Hemifacial spasm is a movement disorder characterised by tonic or clonic contractions of the muscles on one side of the face, which may manifest in short episodes or persistently.1 Vestibular paroxysmia manifests with recurrent episodes of sensation of movement and instability of less than one minute's duration; these generally occur several times per day: episodes may or may not be triggered by posture.2,3 Both entities are included in the group of neurovascular compression syndromes, caused by vascular compression of the entry/exit site of the cranial nerves, close to the brainstem. Neurovascular compression syndromes include superior oblique myokymia, trigeminal neuralgia, hemifacial spasm, vestibular paroxysmia, and glossopharyngeal neuralgia, which are caused by compression of the fourth, fifth, seventh, eighth, and ninth cranial nerves, respectively.1,2,4–6 We describe the case of a patient who simultaneously developed hemifacial spasm and vestibular paroxysmia.
Our patient is a 37-year-old Colombian woman with migraine with aura as the only relevant history. The patient visited our hospital due to a 3-month history of involuntary movements of the right side of the face and paroxysmal episodes of vertigo. Involuntary movements of the face had persisted since onset. Muscle contractions caused narrowing or closure of the right palpebral fissure and, on occasions, a feeling of tightness of the same side of the face with facial droop. She simultaneously started to develop episodes characterised by a sensation of movement lasting a few seconds and not triggered by posture; episodes presented 5-10 times daily. The patient experienced instability and tinnitus between episodes.
The examination revealed tonic contraction of the right orbicularis oculi and lower facial muscles, with occasional clonic movements (Fig. 1). Furthermore, we detected signs of right vestibular hypofunction, with corrective saccades to the right in the head impulse test, and tendency to the right in the pointing test and Romberg test. No other significant findings were observed in the neurological and systemic examinations.
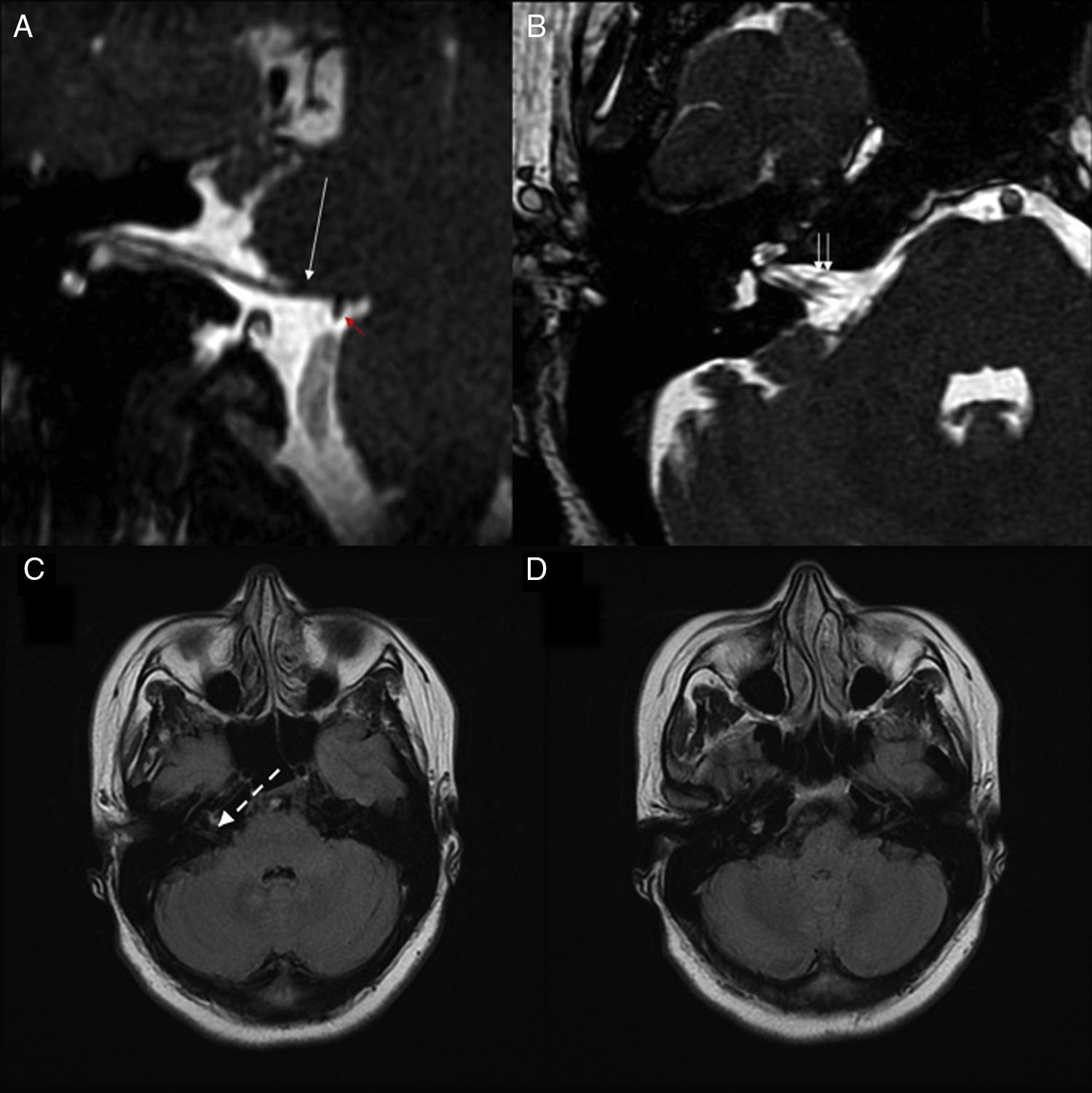
A brain magnetic resonance imaging (MRI) scan was performed with a General Electrics HDxt 1.5 Tesla MRI scanner equipped with an 8-channel high-resolution head coil. T2 weighted images of the internal auditory canals (AIC) were taken using high-resolution 3D fast imaging employing steady-state acquisition (FIESTA) sequences, with a series of 152 0.8-mm-thick slices with 50% overlap, field of view (FOV)=170mm, matrix 320×256, TR=7.1ms, TE=3.0ms. The images obtained revealed an elongation of the right posterior inferior cerebellar artery (PICA), which was in contact with the seventh cranial nerve at the cerebellopontine angle. A vascular loop of the anterior inferior cerebellar artery (AICA) was observed in the entry zone of the right AIC, which was in contact with both the seventh and eighth cranial nerves (Fig. 2). No MRI angiography sequences were obtained.
Reconstruction of the oblique sagittal (A) and axial (B) planes of a high resolution 3D T2-weighted FIESTA sequence of the right cerebellopontine angle. We identified a point of contact with the PICA (red arrow) at the site of emergence of the right seventh cranial nerve (long white arrow). The scan through the right internal auditory canal shows an elongated vessel (double white arrow), corresponding to the AICA, in contact with the right seventh and eighth cranial nerves. (C) and (D) show 2 axial FLAIR sequences of the cerebellopontine angle. The dotted white arrow points to the site of emergence of the right seventh and eighth cranial nerves.
AICA: anterior inferior cerebellar artery; FIESTA: fast imaging employing steady-state acquisition; FLAIR: fluid-attenuated inversion recovery; PICA: posterior inferior cerebellar artery.
To complete the study, we performed an electromyography (EMG) of the right orbicularis oris muscle, which revealed baseline tonic activity with clusters of increased muscle fibre recruitment coinciding with clonic spasms. We also requested hearing tests and auditory evoked potentials, which revealed no significant alterations.
The patient was treated with gabapentin, with the dosage gradually increased to 900mg/day, and displayed partial improvement of vertigo episodes (>50% reduction in number of episodes per day). Subsequently, due to poor tolerance to gabapentin, it was replaced with eslicarbazepine at 800mg/day, achieving excellent symptom control (resolution of vertigo episodes and improved vestibular function). Hemifacial spasm was treated with infiltrations of 7.5U of botulinum toxin type A at 3 points of the right orbicularis oculi muscle, at 3-month intervals, achieving optimal control of the involuntary facial movements.
As described previously, our patient simultaneously presented 2 different clinical entities with the same aetiology: the neurovascular contact involving structures in the posterior fossa. Some authors have proposed that vascular contacts involving the entry/exit zone of the cranial nerves may lead to ephaptic transmission of nerve impulses between adjacent nerve fibres, as a consequence of the demyelination caused by the pulsatile compression of the nerve.7 In our case, the patient presented 2 contacts affecting the seventh and eighth cranial nerves, both due to aberrantly elongated vessels (the right AICA at the ACI and the right PICA at the site of emergence of the facial nerve).
Intercurrent vestibular alterations have been described in some patients with hemifacial spasm, as have vestibular symptoms secondary to decompression of the seventh cranial nerve.8 Furthermore, we found an isolated case series including patients with both vestibular paroxysmia and hemifacial spasm.9 Cases have been reported of patients with hemifacial spasm coinciding with such other neurovascular compression syndromes as trigeminal neuralgia, or even with both trigeminal neuralgia10,11 and glossopharyngeal neuralgia.12 Given the anatomical proximity of the paths of the seventh and eighth cranial nerves through the posterior fossa, it seems logical to consider that coexistence of hemifacial spasm and vestibular paroxysmia should not be exceptional.
Hemifacial spasm presents very specific symptoms and diagnosis is relatively simple. However, vestibular paroxysmia may go unnoticed, since its diagnosis depends on high clinical suspicion, detailed history and exploration, and response to pharmacological treatment with neuromodulators. Given the low prevalence of this syndrome, no randomised controlled clinical trials have been performed; therefore, recommendations regarding treatment are based on individual case reports or case series, and consequently present a low level of scientific evidence. However, response to carbamazepine or oxcarbazepine, even at low doses, is one of the proposed criteria for the diagnosis of vestibular paroxysmia.10,13,14 Furthermore, some cases with response to gabapentin have been published.15 Although there is no evidence on the effect of eslicarbazepine in vestibular paroxysmia, we may expect a similar therapeutic response as with carbamazepine, given its similar action mechanism; this drug also presents a better safety profile, simpler dosing, and higher tolerability.16 Furthermore, although surgical vascular decompression is a therapeutic option for hemifacial spasm, limited evidence is available on whether this technique is indicated for treating vestibular paroxysmia.17,18
Please cite this article as: Silva-Hernández L, Silva-Hernández M, Gutiérrez-Viedma A, Yus M, Cuadrado ML. Espasmo hemifacial y paroxismia vestibular. Concurrencia de 2 síndromes neurovasculares compresivos en una misma paciente. Neurología. 2019;34:131–133.