Glioblastoma (GB) is the most common primary malignant tumour of the central nervous system.1 GB generally involves the subcortical white matter; infratentorial presentation is rare, with less than 1% of tumours involving the cerebellum.2 We present the diagnostic challenge of a case of multifocal GB with striking cerebellar involvement, and the patient’s progression after treatment.
The patient was a 49-year-old woman who consulted in late 2014 due to instability and frequent vomiting of 2 years’ progression, associated with pronounced weight loss. Physical examination detected ataxic gait and no other alterations. A blood analysis including hormones, vitamins, antibodies, and serology testing yielded normal results. A brain MRI scan (1.5 T; Fig. 1a and b) showed tumefactive appearance of the cerebellum, with diffuse involvement of the vermis, and several T2-hypointense lesions on both cerebellar hemispheres, the periaqueductal region of the midbrain, and the caudate nuclei. Contrast administration revealed linear enhancement in the cerebellar lesions (Fig. 1c). These findings were initially interpreted as being suggestive of an inflammatory or granulomatous process. CSF analysis returned normal results, with the exception of elevated levels of angiotensin-converting enzyme (94 U/L; normal range, 8-53). A body CT scan, 18F-FDG PET/CT, and 67Ga-SPECT/CT all yielded normal findings. Suspecting neurosarcoidosis, we started empirical treatment with prednisone and methotrexate; no clinical changes were observed at 6 months. However, a follow-up MRI study (3 T; Fig. 1d) showed a signal alteration in the left mesial temporal lobe, in addition to the known lesions, which had remained stable. Stereotactic biopsy of a tissue sample from the left anterior temporal lobe identified no alterations. In the early months, no clinical or radiological changes were observed, although the larger lesion in the left cerebellar hemisphere presented progression at 12 months (Fig. 1e). A spectroscopy study (Fig. 1f) revealed increased creatine and myo-inositol peak, with a normal choline/N-acetyl aspartate (Cho/NAA) ratio, except in the left cerebellar lesion (which presented a Cho/NAA ratio > 2). An anatomical pathology study of a second biopsy sample from the left hippocampus revealed infiltration due to a high-grade neoplastic proliferation, comprising cells with irregular, hyperchromatic nuclei and small foci of necrosis. Findings from a histology study, immunohistochemical analysis, and fluorescence in situ hybridisation (FISH) were compatible with a diagnosis of GB (Fig. 1g and i)
MRI findings: signal alterations in the cerebellum, midbrain, and caudate nuclei (a and b: 1.5 T, FLAIR sequence). Linear contrast enhancement of the cerebellar lesions (c: gadolinium-enhanced T1-weighted sequence). At 6 months, signal alteration was observed in the left mesial temporal lobe (d: 3 T, FLAIR sequence). At 12 months, the larger lesion, in the left cerebellum, presented pseudonodular contrast uptake (e: gadolinium-enhanced T1-weighted sequence), with a Cho/NAA ratio > 2 on the spectroscopy study (f). Six weeks after onset of radiotherapy and TMZ treatment, the cerebellar signal alteration had reduced (j: FLAIR). A histology study showed infiltration of astrocytes, forming sleeves around blood vessels (g: hippocampus section; haematoxylin and eosin stain; magnification ×400). Immunohistochemistry showed a high proliferative index and perivascular localisation of tumour cells (asterisk) (h: MIB-1 [Ki-67] staining; magnification ×200), and reactive astrocytes (arrows) distributed uniformly in contraposition to the dense perivascular aggregation of tumour cells, with variable expression of glial fibrillary acidic protein (asterisks) (i: GFAP staining; magnification ×200).
The patient was treated with radiotherapy (60 Gy) and temozolomide (TMZ), and presented radiological improvement 6 weeks after treatment onset (Fig. 1j). We continued treatment with TMZ, administering 6 cycles at 160 mg/m2/4 weeks; due to disease progression, the drug was replaced with irinotecan (CPT-11; 150 mg/m2) and bevacizumab (BVZ, 10 mg/kg) every 4 weeks. The patient died 14 months after onset of antitumour therapy.
Multicentric glioma is rare, and most commonly involves supratentorial regions.3 In our patient, the presence of both supra- and infratentorial tumours, with most pronounced involvement in the cerebellum, and the uncertain clinical course, led to uncertainties about the cause. This case also shows that higher-resolution MRI studies can reveal less visible lesions; spectroscopy studies can also provide key information for guiding aetiological diagnosis towards neoplastic processes.4
The standard treatment for GB is based on surgery, where possible, as well as radiotherapy and chemotherapy with TMZ.5,6 CPT-11, a topoisomerase I inhibitor, and BVZ, a monoclonal antibody that inhibits vascular endothelial growth factor (VEGF), are used for second-line treatment, given the high level of vascularisation of these tumours.7 Multifocal presentation and cerebellar localisation have been described as independent risk factors for poor prognosis.8,9 In this case, the survival time after diagnosis was similar to the overall mean for patients with GB, approximately 12 months.10
In conclusion, multifocal presentation of GB constitutes a diagnostic challenge, and must be differentiated from such other processes as inflammatory or granulomatous lesions. Spectroscopy can be a valuable tool in guiding diagnosis. Finally, this entity must be considered in patients presenting cerebellar lesions, despite its low prevalence.
FundingThe authors have received no funding for this study.
Please cite this article as: Rouco Axpe I, Mateos Goñi B, Zaldumbide Dueñas L, Fernández-Lomana Idiondo E. Dificultad en el diagnóstico de un glioblastoma de presentación inhabitual. Neurología. 2021;36:241–243.




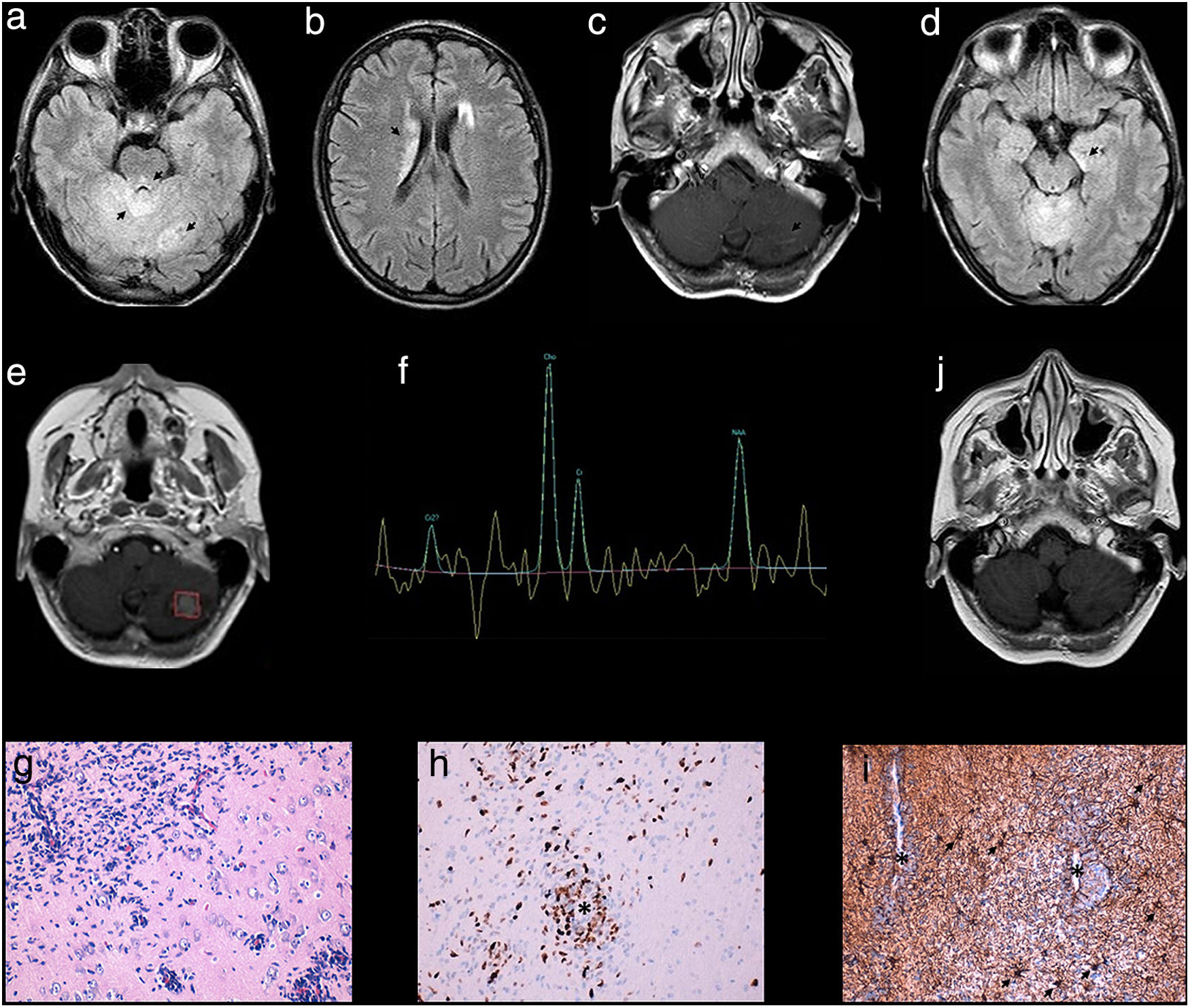
![MRI findings: signal alterations in the cerebellum, midbrain, and caudate nuclei (a and b: 1.5 T, FLAIR sequence). Linear contrast enhancement of the cerebellar lesions (c: gadolinium-enhanced T1-weighted sequence). At 6 months, signal alteration was observed in the left mesial temporal lobe (d: 3 T, FLAIR sequence). At 12 months, the larger lesion, in the left cerebellum, presented pseudonodular contrast uptake (e: gadolinium-enhanced T1-weighted sequence), with a Cho/NAA ratio > 2 on the spectroscopy study (f). Six weeks after onset of radiotherapy and TMZ treatment, the cerebellar signal alteration had reduced (j: FLAIR). A histology study showed infiltration of astrocytes, forming sleeves around blood vessels (g: hippocampus section; haematoxylin and eosin stain; magnification ×400). Immunohistochemistry showed a high proliferative index and perivascular localisation of tumour cells (asterisk) (h: MIB-1 [Ki-67] staining; magnification ×200), and reactive astrocytes (arrows) distributed uniformly in contraposition to the dense perivascular aggregation of tumour cells, with variable expression of glial fibrillary acidic protein (asterisks) (i: GFAP staining; magnification ×200). MRI findings: signal alterations in the cerebellum, midbrain, and caudate nuclei (a and b: 1.5 T, FLAIR sequence). Linear contrast enhancement of the cerebellar lesions (c: gadolinium-enhanced T1-weighted sequence). At 6 months, signal alteration was observed in the left mesial temporal lobe (d: 3 T, FLAIR sequence). At 12 months, the larger lesion, in the left cerebellum, presented pseudonodular contrast uptake (e: gadolinium-enhanced T1-weighted sequence), with a Cho/NAA ratio > 2 on the spectroscopy study (f). Six weeks after onset of radiotherapy and TMZ treatment, the cerebellar signal alteration had reduced (j: FLAIR). A histology study showed infiltration of astrocytes, forming sleeves around blood vessels (g: hippocampus section; haematoxylin and eosin stain; magnification ×400). Immunohistochemistry showed a high proliferative index and perivascular localisation of tumour cells (asterisk) (h: MIB-1 [Ki-67] staining; magnification ×200), and reactive astrocytes (arrows) distributed uniformly in contraposition to the dense perivascular aggregation of tumour cells, with variable expression of glial fibrillary acidic protein (asterisks) (i: GFAP staining; magnification ×200).](https://static.elsevier.es/multimedia/21735808/0000003600000003/v1_202103190809/S2173580821000080/v1_202103190809/en/main.assets/thumbnail/gr1.jpeg?xkr=ue/ImdikoIMrsJoerZ+w96p5LBcBpyJTqfwgorxm+Ow=)

