Objective: To evaluate clinical results and proinflammatory cytokine level in the tear film after PRK vs Epi.Lasik.
Methods: Twenty eight eyes of 14 patients for the correction of low myopia (<-3.0 Diopters S.E.) were included. PRK was done in one eye and Epi-Lasik in the fellow eye. In the cases with PRK, epithelium was removed with a rotary brush. Cases with Epi-LASIK, an epithelial flap was created with the Amadeus II microkeratome (AMO). Aspheric ablation was done using the excimer Schwind laser in both techniques. During the first four days re-epithelization time and discomfort were evaluated; visual acuity, manifest refraction and high order aberrations were evaluated at the 1st week, 1st and 3rd months. A sample of 50 microliters of tears was obtained to measure IL-1beta, IL-6, IL-8 and TNF- alpha by ELISA prior to surgery and at 1st and 4th day
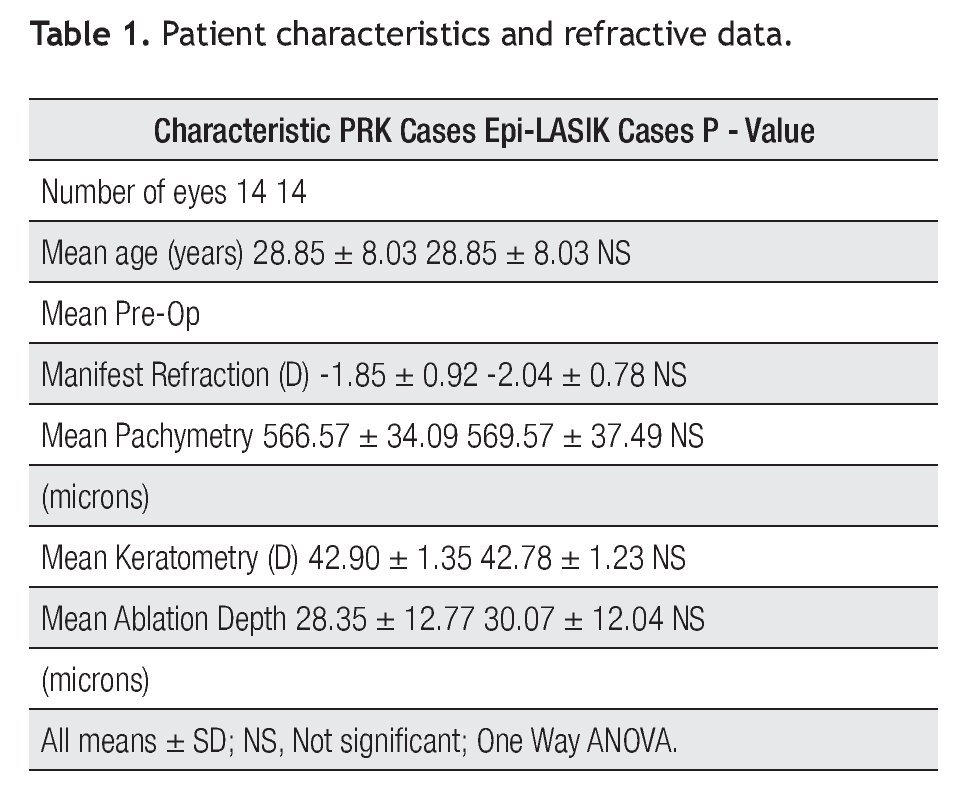
Results: Fourteen eyes underwent PRK (-1.85D +/- 0.92) and 14 eyes underwent Epi-LASIK (-2.04 D +/- 0.78). At three months, manifest refraction was -0.15 D (+/- 0.42 D) and - 0.12D (+/- 0.35). Efficacy index was 0.86 and 0.89, in cases with PRK and Epi-LASIK respectively. Safety index was one in both techniques. No significant differences between groups (p>0.05) was found in high order aberrations at three months. Pain was higher (p<0.05) only in the first day in PRK cases. In both surgical techniques, IL-6 concentrations in the tear film were statistically significant higher on day one, and lower but significant different on day four (p<0.05). IL-8 was statistically significant higher on day four, (p<0.05), and not different between control and the 1st day in both procedures (p<0.05). There were no measurable levels of IL1-beta and TNF-alpha in both techniques. No differences between groups were found for any cytokine comparing concentration by day.
Conclusions: In this study good predictability, efficacy and safety was found in both techniques. Similar level of total high order aberrations were induced in both procedures. Faster re-epithelization and less pain were found in the cases with epi-LASIK. Similar level of haze was induced in both procedures. IL-8 and IL-6 seem to play a role during the acute phase of wound healing of corneal surface ablation. The level of cytokines was not significantly different between both groups.
Objetivo: Evaluar resultados clínicos y niveles de citoquinas proinflamatorias en la lágrima después de PRK vs Epi-Lasik.
Métodos: Se incluyeron 28 ojos de 14 pacientes programados para la corrección de baja miopía (<-3.0 Dioptrias). Se realizó PRK en un ojo y Epi-Lasik en el ojo contralateral. En los casos con PRK, el epitelio fue removido con cepillo rotatorio. En los de Epi-LASIK, el flap epitelial fue realizado con el microkeratomo Amadeus II. La ablación asférica fue realizada usando el laser excimer Schwind, en ambas técnicas. El tiempo de re-epitelizacion, y las molestias fueron evaluadas durante los primeros cuatro días. La agudeza visual, la refracción manifiesta y las aberraciones de alto orden, fueron evaluadas a la semana y al primer y tercer mes. Una muestra de 50 microlitros de lágrima fue obtenida antes de la cirugía, y al primer y cuarto día después de la cirugía, para medir la cantidad de IL-1 beta, IL-8, and TNF-alfa por medio de la técnica de ELISA.
Resultados: Catorce ojos fueron operados con la técnica PRK (-1.85D +/- 0.92) y 14 ojos con Epi-LASIK (-2.04D +/- 0.78), a los tres meses la refracción manifiesta fue en promedio -0.15 D (+/- 0.42 D) y -0.12 D (+/- 0.35), respectivamente. El índice de eficacia fue 0.86 y 0.89 en PRK y Epi-LASIK, respectivamente. El índice de seguridad fue uno en ambas técnicas. No hubo diferencias estadísticas significativas (p >0.05), en cuanto a las aberraciones de alto orden, a los tres meses entre los dos procedimientos. El dolor fue más alto (p< 0.05), solo en el primer día en los casos de PRK. En ambas técnicas, las concentraciones de IL-6 en la película lagrimal fueron estadísticamente significativamente más altas en el primer día y significativamente más bajas al cuarto día (p<0.05). La concentración de IL-8 fue estadísticamente significativa más alta al cuarto día (p<0.05) en ambos grupos, pero no diferente entre el grupo control y el primer día (p<0.05). No hubo niveles medibles de IL-1 beta y TNF-alfa, en ambas técnicas. Tampoco existieron diferencias significativas de alguna citoquina comparándola entre los dos grupos.
Conclusiones: En ambas técnicas quirúrgicas se encontró buena predictibilidad, eficacia y seguridad. Niveles similares de aberraciones de alto orden fueron inducidos en ambos procedimientos. Una re-epitelizacion más rápida y menos dolor, se encontró en los casos de Epi-LASIK. Un nivel similar de visión borrosa fue inducido en ambos procedimientos. IL-8 e IL-6 parecen tener niveles de importancia durante la fase aguda del proceso de curación inmediata, después de cirugía de superficie corneal. El nivel de citoquinas no fue significativamente diferente entre ambos grupos.
Introduction
Different variations on conventional surface ablation procedures for refractive corneal surgery have been developed in recent years including PRK with application of mitomycin C, LASEK, and more recently Epi-LASIK.1-5 Each of the techniques aims to reduce postoperative pain, speed visual recovery and eliminate haze. Otherwise, numerous strategies have been investigated to control the inflammatory acute phase produced by these techniques.6-9 Knowledge with respect to immunology of corneal wound healing after corneal surface ablation, could help to understand effects on corneal clarity, predictability and stability.6-7 On the other hand, evidence of the presentation of tear film inflammatory mediators in dry eye,10 contact lens wearers,11 and infection,12 including proinflammatory cytokines and chemokines, have been detected in the tear fluid and/or conjuntival epithelia. In this study, we evaluated clinical results and different cytokine levels in the tear film during the acute phase after PRK and Epi-LASIK.
Methods
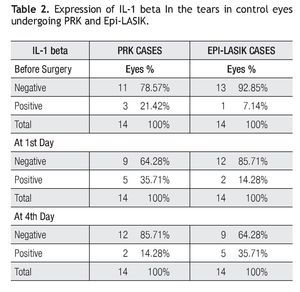
Twenty eight eyes of 14 patients with low myopia (< 3.00 D) scheduled for corneal refractive surface ablation by the same surgeon (F.V.P) at the Centro de Oftalmologia San Jose were studied prospectively (Table 1). One eye was operated with PRK and the fellow eye with Epi-LASIK, assignment of which eye received each treatment was determined randomLy. All patients received an explanation about the surgeries, and signed an informed consent in accordance with institutional guidelines and the Declaration of Helsinki.
Ophthalmic examination previous to the surgery included: Manifest refraction, cycloplegic examination, keratometry, topography, elevation map, slit lamp biomicroscopy and fundus examination. Patients with Diabetes mellitus, connective tissue disease, corneal disease, and glaucoma were excluded from the study. All users of contact lenses (four eyes of two patients) interrupted the use for at least two weeks and were examined before the surgery.
Surgical technique
The PRK procedures were performed in a standardized manner. A drop of tetracaine hydrochloride 0.5% was instilled five minutes before and just prior to the procedure. Afterwards, asespis was done on the lids using povidone - iodine 5% and a speculum with suction was placed in the operative eye. The corneal epithelium was removed with a rotary brush and the detritus removed with a sponge; immediately after ablation. The corneal stroma was irrigated with a balanced salt solution and a contact lens was placed.
The Epi-LASIK procedures were done as follows: Lid preparation was done as PRK cases. Afterwards, an epithelial flap was created with the Amadeus II microkeratome (Advanced Medical Optics®, Inc., Santa Ana, CA).
It achieves epithelial separation by movement of 11 000 oscillations per second of a disposable poly (methylmethacrylate) (PMMA), with an advance speed of 2 mm/s. In our cases, nasal hinge was intended to 0.8 mm to obtain a free epithelial flap in the most cases. The microkeratome comes in four different sizes: 8.5, 9.0, 9.5, and 10.0 mm. We found larger ring sizes advantageous and performed most of our cases with the 9.5 mm ring. Finally, the unit also allows for altitude adjustment in order to provide maximum suction performance. The preassembled microkeratome is applied to the operative eye, its central circular opening around the limbus. When suction is activated, a Barraquer tonometer ensures adequate suction, during the cut drops of balanced salt solution acts as lubricant to the cornea. Afterwards, the foot pedal is pressed, oscillating block runs parallel to the horizontal cornea plane, separating the epithelial flap, after the PMMA block runs on reverse movement, when it reaches final position the suction is released and the device is removed from the eye. With the use of a forceps, epithelial sheet is removed when a free flap is achieved or amputed when a residual hinge is obtained. With the use of a Merocel sponge, corneal stroma is dried to be ablated, and a therapeutic contact lens is applied to the eye as a bandage, as usual postoperative care in corneal surface surgery. In both techniques, drops of topical gatifloxacin 0.3% were instilled previous speculum remotion. All treatments were performed using aspheric ablation with the Schwind (Schwind eye-tech-solutions) excimer laser in both techniques.
Tear collection
Immediately previous to the surgery and at the 1st and 4th day, a tear fluid sample was collected with a 50-μL volume glass capillary tube (Eppendorf®, Freemont, CA) as described by Jones et al.13 by capillary action from the tear meniscus in the lateral canthus. The tear solution was stored at -20°C until ELISA was performed.
The concentrations of cytokines were determined with commercial ELISA kits. For these assays, tear samples were diluted in ELISA buffer (supplied by the manufacturer) to a final volume of 100 to 200 microliters. These assays were performed as recommended by the manufacturer (Pierce®, Rockford, IL). Samples were thawed, and aliquots of each sample applied to a PD-10 G-25 column (pharmacia®, Piscataway, NJ) preequilibrated with 3mm Tris-HCL, ph 7. The flow-through was discarded and protein eluted with 3.5 mL. This procedure desalts the specimens but recovers more than 98% of the protein, Samples were snap-frozen on dry ice, lyophilized, and resuspended in phosphate-buffered saline and aliquots used for analyses of cytokines. Samples were assayed for protein concentration using the BCA assay (Pierce®, Roc-®, Roc-, Rockford, IL). Aliquots were assayed for IL-1 beta, IL-6, IL-8 and TNF-alpha by enzyme-linked immunoadsorbent assay using commercially available kits.
The results were evaluated using One-Way ANOVA with SPSS statistical analysis software (version 17.0, SPSS® Inc.). A p value less than 0.05 was considered significant. All patients completed the follow-up to obtain samples of tears, and three months to evaluate clinical results.
Postoperative Care
Immediately after the surgery, patients were instructed to apply medications in both eyes as follows: during the first week gatifloxacin 0.3%, one drop four times daily and diclofenac one drop two times daily. During the first three days, all patients were offered oral ketorolac 10 mg two-four times a day for pain throughout the postoperative course. After complete corneal re-epithelization, the therapeutic contact lens was removed and fluorometholone acetate 0.1% one drop four times daily was prescribed during the first month and then tapered over three months. Topical corticosteroids were used for prolonged periods in any patient who developed haze.
Postoperative subjective pain was measured using the Visual Analogue Scale (VAS). The patient was asked to place a mark on a 100 mm long line that showed a continuum from no pain to severe pain.14
Corneal subepithelial haze was graded from 0 to 4 as follows: 0 = clear; 0.5 = faint; 1 = mild, seen only by oblique indirect illumination; 2 = moderate, seen by direct illumination; 3 = easily visible but not affecting refraction; 4 = dense, impairs view of intraocular structures and possibly affects refraction.
Results
Ephitelium
Complete re-epithelization occurred in all eyes between three to five days postoperatively. Three eyes (21.43%) in the PRK group and five (35.71%) in the Epi-LASIK group had healed within three days postoperatively, while 11 eyes (78.57%) and 14 (100%), (p<0.05) within four days in the PRK and Epi-LASIK group respectively. All eyes had complete re-epithelization within five days and no eye had recurrent corneal erosions, infection or other reepithelization complication.
Pain
At one day postoperatively the mean VAS score was 8.50 + 1.09 (SD) in the PRK group and 5.92 + 2.09 in the Epi-LASIK group; the difference between groups was significant on day one (p<0.05). Two days after the procedure, the mean VAS score was 3.21 + 1.18 in the PRK group and 2.71 + 0.91 in the epi-LASIK group, and was not statistically significant between groups in the VAS score (p>0.05). All patients reported their eyes felt normal one week after the procedure.
Manifest Refraction
Before the procedure, the mean spherical equivalent (MSE) was -1.85D + 0.92 (SD) in the PRK group and -2.04 D + 0.78 (SD) in the Epi-LASIK group with no significant difference (p > 0.05). After three months, MSE was 0.15 D + 0.42 (SD) in the PRK group and 0.12 + 0.35 (SD) in the Epi-LASIK group, and with also no significant difference (p>0.05).
Efficacy and safety
Three months after surgery, efficacy index (mean postoperative UCVA / preoperative BSCVA) was 0.86 and 0.89 in cases with PRK and Epi-LASIK respectively, with no significant differences between these groups (p>0.05). Safety index (mean post-BSCVA / mean pre - BSCVA) was 1.0 in both groups, with also no significant differences (p>0.05).
Corneal Haze
One month postoperatively, the mean haze score was 0.33 (range 0-1) and 0.25 (range 0-1) in the PRK and Epi-LASIK groups respectively. Two months later, it was 0.08 in the both groups, with no significant differences between the groups during all follow-up.
RMS
Before the surgery, root mean square (RMS) error of total high order aberrations were 0.27 microns + 0.07 (SD) and 0.27 microns + 0.1 (SD) and at three months 0.29 microns + 0.1 (SD) and 0.30 microns + 0.1 (SD), in the PRK and Epi-LASIK cases respectively. There was no significant difference between groups (p>0.05).
IL-1 beta in tear fluid
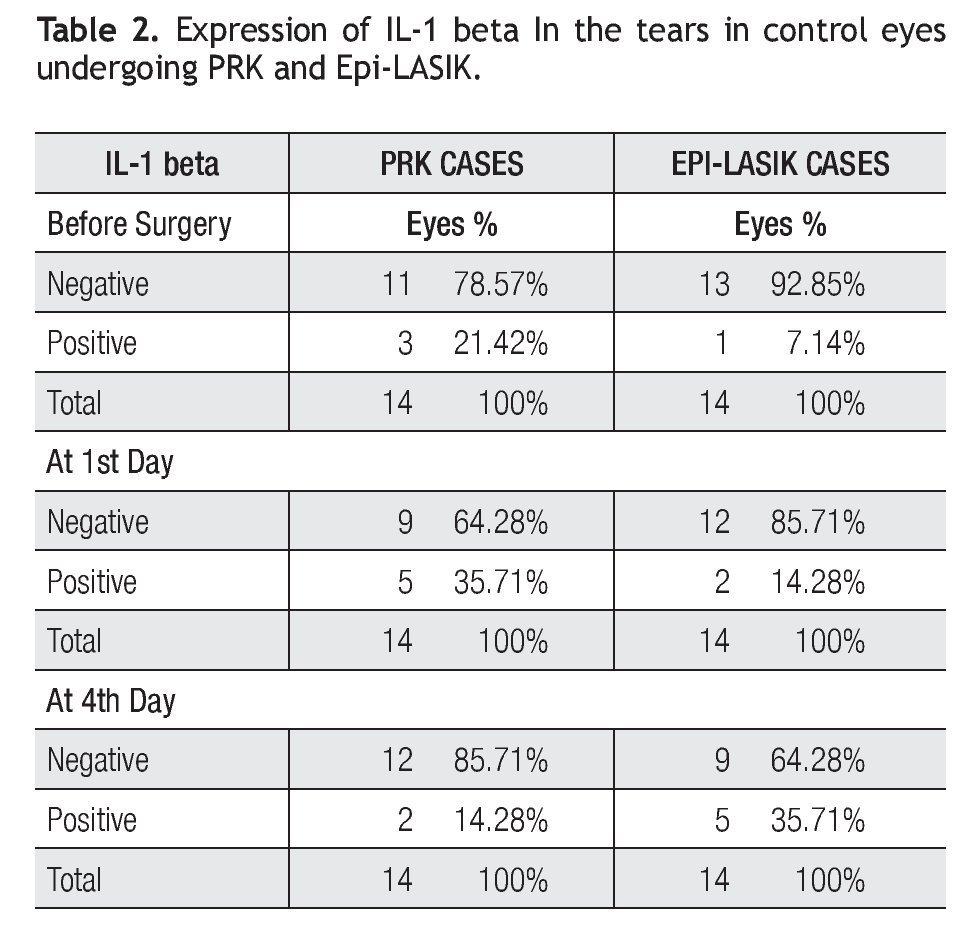
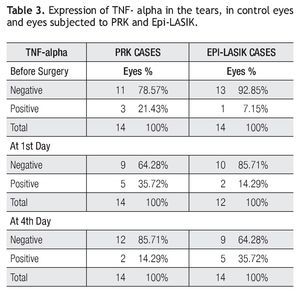
The concentration of the IL-1 beta in the control period and during the acute phase after the procedure are presented (Table 2). In fact, concentrations were not detected in all cases and hardly detected in some samples. In the PRK cases group as follows: before the surgery, detected in three samples (range 0 to 9.65 pg/mg), on day one, detected in six samples (range 0 to 12.9 pg/mg) and on day four, detected in two samples (range 0 to 6.12 pg/ mg). In the Epi-LASIK cases as follows: before the surgery, detected in one case with 8.5 pg/mg, on day one detected in two samples ( Range 0 to 3.18 Pg/mg), and on day four, detected in five samples (range 0 to 16.12 pg/mg) (Table 2). The 1st day after PRK and 4th day after Epi-LASIK more quantities of IL-1 beta were detected, nevertheless the differences were not statistically significant between the procedures and between the concentra- tion before and after the surgery.
IL-6 in tear fluid
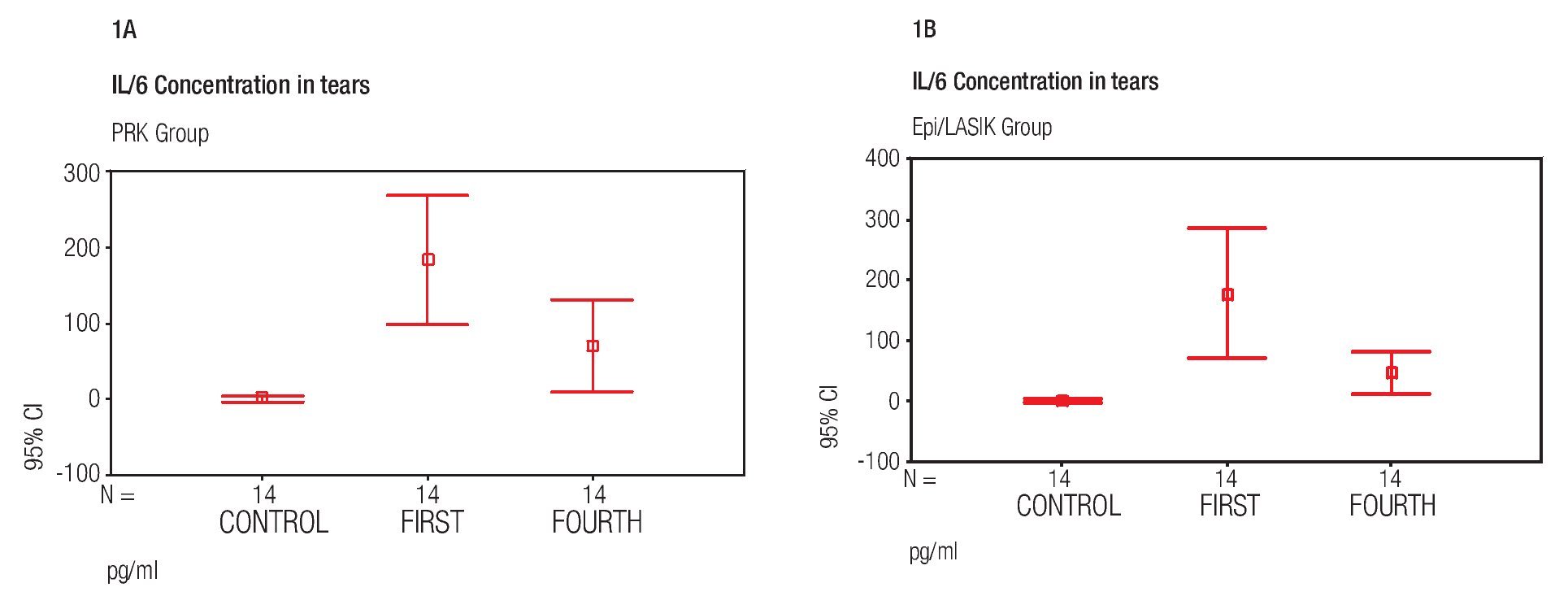
In this study, we found wide variations in the level of IL-6 concentration before and after surgery. In the PRK group, prior to surgery, concentrations were detected in only three cases with a mean of 2.23 pg/mL +/- 5.87 (SD), ranging from undetectable to 21.16 pg/mL. In contrast; On day one (mean, 183.74 pg/mL +/- 148.42) ranging from 64.02 pg/mL to 662.59 pg/mL; and on day four (mean, 71.76 pg/mL +/- 106.52) ranging from 20.27 pg/mL to 428.48 pg/mL. In Epi-LASIK cases, before surgery, concentrations were detected in three cases, with a mean of 1.71 pg/mL +/- 3.91, ranging from undetectable to 13.25 pg/mL; on day one (mean of 177.89 pg/mL +/- 184.54) ranging from 4.02 pg/mL to 575.09 pg/mL; and on day four (mean 47.41 pg/mL +/- 63.09) ranging from 0.94 pg/ mL to 252.94 pg/mL (Figure 1A and 1B). There was a significant difference between the control group before surgery and the 1st and the 4th day after surgery in the PRK group and in the Epi-LASIK group procedure (p<0.05), but the values of the PRK group and Epi-LASIK group were not significantly different (p>0.05) between them.
Figure 1. Mean IL-6 concentration before surgery (control) and at first and fourth day after the PRK surgical procedure (error bar = standard deviation SD) A). Mean IL-6 concentration before surgery (Control) and at first and fourth day after the Epi-LASIK surgical procedure (error bar = standard deviation SD) B).
IL-8 concentration in the tear fluid
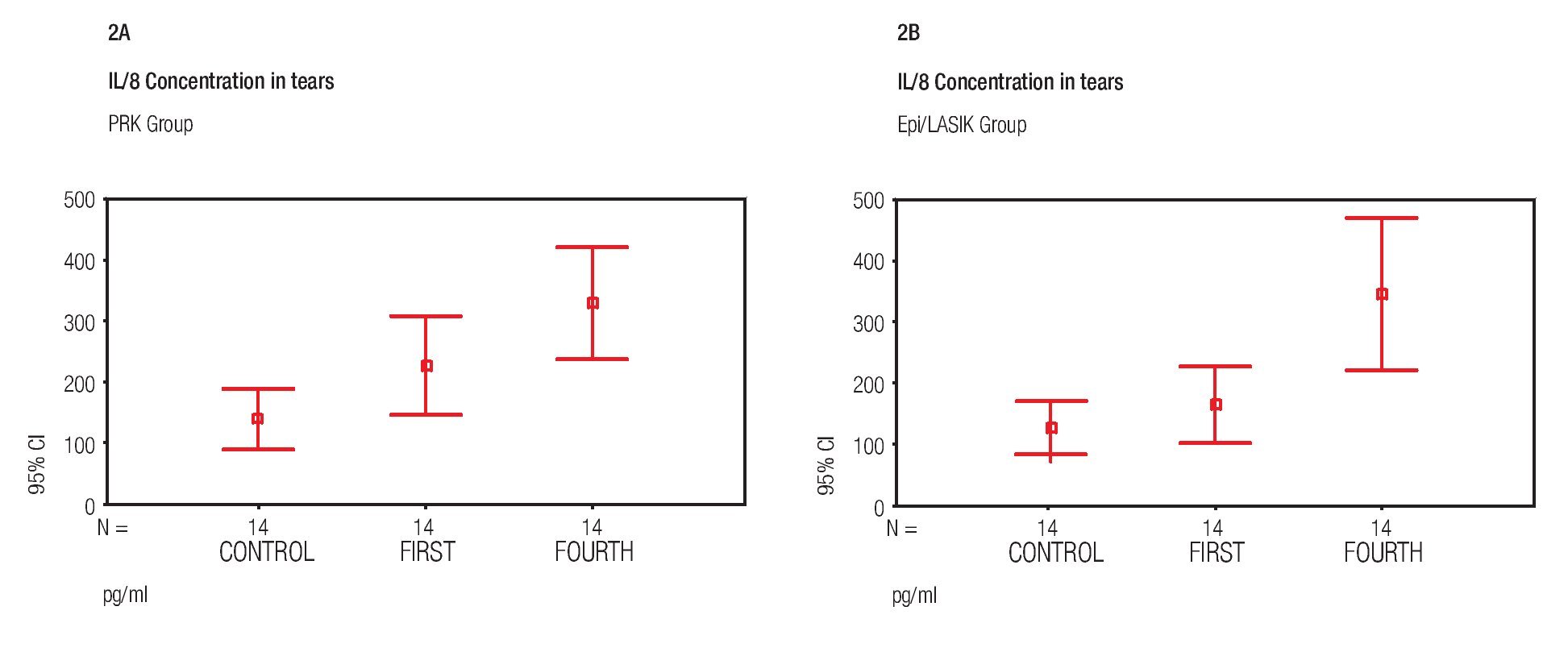
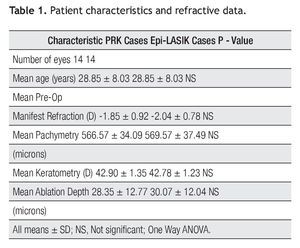
Concentrations of IL- 8 were the only interleukin detected in all samples of both surgical procedures, before and after the surgery. The variations were found as follows: In the PRK group, mean concentration of the proinflammatory cytokine IL-8 pg/mL was 140.73 pg/mL +/- 85.60 (SD) prior to surgery, 228.51 pg/mL +/- 139.85 (SD) on day one and 328.76 pg/mL +/- 157.57 (SD) on day four. In the Epi-LASIK group, mean concentration was 130.15 pg/mL`+/- 73.40 (SD) prior to surgery, 165.38 pg/mL +/-104.58 (SD) on day one, and 344.52 pg/mL +/- 215.29 (SD) at 4th day (Figure 2A and 2B). There was no statistically significant difference (p>0.05) between the values obtained prior to surgery and those obtained on day one, but there was, between values of day one and values of day four (p<0.05) of both procedures. No statistical difference was found (p>0.05) comparing values of samples obtained the same day between PRK and Epi-LASIK.
Figure 2. Mean IL-8 concentration before surgery (Control) and at first and fourth day after the PRK surgical procedure (error bar = standard deviation SD) A). Mean IL-8 concentration before surgery (Control) and at first and fourth day after the Epi-LASIK surgical procedure (error bar = standard deviation SD) B).
TNF-alpha concentration in the tear fluid
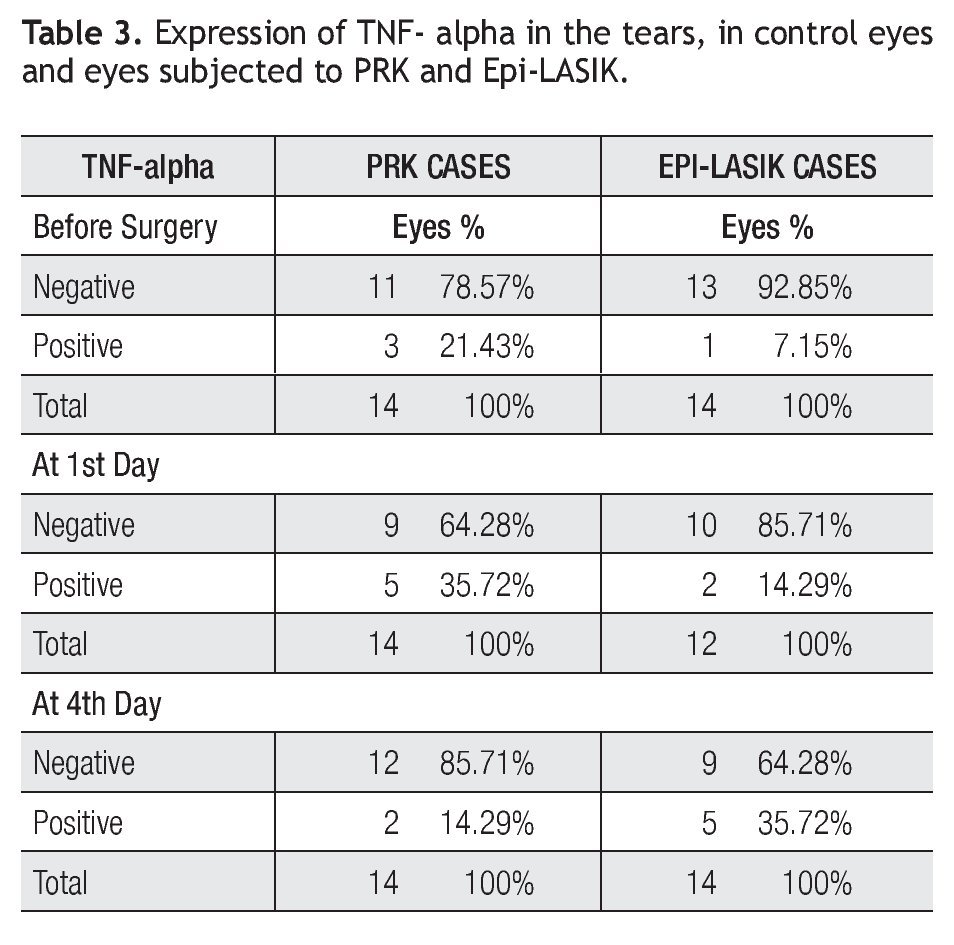
TNF-alpha was the least detected interleukin in this study. From 12 samples obtained per group, concentrations were detected in only two eyes of two different patients during the study period. In the group of PRK, values obtained were 35.71 and 46.96 pg/mL in the control day before surgery, 24.04 and 71.13 pg/mL at 1st day after surgery and 53.21 and 48.21 pg/mL at 4.th In the group of Epi-LASIK values obtained were 52.79 and 56.96 pg/ mL in the control day before surgery, 29.88 and 57.79 pg/mL at 1st day after surgery, and 64.76 and 50.71 pg/mL at 4th (Table 3). No statistically differences (p>0.05) were found during all follow-up.
Discussion
The original purpose of this study was to evaluate clinical results and the production of different proinflammatory cytokines in the tear film, in PRK vs Epi-LASIK. With respect to the technique used in this study, in PRK cases; using a rotary brush, we tried to remove the least amount of the epithelium, (around 8 mm) this maneuver being dependent of the surgeon and patient cooperation, on the other hand in Epi-LASIK cases intended flap diameter was 8.5 mm in all cases, taking into account the ablation diameter zone of 6.5 millimeters. Nevertheless, complete epithelial regeneration was presented in all Epi-LASIK cases at least at 4th day, while three cases had to wait one day more in PRK cases. With respect to the Epi-LASIK technique, the epithelium flap was gently detached using a microsponge, because in previous studies the remaining epithelial flap had been associated to pain increment,14,15 and we believe that this maneuver contributed to decrease the inflammatory response and some degree of discomfort. In fact, the measured pain differences between the two treatments studied revealed statistically more pain using VAS scale, on postoperative day one and day three, although on day four differences were only marginally higher in the PRK group (p=0.029).
In our study, we noticed that post-op manifest refraction was not dependent of the technique (p>0.05), further demonstrating good predictability, efficacy and safety in both techniques and perhaps in accordance with other surface ablation techniques, no significant level of haze in this group of myopia (<3.0D).16,17 Otherwise, customized aspheric ablation induced a small increase in corneal higher-order aberrations with no differences between both procedures.
Corneal wound healing after excimer surface ablation can be divided into acute, intermediate and late phases.18 The acute phase, which begins immediately after PRK and lasts several days, is marked by a large influx of polymorphonuclear leukocytes in rabbits.19 The number of stromal keratocytes increases above baseline in the intermediate phase, which begins a few days after PRK, when re-epithelization is complete.20 After re-epithelization, compensatory epithelial hyperplasia occurs. This hyperplasia may return to normal in three to 12 weeks.19 From a few weeks to months after PRK, during the late phase, the number of keratocytes tend to gradually return to baseline.18 After PRK, prostaglandins, leukotrienes, growth factors and other cytokines help regulate the corneal wound healing. This process has been studied jointly with alteration in the composition of the basal tear film (lactoferrin, secretory IgA and lysozyme)21,22 compared with reflex tears.23 Recently, studies have contributed to determine the expression and the functions of immune molecules in the tear fluid and corneal epithelia24,25 as well as alteration in the level of pro-inflammatory cytokines detected in the tears in some conditions as dry eye,26 contact lens wearers,27 ocular rosacea,28 keratoconus,29 after LASEK and Epi-LASIK,30 eyes with glaucoma treated topically,31 anterior segment eye diseases,32 and cystic fibrosis.33 Further, many studies have shown expression of immune mediators during corneal wound healing.30,34,35 IL-1 beta is capable of initiating a series of immunomodulatory chain reactions influencing the behavior of cells of the innate and adaptive immune systems at the ocular surface.36 Nevertheless, it has not been consistently detected in the tear during corneal inflammatory processes.37,38 In a previous study IL-1 beta concentration differences in tears were not statistically significant after LASIK.39 In our study, levels of IL-1 beta were undetectable or low in the tear film of a few samples with no significant differences between both groups, which did not correlate with levels in others published studies. For example in subjects with dry eye,40 levels ranged from 80 to 180 pg/mL, whereas that of healthy controls was 30 pg/mL. This data suggests that IL-1 beta may be just below the detection level of the assays performed, probably because it is bound to receptors remaining undectectable in this form,40 or it may not play a role in eyes undergoing corneal surface excimer laser surgery.
Interleukin IL-6 is a pleiotropic cytokine that plays an important role in the regulation of a variety of host immune responses, including inflammation, acute-phase responses, hematopoiesis, and differentiation of B-cells.41 Higher concentration in the tears has been associated to neutrophil activation during corneal infection,42 and it decreases with corneal healing.43 In this study IL-6 concentrations in tears were elevated from undetectable or low levels before the surgery to statistically significant higher concentrations on day one, and lower but significant different concentrations on day four, perhaps correlating with corneal re- epithelization. Significantly different concentrations of IL-6 between the two procedures were not found (p<0.05). Nevertheless, although higher levels were found in the Epi-LASIK group compared with the PRK group on day one, they were not statistically different (p>0.05). IL-6 probably rises very early during inflammatory processes, and after 24 hours their concentration perhaps decrease below detectable levels.
In this study, the proinflammatory cytokine, which played a major role between those studied, was IL-8. Previous studies have shown release of IL-8 by epithelial corneal cells during corneal wound healing.24 Further, high concentrations in the tears remained after corneal re-epithelization.43 In our study, IL-8 was statistically significant higher on day four, (p<0.05), and not different between control and on day one in both procedures (p>0.05). During follow up no differences were found between the two procedures (p>0.05).
Although elevation of TNF-alpha concentration in the tears has been associated with corneal inflammatory processes,31,37 undetectable or low levels of TNF-alpha with no significant variation in both procedures were found in our study (p>005). Perhaps this production was low or these molecules remained attached to the receptor, doing their function.
Similar studies have used ELISA protocols to obtain this type of data. Sample size and assay sensitivity limits among commercially available kits have usually limited analysis to the measurement of one of a few proteins in the tear sample.44 Given the complexity of biological process, it has long been recognized that it would be of great value if one could obtain differential data on a broad range of proteins in tear samples (Figure 1 and 2).
In summary, with exception of less pain on day one and faster reepitelization observed in the epi-LASIK cases, similar good clinical results were observed in both surgical procedures, with evidence of the participation, detected in the tears, of IL-6 and IL-8 during the post-operative acute phase, with no significant different concentration of pro-inflammatory cytokines studied between PRK and epi-LASIK cases.
The authors have no proprietary interest in the development or marketing of any equipment or device used in this research.
Corresponding author: Fortino Velasco M.D.
Centro de Oftlamología San Jose. Av Morones Prieto Pte 3 000. C.P. 64710. Monterrey, Nuevo Leon. Mexico.
Phone: (52) 81 8333 5030. Fax: (52) 81 8333 4601.
E-mail: fortinovelasco@yahoo.com.mx
Received: March 2011.
Accepted: October 2011