Gynecomastia is defined as a benign proliferation of breast tissue in males and results from an imbalance between estrogenic and androgenic activities.1 It may be due to decreased androgen production, and increased estrogen production, or an increased peripheral conversion of androgenic precursors. Other potential mechanisms include the blockade of androgenic receptors and increased androgen binding to its transporter protein (SHBG).2
Its less common causes include testicular tumors, which account for approximately 2% of cases of gynecomastia.3 We report a case of gynecomastia secondary to production of chorionic gonadotropin (HCG) by a testicular germ cell tumor.
A 27-year-old male patient with an unremarkable history except for a septoplasty procedure and who smoked 10 cigarettes daily attended the clinic for painful bilateral gynecomastia over the previous three months. He reported no associated clinical signs of hypogonadism, body weight changes, use of drugs or cosmetic products, nor illegal drug consumption. His family history was unremarkable.
Physical examination revealed a normal male phenotype, with adequate body hair distribution. Weight was 71.7kg and height 1.71cm (body mass index 25kg/m2), and blood pressure was normal (130/80mmHg). A bilateral, grade II, symmetrical increase in glandular breast tissue was palpated, with no nodules and no secretion on expression. No goiter was palpated, and cardiopulmonary auscultation was normal. The abdomen was soft and amenable to pressure, with no masses or visceromegalies. Both testes were inside the scrotum, with volumes of 25mL and 15–20mL for the right and left testis, respectively. No testicular masses were palpated.
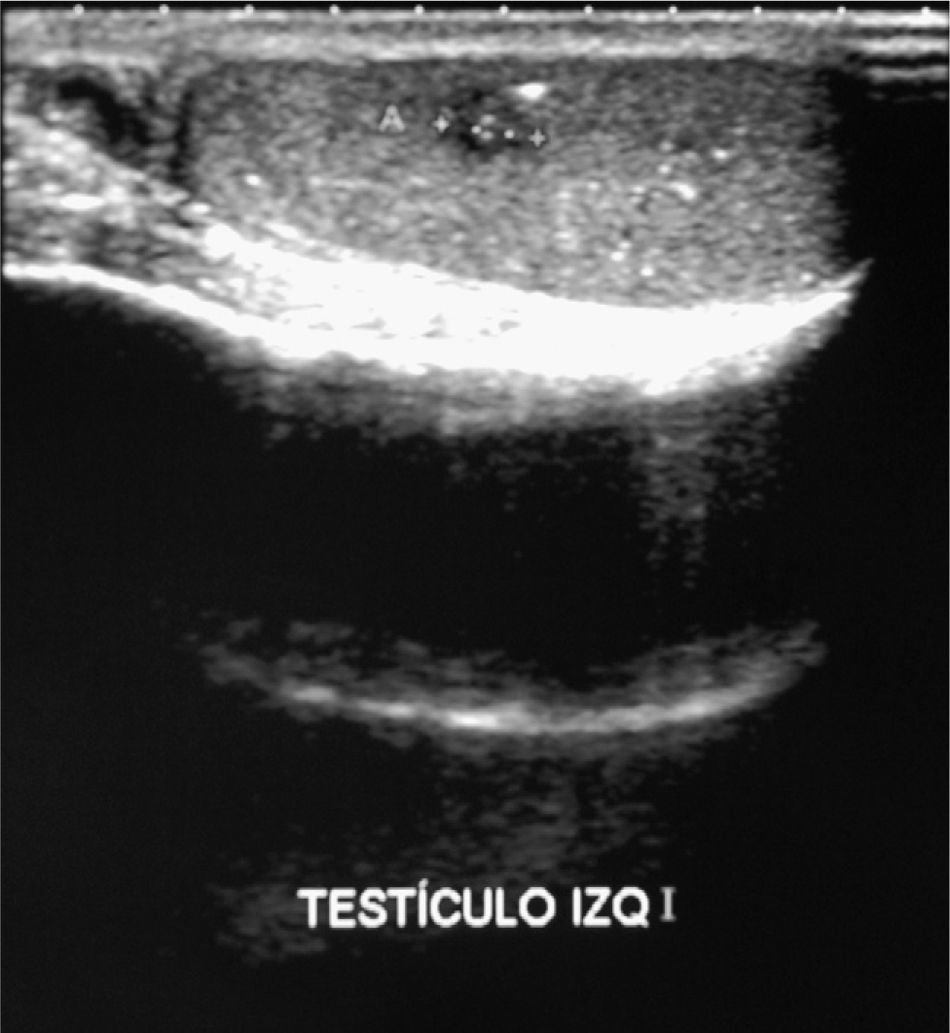
Based on this recent onset of bilateral gynecomastia with no identifiable cause, laboratory tests were requested and showed the following values: HCG 27,703IU/L (normal less than 2.5IU/L), LH 16.2IU/L (normal 0.87–7.6), FSH less than 0.1IU/L (normal 0.7–11.1); estradiol 143ng/L (normal 0–80); free testosterone 53ng/L (normal 8.8–27). Results of all other general laboratory parameters and thyroid hormone and alpha-fetoprotein levels were normal. A testicular ultrasound examination reported a normal right testis and a left testis with diffuse hypoechogenicity, with multiple scattered hyperechogenic images related to microlithiasis. A 6-mm hypoechoic area with ill-defined margins was seen in the middle part of the left testis (Fig. 1). Dilatation of left pampiniform plexus related to varicocele. Chest X-rays were normal.
The patient was referred to urology, where left transinguinal orchiectomy plus placement of testicular prosthesis were successfully performed. A CT scan of the chest, abdomen, and pelvis showed no adenopathies of lesions suggesting metastatic disease.
The pathological report described a burned-out testicular tumor associated with intratubular germ neoplasm with a lesion-free surgical margin. There was a whitish area consistent with scar tissue together with a granulomatous lesion with heavy histiocyte reaction, with no residual tumor cells. Therefore, a definitive pathological diagnosis was not possible. There was no apparent vascular infiltration. There was Leydig cell hyperplasia and intratubular stones.
In laboratory tests performed after surgery, HCG concentrations continued to be high (83,117IU/L). Chemotherapy was therefore decided upon at a multidisciplinary session. The patient lived in another region and said that he would rather continue treatment in his home town. His reference hospital was therefore contacted for transfer.
Gynecomastia results from an imbalance between estrogens and androgens. Physiological gynecomastia is very common in the neonatal period, adolescence, and males aged 50–80 years, and resolves spontaneously in a majority of cases.1 The most common causes in adults include persistent pubertal gynecomastia, drugs, or idiopathic gynecomastia. Other conditions such as liver cirrhosis, renal failure, malnutrition, hypogonadism, or hyperthyroidism are associated with gynecomastia.4
The multiple causes of pathological gynecomastia in young adults include tumors, mainly testicular tumors. In such cases, gynecomastia is due to HCG production in germ cell tumors or to estrogen production in Leydig cell tumors.5 Other tumors which have been associated with gynecomastia include feminizing adrenocortical tumors and neoplasms with ectopic HCG production (lung, gastric, renal, etc.).1
Testicular tumors are the most common solid neoplasms in males aged 15–35 years. Germ cell tumors account for 95% of all testicular cancers, and gynecomastia occurs in 2.5–6% of cases.6 This is associated with HCG production by foci of choriocarcinoma or trophoblastic cells in the tumor, and is considered a sign of poor prognosis.7
Germ cell tumors are divided into two main categories, seminomas and non-seminomatous tumors. Seminomas account for 50% of germ cell tumors and are usually confined to the testis at diagnosis. Tumor markers are normal, but HCG may be slightly elevated (usually to less than 100IU/L). Non-seminomatous tumors occur in younger patients, usually consisting of two or more histological subtypes, and up to 80–85% have elevated HCG or alpha-fetoprotein levels. The most aggressive histological variant is choriocarcinoma, which rarely occurs as the only component, but is present in 10% of mixed tumors. In these, HCG levels are usually higher than 1000IU/L.
In ultrasound examination, seminomas usually appear as well-defined hypoechoic lesions, while non-seminomatous tumors are typically heterogeneous, with calcifications, cystic areas, and irregular margins.8
The recommended treatment is radical inguinal orchiectomy, which allows for histological diagnosis and local control of the disease. Radiographic evaluation using CT of the abdomen and pelvis is recommended7,8 because regional metastases occur frequently in the retroperitoneal lymph nodes. If lung involvement is suspected, a CT scan of the chest should be performed. Although tumor markers (HCG and alpha-fetoprotein) are useful for the diagnosis of testicular tumors and have prognostic value, their greatest value is for disease monitoring following primary treatment.9
In this patient, HCG levels and the ultrasonographic characteristics of the tumor suggested a non-seminomatous germ tumor, but pathological analysis was not conclusive because it was a “burned out” tumor, a relatively common occurrence in testicular tumors. After surgery, the patient showed persistently high HCG levels, and chemotherapy was therefore decided upon. No data subsequent to chemotherapy are available, as this treatment was performed elsewhere.
Clinical history and physical examination allow for etiological diagnosis in most patients with gynecomastia.2 Liver, kidney, and thyroid function tests are useful to rule out disease in those organs. When gynecomastia has recently occurred, particularly in young adults, HCG, LH, testosterone, estradiol, and prolactin levels should be measured, unless the cause is apparent after a careful perusal of the clinical history and a physical examination including a testicular examination.1 It should be noted that testicular tumors may not be evident upon palpation in some cases.10 Early diagnosis of these uncommon cases is essential for early treatment and improved prognosis.
Please, cite this article as: Ollero García-Agulló D, et al. Ginecomastia de origen tumoral. Endocrinol Nutr. 2011;58:554–5.