The term “psychosis” comes from the Greek prefix “ψυχo-” (“soul” or “mind”) and suffix “σιζ-” (“abnormal condition”).1 It is used to describe an altered mental state characterised by loss of the ability to differentiate between inner mental experiences and the external reality.2 Epileptic patients frequently present psychiatric comorbidities, including psychotic disorders.3–6 Levetiracetam-induced psychosis affects 1%-1.3% of all patients treated with this antiepileptic drug (AED), and represents a diagnostic and therapeutic challenge.3,7,8 Several risk factors for AED-induced psychotic disorders have recently been identified, and include female sex, temporal lobe involvement, and use of levetiracetam.3 We present a series of 3 patients who developed levetiracetam-induced psychosis and who met the aforementioned criteria.
Our patients were 3 women with acquired brain damage secondary to cerebrovascular disease and presenting no history of psychiatric disorders. Mean age was 49 years (range, 32-63), and all 3 had recently been diagnosed with focal seizures (secondarily generalised) with structural causes9–11 in the context of vascular epilepsy.12 All 3 patients presented delusional ideation and disorganised thinking and speech within 10-14 days after starting treatment with levetiracetam 500-1000mg/12hours; 2 also reported visual hallucinations (patients 2 and 3).
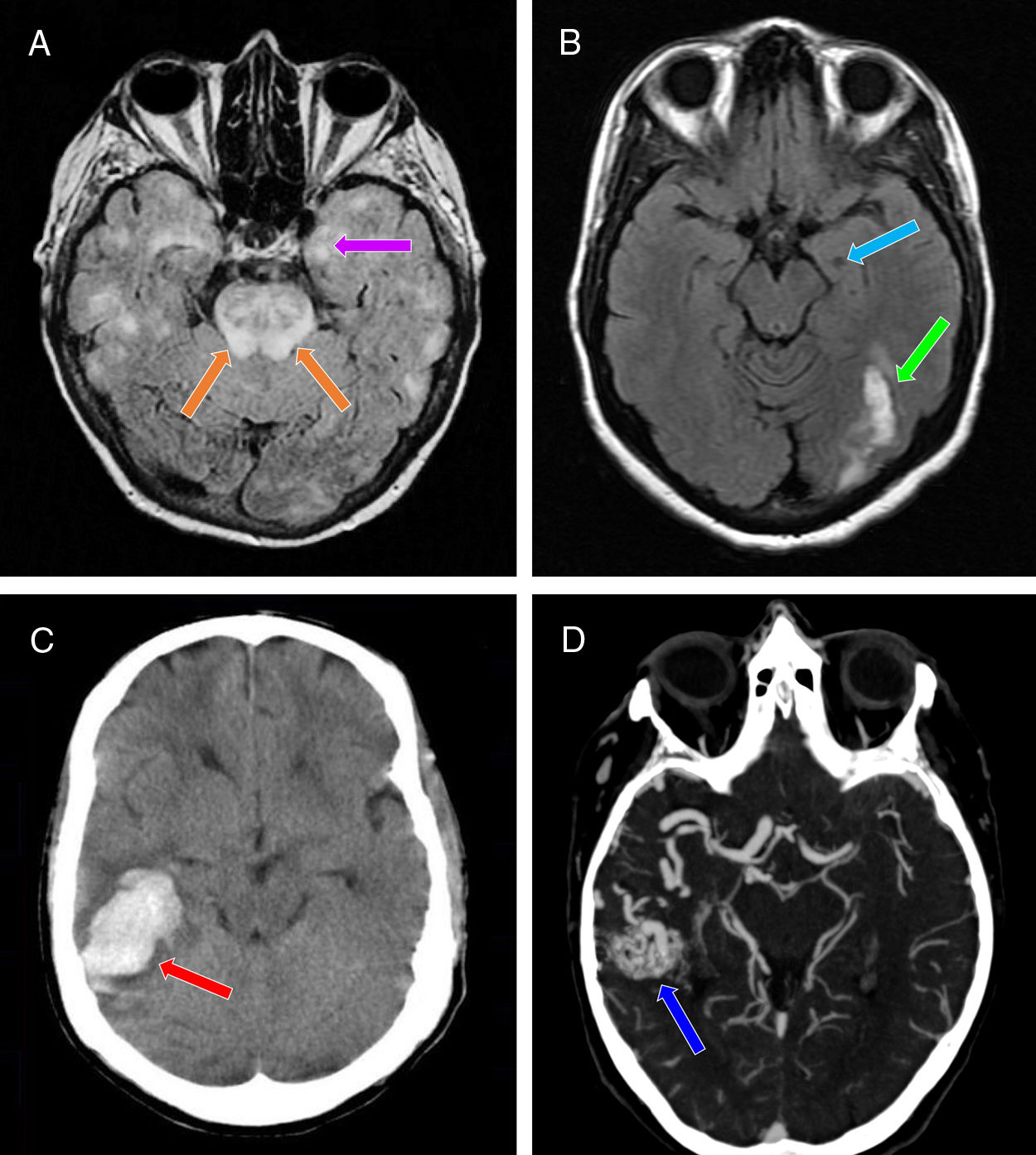
Patient 1Our first patient was a 63-year-old woman diagnosed with postanoxic encephalopathy caused by a haemodynamic mechanism in the context of cardiogenic shock secondary to acute myocardial infarction (Killip IV), with bilateral temporal lobe ischaemia (Fig. 1A). After a month, she developed non-motor, focal seizures associated with impaired consciousness, behavioural alterations, and progression to bilateral tonic-clonic seizures.13 She was prescribed levetiracetam, with doses increasing every 2 weeks up to 1000mg/12hours (initially with intravenous and subsequently with oral administration). Twelve days later she presented aggressiveness and persecutory delusions.
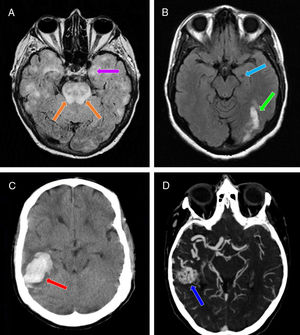
Neuroimaging studies performed in our 3 patients. A) Brain MRI scan (axial plane, FLAIR sequence) showing multiple patchy areas of corticosubcortical hyperintensity bilaterally, including the hippocampus (particularly the left; purple arrow) and the pons (orange arrows). B) Brain MRI scan (axial plane, non-contrast T1-weighted sequence) showing a hypointense region in the left hippocampus (suggestive of chronic progression; light blue arrow) and signal hyperintensity in the ipsilateral occipital region (suggestive of subacute lesion; green arrow). C) Non-contrast head CT scan (axial plane) revealing a hyperdense area in the right temporal lobe, with no intraventricular extension, and surrounding vasogenic oedema (red arrow). D) CT angiography of the supra-aortic trunks and circle of Willis confirming the presence of an arteriovenous malformation in the right temporal region (dark blue arrow), mainly supplied by the right middle cerebral artery, as well as by the right posterior cerebral artery through the lateral posterior choroidal arteries, and with venous drainage through the right transverse and sigmoid sinuses.
The second patient was a 52-year-old woman diagnosed with intraparenchymal haemorrhagic stroke in the left occipital lobe, and left temporal ischaemia due to vasospasm (Fig. 1B) secondary to holocranial vasculopathy, which presented after radiation therapy for the treatment of a fourth ventricle ependymoma 30 years previously. She presented another stroke episode, characterised by holocranial headache and right homonymous hemianopsia, and a week later presented a motor-onset epileptic seizure associated with impaired consciousness, clonic movements in the right side of the face, and bilateral progression to tonic-clonic seizures.13 The patient received levetiracetam, with doses increasing every 2 weeks up to 500mg/12hours (initially intravenous and subsequently oral administration). Ten days later, the patient presented persecutory delusions and visual hallucinations.
Patient 3Our third patient was a 32-year-old woman with right temporal intraparenchymal haemorrhagic stroke (Fig. 1C) associated with bleeding from an arteriovenous malformation (Fig. 1D). Five days later, she presented 2 focal motor-onset seizures, which initially were not associated with altered level of consciousness and were accompanied by left faciobrachial clonic movements; the second seizure progressed to a bilateral tonic-clonic seizure, with subsequent loss of consciousness.13 After 14 days of treatment with levetiracetam, with doses increasing every 2 weeks up to 500mg/12hours (initially intravenous and subsequently oral administration), the patient presented messianic delusions and visual hallucinations.
According to a thorough neuropsychiatric examination, none of the patients met criteria for postictal psychosis (which usually appears within a week of an epileptic seizure)14 or confusional syndrome (while all 3 patients presented impaired consciousness, level of consciousness did not fluctuate over the course of a day, and no sleep-wake cycle alterations or cognitive impairment [eg, disorientation]15 were observed), and the patients did not show forced normalisation (hypothetical psychosis induced by clinical control of epileptic seizures and the dramatic improvement in epileptiform activity in the temporal lobe after onset of antiepileptic treatment with levetiracetam, which would not have resolved after switching from levetiracetam to eslicarbazepine acetate).14,16 A blood analysis ruled out toxic, metabolic, and infectious causes, and plasma levetiracetam levels were within the therapeutic range (12-46mg/L).17
Due to suspicion of a causal association between psychotic symptoms and onset of treatment with levetiracetam, the drug was progressively withdrawn and oral eslicarbazepine was introduced, with doses increasing up to 800mg/24hours (the medication was taken with dinner in all 3 cases). Symptoms resolved completely and the patients presented no further seizures, with no need for antipsychotic treatment; we established a diagnosis of levetiracetam-induced psychosis (DSM-5 code: 292.9, F19.259).18 According to the scale proposed by Naranjo et al.,19 our patients present a probable association between symptoms and treatment with levetiracetam (score, 8/13). All 3 patients presented good tolerance to eslicarbazepine and remained seizure-free during follow-up (39 months for patient 1, 29 months for patient 2, and 36 months for patient 3).
DiscussionChen et al.3 conducted a case-control study including 2630 patients with epilepsy and found that AED-induced psychotic disorders were positively correlated with 3 risk factors: female sex, temporal lobe involvement, and treatment with levetiracetam. Treatment with carbamazepine, on the contrary, was found to be negatively correlated with presence of AED-induced psychotic disorders. More recently, Josephson et al.20 conducted a retrospective cohort study including 1173 patients with epilepsy who were receiving levetiracetam, identifying 4 risk factors for psychiatric adverse reactions to levetiracetam: female sex, depression, anxiety, and recreational drug use. This study also found greater risk of AED-induced psychiatric adverse reactions in patients with more risk factors: 8% for patients with no risk factors, 11%-17% for patients with one risk factor, 17%-31% for 2, 30%-31% for 2, 30%–" is deleted.--!>42% for 3, and 49% for 4 risk factors. They also observed that, for each stratum, depression and/or drug addiction were consistently associated with higher risk.
The exact aetiopathogenic mechanism of levetiracetam-induced psychosis is unknown; however, loss of hippocampal neurons and disruptive excitatory synaptic reorganisation in the mesolimbic pathway are thought to play a role.3,4
We opted for eslicarbazepine in all 3 cases, for several reasons: the drug has a similar action mechanism to that of carbamazepine (it is a sodium-channel blocker, but it has the particular characteristic that it stabilises the inactive state of voltage-dependent sodium channels)21; it is approved for use in monotherapy in patients with focal seizures, with or without secondary generalisation (in adults with a recent diagnosis of epilepsy)22; its posology was more convenient (a single dose taken at night); it does not interfere with neurorehabilitation; its effectiveness is not inferior to that of carbamazepine23; and it is associated with a lower risk of hyponatraemia than carbamazepine and oxcarbazepine.21,22
Our patients all presented the 3 risk factors described by Chen et al.3 and one of those established by Josephson et al.20 (female sex), and progressed favourably after switching from levetiracetam to eslicarbazepine. Close clinical monitoring is necessary for early detection of levetiracetam-induced psychosis in patients presenting these risk factors, particularly if they have history of psychosis: levetiracetam-induced psychosis is potentially reversible with discontinuation of the drug, enabling us to avoid the use of neuroleptics that may lower the seizure threshold and trigger and/or exacerbate seizures. Eslicarbazepine should be considered in these patients, particularly in those showing difficulties adhering to treatment with several daily doses.
Finally, to answer the question posed in the title, we may establish a profile of patients with epilepsy who are more likely to present levetiracetam-induced psychosis both from a biological and a neurostructural viewpoint (female sex and temporal lobe lesions, respectively). The use of levetiracetam (the exact dose has not been established) in these patients may trigger the disorder through a mechanism that is yet to be understood. However, further well-designed randomised, controlled, prospective studies with larger patient samples are needed to confirm the hypothesis of a causal association.
Our study is not without limitations. Firstly, this case series includes only 3 patients. And secondly, our patients had a clinical diagnosis of epilepsy: diagnosis of epilepsy is mainly clinical, and requires a detailed semiological description and/or video recordings of the episodes by a witness; this should be reflected in the clinical history.11,24–26 None of the patients underwent an EEG study, given the favourable progression of psychotic symptoms after switching to eslicarbazepine. However, EEG and/or video-EEG results may have provided useful complementary data to confirm diagnosis and rule out forced normalisation.14,16
FundingThis study received no public or private funding.
Please cite this article as: León Ruiz M, Rodríguez Sarasa ML, Sanjuán Rodríguez L, Benito-León J, Álvarez de Toledo O, Pérez Nieves MT, et al. Trastorno psicótico de novo inducido por levetiracetam: ¿existe un perfil de paciente epiléptico neuroestructural y/o biológicamente más vulnerable a desarrollarlo? Neurología. 2020;35:684–687.
This study was presented in poster format at the 70th Annual Meeting of the Spanish Society of Neurology (Seville, 20 November 2018) and as an e-poster at the 5th Congress of the European Academy of Neurology (Oslo, 30 June 2019).