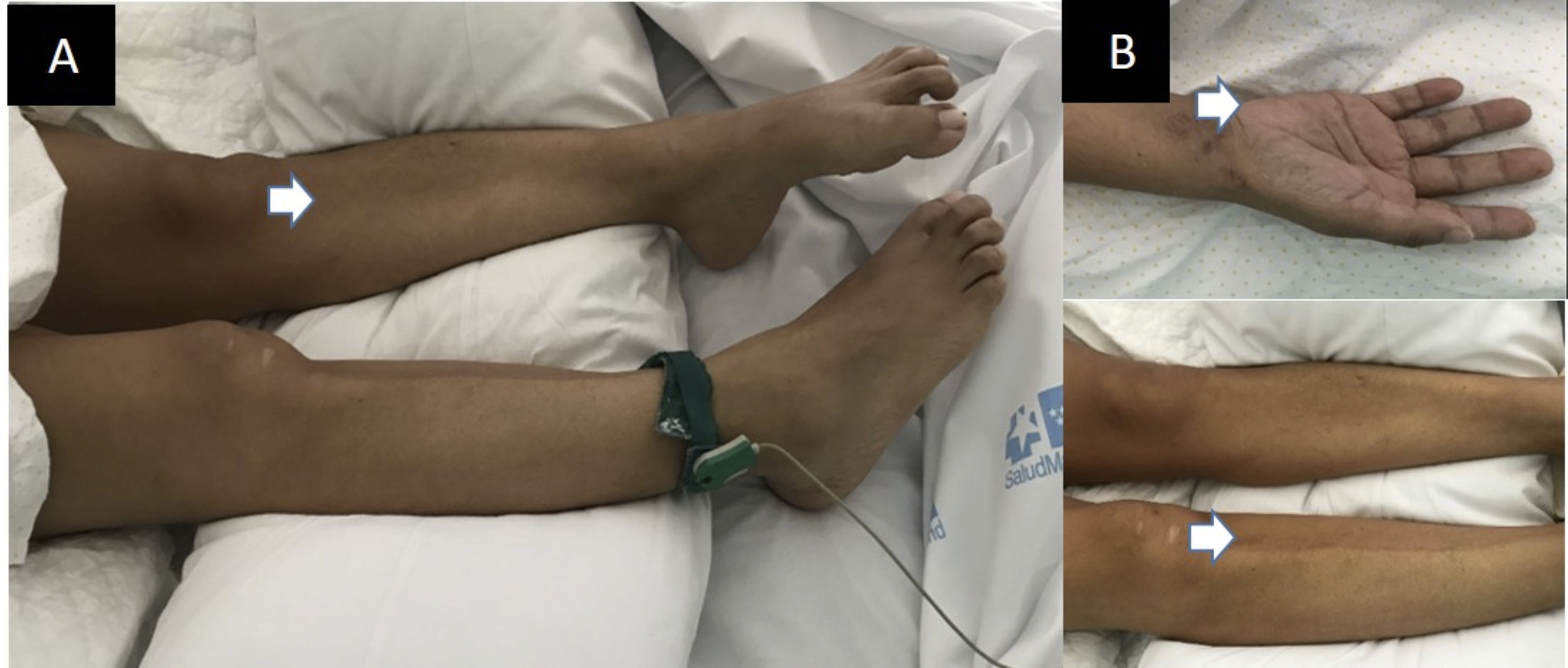
We present the case of a 45-year-old woman with no relevant history who was admitted to the ICU due to severe respiratory insufficiency secondary to bilateral pneumonia, with positive nasal swab PCR results for SARS-CoV-2; therefore, the patient met the World Health Organization criteria for COVID-19. She stayed in the ICU for 2 weeks, and was treated with hydroxychloroquine, azithromycin, tocilizumab, and ceftriaxone, and a 5-day cycle of high-dose intravenous corticosteroids (250 mg/day). Upon discharge from the ICU, she presented generalised weakness and myalgia. Physical examination revealed marked global muscle atrophy, which was more severe in the lower limbs, especially the tibialis muscles, and in the intrinsic muscles of the hands; no alterations were observed in tactile or pain sensitivity or in proprioception (Fig. 1). Muscle strength was 2/5 in the lower limbs and 3+/5 in the upper limbs. Patellar and biceps reflexes were present. She presented no cognitive alterations or cranial nerve involvement. A brain MRI scan and EEG study performed within 48 hours of admission yielded normal results.
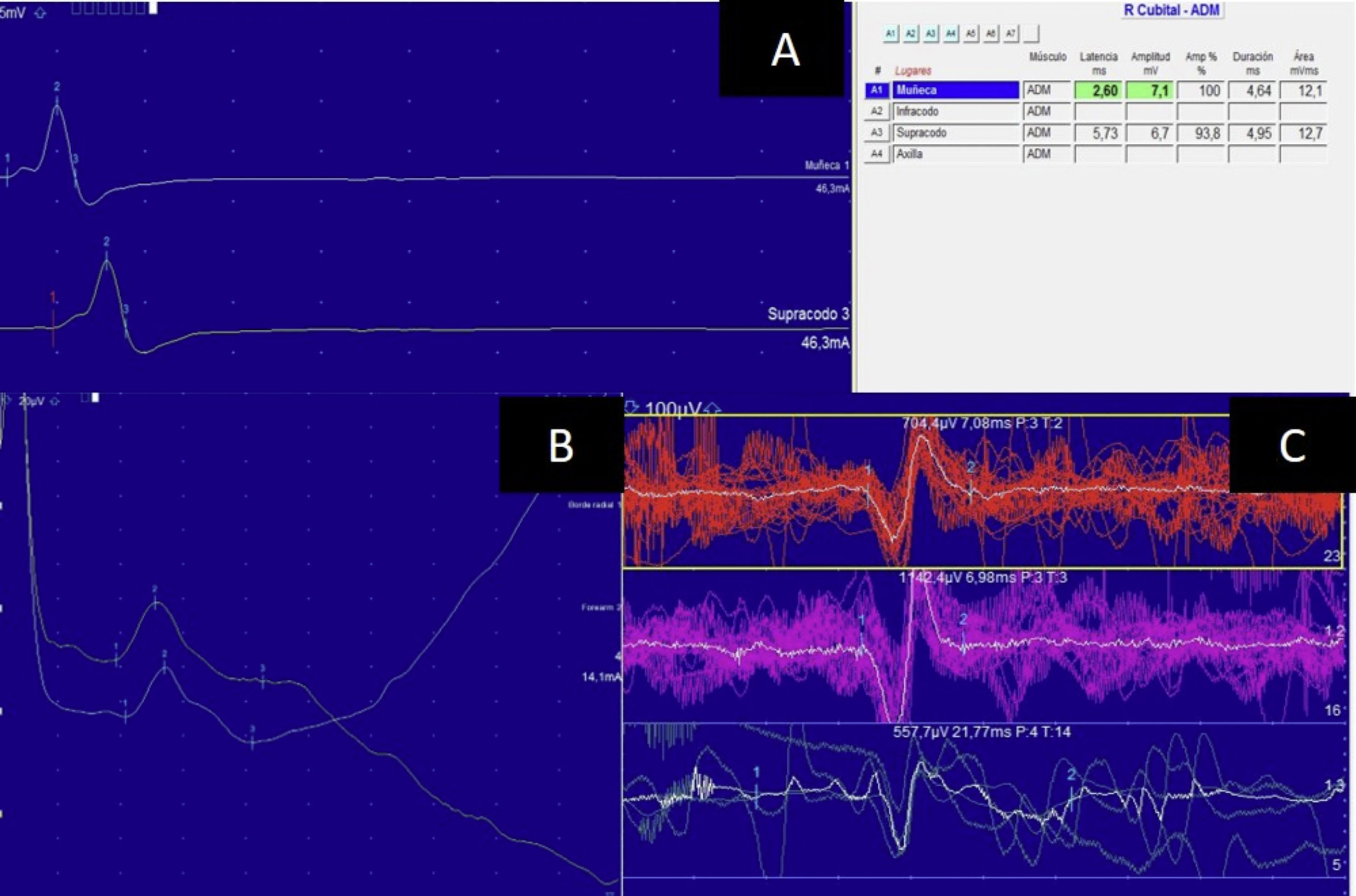
An electroneuromyography study showed completely normal motor conduction velocity in the median, ulnar, sural, and tibial nerves. F-wave duration was 48 ms in the tibial nerve. The muscle study showed positive sharp waves at rest, reduced motor unit potential amplitude and duration, and no polyphasia, especially in the abductor digiti minimi and the tibialis anterior (Fig. 2). Creatine phosphokinase levels ranged from 642 to 900 U/L.
Electroneuromyography study showing normal conduction velocity and amplitude in the left ulnar nerve (A). Sensory amplitude in the superficial radial nerve was normal (B). Muscle study of the right tibialis anterior revealed small motor unit potential amplitudes, without polyphasic motor unit potentials (C).
We initially suspected Guillain-Barré syndrome or critical illness polyneuropathy; however, given the presence of patellar and biceps reflexes and the short ICU stay, we opted to perform further testing. While creatine phosphokinase levels were not especially elevated, muscle atrophy was very pronounced, given the short duration of immobilisation. Hydroxychloroquine and corticosteroids can cause symptoms of myopathy, but we consider this hypothesis unlikely as the patient only received these drugs for one week. Our final diagnosis was acute inflammatory myopathy secondary to SARS-CoV-2 infection. Progression at 12 weeks was very favourable, and the patient was practically asymptomatic at the end of this period.
Myalgia and asthenia are very common in patients with COVID-19, and appear to be caused by the systemic inflammatory reaction to the virus; these cases of EMG-confirmed myopathy may also be explained by inflammatory causes.
This study was approved by our centre’s research committee.
Please cite this article as: Abenza Abildúa MJ, Ramírez Prieto MT, Moreno Zabaleta R, Algarra Lucas C, Pérez López C. Miopatía asociada a infección grave por SARS-CoV-2. Neurología. 2020;35:706–708.