Hepatic adenomas (HA) are benign tumours which can present serious complications, and hence all were resected in the past. It has now been shown that those smaller than 3cm not expressing β-catenin only result in complications in exceptional cases and therefore the therapeutic strategy has been changed.
Materials and methodRetrospective study in 14 HPB units. Inclusion criteria: patients with resected and histologically confirmed HA. Study period: 1995–2011.
Results81 patients underwent surgery. Age: 39.5 years (range: 14–75). Sex: female (75%). Consumption of oestrogen in women: 33%. Size: 8.8cm (range, 1–20cm). Only 6 HA (7.4%) were smaller than 3cm. The HA median was 1 (range: 1–12). Nine patients had adenomatosis (>10 HA). A total of 51% of patients displayed symptoms, the most frequent (77%) being abdominal pain. Eight patients (10%) began with acute abdomen due to rupture and/or haemorrhage. A total of 67% of the preoperative diagnoses were correct. Surgery was scheduled for 90% of patients. The techniques employed were: major hepatectomy (22%), minor hepatectomy (77%), and one liver transplantation. A total of 20% were performed laparoscopically. The morbidity rate was 28%. There were no cases of mortality. Three patients had malignisation (3.7%). The follow-up period was 43 months (range 1–192). Two recurrences were detected and resected.
DiscussionPatients with resected HA are normally women with large lesions and oestrogen consumption was lower than expected. Its correct preoperative diagnosis is acceptable (70%). The major hepatectomy rate is 25% and the laparoscopy rate is 20%. There was a low morbidity rate and no mortality.
El adenoma hepático (AH) es un tumour benigno que puede presentar graves complicaciones por lo que, clásicamente, todos eran resecados. Actualmente se ha demostrado que los menores de 3cm, y si no expresan β-catenina, solo se complican excepcionalmente, lo que ha cambiado la estrategia terapéutica.
Material y métodosEstudio retrospectivo en 14 unidades HPB. Criterio de inclusión: pacientes con AH resecado y confirmado histológicamente. Periodo de estudio: 1995–2011.
ResultadosFueron intervenidos 81 pacientes. Edad: 39,5 años (rango: 14-75). Sexo: mujeres (75%). Consumo de estrógenos en mujeres: 33%. Tamaño: 8,8cm (rango: 1-20cm). Solo 6 AH (7,4%) eran menores de 3cm. La mediana de AH fue 1 (rango: 1-12). Nueve pacientes presentaban adenomatosis (> 10 AH). El 51% de los pacientes presentaban síntomas; el más frecuente (77%) era dolor abdominal. Ocho pacientes (10%) comenzaron con abdomen agudo por rotura o hemorragia. El 67% de los diagnósticos preoperatorios fueron correctos. La cirugía fue programada en el 90% de los pacientes. Las técnicas fueron: hepatectomías mayores (22%), menores (77%) y un trasplante hepático. Un 20% fueron realizadas por laparoscopia. La morbilidad fue 28%. No hubo mortalidad. Tres pacientes presentaron malignización (3,7%). El seguimiento fue 43 meses (rango: 1-192). Se detectaron 2 recidivas que fueron resecadas.
DiscusiónLos pacientes con AH resecados son habitualmente mujeres con lesiones grandes, con un consumo de estrógenos inferior al esperado. Su diagnóstico preoperatorio correcto es aceptable (70%). La tasa de hepatectomías mayores es 25% y la de laparoscopia, 20%. Hemos obtenido una baja morbilidad y nula mortalidad.
Hepatic adenoma (HA) is a benign liver tumour which can present serious complications (rupture, haemorrhage, malignisation…). For this reason, traditionally most diagnosed HA have been resected.1–6 Some series have demonstrated that HA of less than 5cm that do not express certain markers (β-catenin) only rarely produce complications.1,7 Therefore, it is now being suggested that some HA1,7 should be treated conservatively. Given this therapeutic change, the Hepato-Biliary Pancreatic (HPB) section of the AEC (Spanish Association of Surgeons) suggested performing a retrospective study of resected HA prior to undertaking a prospective study of the current treatment of hepatic adenoma.
Materials and MethodsThis was a retrospective study performed in 14 HBP surgical units from January 1995 to December 2011. The inclusion criteria were patients with HA that had been resected and confirmed histologically. We were not able to establish how many patients with HA were assessed but not resected.
Data were gathered using a questionnaire which included the following variables: age, gender, oestrogen consumption, presence of HA related disease (glycogenosis, polyposis, tyrosinaemia…), size and number of HA, symptoms caused by the HA, preoperative radiological diagnosis and preoperative embolisation, type of surgery (elective or emergency), surgical technique used, and type of approach (laparotomy vs laparoscopy). Morbidity was measured using the Dindo-Clavien8 classification, histological studies were included and follow-up to quantify recurrence. Adenomatosis was defined as more than 10 HA.
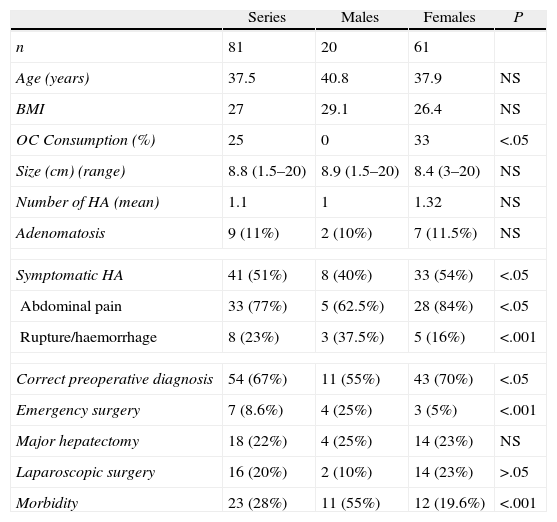
Results81 patients with HA underwent surgical resection (Table 1). The average age was 39.5 (range: 14–75). 75% of the patients were women. The mean body mass index was: 27kg/m2 (range: 21–41) Only 33% of the women had taken oral contraceptives (OC). No patient acknowledged having taken androgens or other anabolic steroids. One patient presented glycogenosis. The average size of the HA was 8.8cm (range: 1–20cm). The radiological diagnosis was made by ultrasound and computerised axial tomography (CT) in 100% of cases, and complemented with magnetic resonance imaging (MRI) in 67% of the patients. Only 6 HA (7.4%) were under 3cm. The median nodules count in the patients who did not have adenomatosis was 1 (range 1–6). Nine patients presented adenomatosis (>10 HA) (11%): 8 multifocal and one massive.9 51% of the patients presented symptoms, the most common being abdominal pain. Preoperative diagnosis of HA was correct in 67% of the patients (n=54). The most common incorrect preoperative diagnoses were: hepatocarcinoma (n=5), focal nodular hyperplasia (FNH) (n=4), angiomyolipoma (n=1), and metastasis (n=1). In the remainder (n=16) there were doubts between FNH and HA.
Surgery on 81 Patients With HA.
| Series | Males | Females | P | |
| n | 81 | 20 | 61 | |
| Age (years) | 37.5 | 40.8 | 37.9 | NS |
| BMI | 27 | 29.1 | 26.4 | NS |
| OC Consumption (%) | 25 | 0 | 33 | <.05 |
| Size (cm) (range) | 8.8 (1.5–20) | 8.9 (1.5–20) | 8.4 (3–20) | NS |
| Number of HA (mean) | 1.1 | 1 | 1.32 | NS |
| Adenomatosis | 9 (11%) | 2 (10%) | 7 (11.5%) | NS |
| Symptomatic HA | 41 (51%) | 8 (40%) | 33 (54%) | <.05 |
| Abdominal pain | 33 (77%) | 5 (62.5%) | 28 (84%) | <.05 |
| Rupture/haemorrhage | 8 (23%) | 3 (37.5%) | 5 (16%) | <.001 |
| Correct preoperative diagnosis | 54 (67%) | 11 (55%) | 43 (70%) | <.05 |
| Emergency surgery | 7 (8.6%) | 4 (25%) | 3 (5%) | <.001 |
| Major hepatectomy | 18 (22%) | 4 (25%) | 14 (23%) | NS |
| Laparoscopic surgery | 16 (20%) | 2 (10%) | 14 (23%) | >.05 |
| Morbidity | 23 (28%) | 11 (55%) | 12 (19.6%) | <.001 |
OC: oral contraceptives; HA: hepatic adenoma; BMI: body mass index.
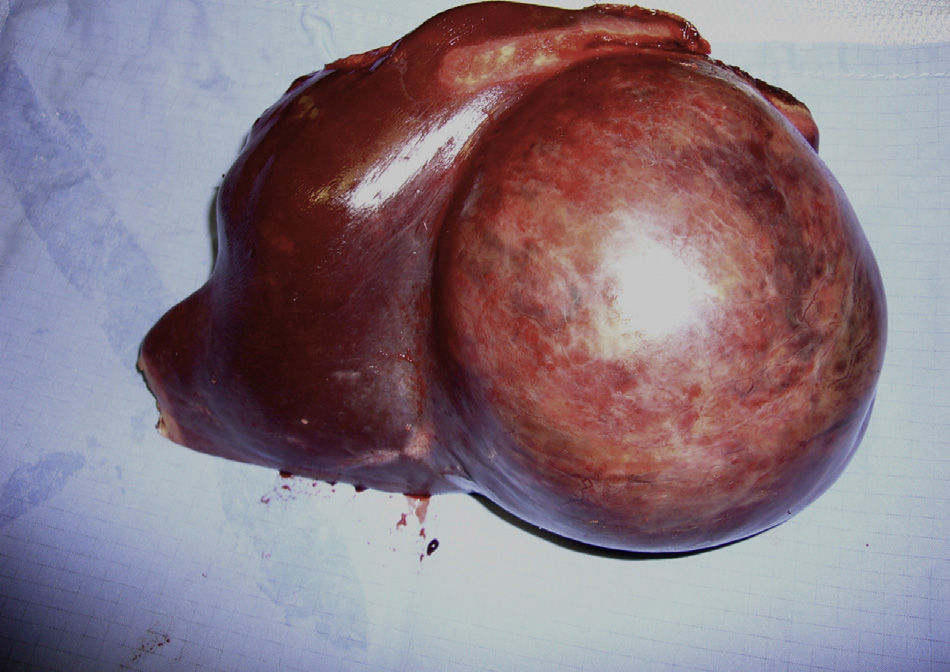
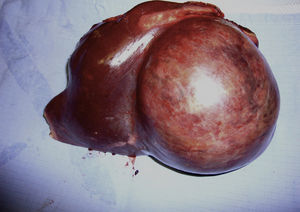
Surgery was elective in 90% of cases (73/81). The 7 emergency operations were performed on complicated HA (rupture/haemorrhage); the eighth patient with haemorrhage was embolised and underwent elective surgery. The techniques used were: major hepatectomy (22%), minor hepatectomy (77%) and a liver transplant after prior resection as new lesions had appeared (right-sided hepatectomy). 20% of the operations were performed using a laparoscopic approach. Morbidity was 28% (n=23). The morbidity was divided into two groups according to severity: Clavien I–II: 82% (n=19) and major Clavien: III–IV: 18% (n=4). There was no mortality. In the histological study, 2 patients presented malignisation of the HA to hepatocarcinoma, and foci of severe dysplasia were found in another patient, which represented 3.7% of the total patients. Of these patients, one was a male with a single tumour of 11cm (Fig. 1) and 2 were women: one had a tumour of 6cm and the other had hepatic adenomatosis. The very few patients with malignisation do not enable statistical conclusions to be extracted on any parameter associated with malignisation. The median follow-up was 43 months (range: 1–192). 2 recurrences were detected and they were reoperated on.
A comparison of the parameters studied was performed between males and females (Table 1). The fundamental difference is a greater number of males presenting with rupture and haemorrhage, which implied more emergency surgery, greater morbidity and a lower rate of laparoscopic surgery. The women more frequently presented with abdominal pain.
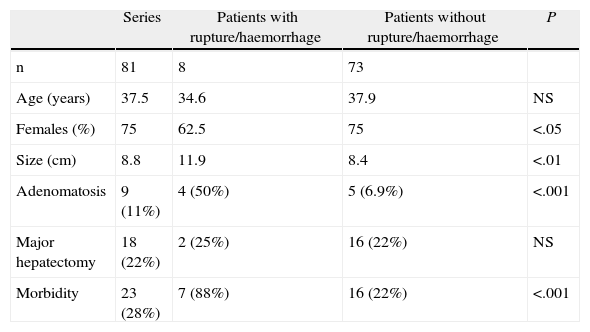
We made a comparison between the patients presenting with acute abdomen (rupture/haemorrhage) and those which were uncomplicated (Table 2). The size, presence of adenomatosis and postoperative morbidity was greater in the group of complicated patients (P<.05).
Comparison Between the Patients who Started With Acute Abdomen (Rupture/Haemorrhage) and Those who Were Uncomplicated.
| Series | Patients with rupture/haemorrhage | Patients without rupture/haemorrhage | P | |
| n | 81 | 8 | 73 | |
| Age (years) | 37.5 | 34.6 | 37.9 | NS |
| Females (%) | 75 | 62.5 | 75 | <.05 |
| Size (cm) | 8.8 | 11.9 | 8.4 | <.01 |
| Adenomatosis | 9 (11%) | 4 (50%) | 5 (6.9%) | <.001 |
| Major hepatectomy | 18 (22%) | 2 (25%) | 16 (22%) | NS |
| Morbidity | 23 (28%) | 7 (88%) | 16 (22%) | <.001 |
HA is an uncommon benign hepatic tumour, first described by Frerichs in 1861.1–5,10–14 They generally appear in healthy livers, there is usually only one and they tend to be of a variable size.1,2,13,15–18 Their incidence is approximately 1.3–5 cases per million inhabitants.2,4,6,10,12,19 They mainly, but not exclusively, occur in young women (15–45) who have received treatment with OC,1,2,5,10,16,18–20 and they only appear in 4.3% of males who have generally consumed anabolic steroids.1,13,21 Other risk factors for HA in males are: alcoholism, smoking and a high body mass index.19In our multicentre study 2 epidemiological findings are worthy of note: the high percentage of males comprising the series (25%), and the very low consumption of OC in the women (33%). This data is probably biased by retrospective gathering of information, because the rate of CO consumption in European patients with HA is usually 85%–95%.16 Curiously, both of these findings, the high amount of males and low OC consumption, have been observed in a different study performed on HA in China.10
The etiopathogenesis of HA has not been fully clarified, although the relationship between the consumption of OC or anabolic steroids containing androgens and the formation of HA is obvious, and is also dose-dependent.1,2,4,5,13,14,21HA is more frequent in patients with type Ia, III, and VI glycogenosis, where there is a greater risk of the HA becoming malignant, tyrosinaemias, galactosaemia, β-thalassaemias, steatohepatitis, haemochromatosis, familial polyposis and Fanconi because of the use of androgens as treatment.1,5,6,11,13,16,19,22,23 One of our patients presented glycogenosis as a disease associated with the HA.
Depending on the genotype, HA are classified as: HA with mutations in the HNF1α gene, HA with mutations of the β-catenin gene and with no markers.16,19,20,23,24 This classification has prognostic importance, as the HA group with mutation of the β-catenin gene has a greater risk of malignisation.6,19,20 For reasons of financial costs, genotype markers are not used routinely in Spain. Histologically, they are subdivided into steatosic (when they present more than 60% macronodular steatosis), inflammatory or telangiectasic and unclassifiable.1 This classification is important for therapeutic decision-making because, for example, telangiectasic HA have a greater tendency to rupture.1
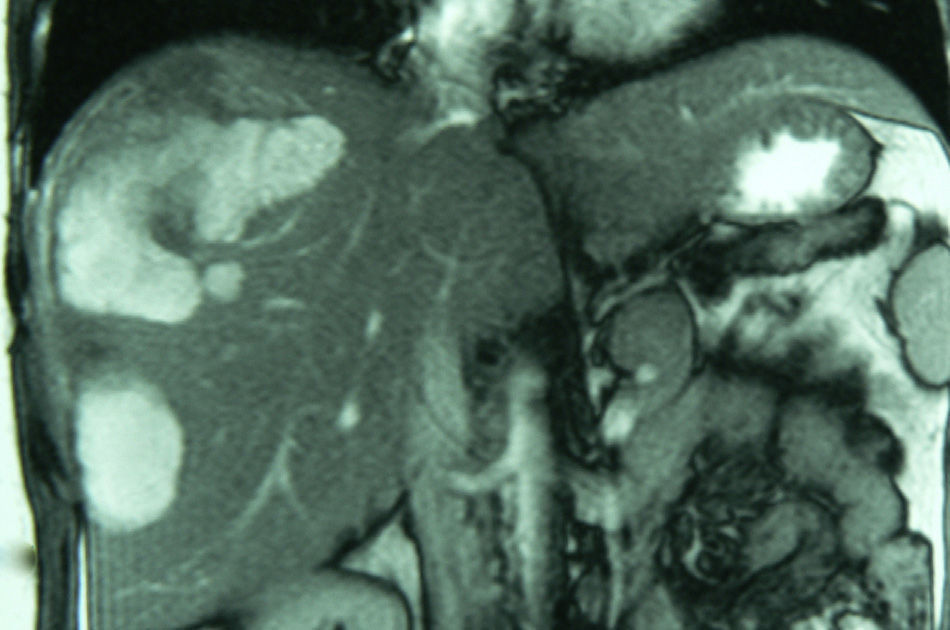
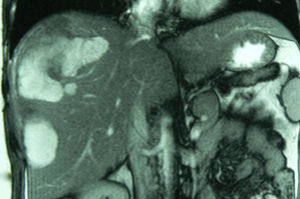
Diagnosing HA radiologically is not easy.10,25 The combined diagnostic sensitivity of the radiological techniques was 80%.12,17 In our series, which involved a long period of time, it was 54%. Traditional ultrasound, eco-Doppler and contrast ultrasound, multiphase CT scan and MRI,6,24,26–28 in particular, were the diagnostic methods most commonly used (Fig. 2). MRI with hepato-specific contrast can achieve 97% sensitivity and also enables the HA to be classified according to phenotype (steatosic vs inflammatory) which has therapeutic implications.24,27 PET/CT has been used to differentiate HA and FNH, obtaining promising results in a short series.6,26,27
The first measure after diagnosis of HA is for the patient to stop taking CO or anabolic steroids, if they had been taking them, and for patients with glycogenosis to start dietary measures, and a radiological reassessment should be made after 3–6 months.4,6,7,10,16,28 A reduction in size and even the complete disappearance of lesions has been observed.2,4–6,16,28 Subsequently it has to be decided whether the HA should be resected.
The therapeutic strategy has changed in recent years. Traditionally, almost all HA were resected because of possible complications,1,5,7 but currently a more conservative strategy is being adopted and it is only accepted that HA greater than 5cm6,11,16,22,29 should be resected. This cut-off point is because the risk of HA smaller than 5cm becoming malignant or haemorrhaging is extremely low.2,13,22,29 Another accepted indication for resection is an HA which grows more than 1cm between 2 successive controls,2,13 and another more contentious indication is to resect all HA diagnosed in males or in patients with glycogenosis because of the greater risk of complications.16,19,23
There are differing opinions as to how to act with tumours under 5cm. The debate concerns those measuring between 3 and 5cm in particular, as there is a degree of consensus that HA of less than 3cm should be monitored initially.1,2,13 There are no established protocols for the follow-up of unresected HA, although the most accepted is an annual MRI scan.4,16 Some authors recommend, almost systematically, puncture of the HA and inmunohistochemical analysis of the material obtained. If the result is positive for β-catenin, they recommend resection, because of the greater risk of malignant transformation. But not all authors agree with this approach, as it involves biopsy of all HA, which can entail complications, and the results with β-catenin are not all that reliable.1,7,13,16,23
Surgery can be via laparotomy, or by the increasingly common laparoscopic approach, when feasible.4,7 The percentage of major hepatectomies in our series is similar to that published in the literature (25%).10 In elective surgery, mortality does not usually exceed 1% (none in our study) and morbidity varies between 5% and 30% (28% in our series). These figures rise considerably in emergency surgery.29 The recurrence rate after resection in patients with single tumours is exceptional.16
The most serious long-term complication of HA, although uncommon, is malignisation.2,13,19,22,25 The exact mechanisms of malignant transformation are not known.23 An evolution from adenoma to carcinoma due to the presence of a carcinoma inside the adenomatous tissue has been suggested, but it has not been possible to confirm this. Preoperative differentiation using imaging tests of HA which have become malignant and those which have not is very difficult.7 Serum α-fetoprotein is not helpful as it is usually normal.10,22
The real incidence of malignisation of HA has not been clearly defined and is placed between 5% and 18%.1,6,7,22 The systematic review of the literature on HA and malignisation undertaken in 2010 by Stoot et al. found an incidence of malignant transformation of 4.2% in all HA studied, and 4.5% in resected HA.13 This rate of malignant change is very similar to that of our series (3.7%). The average size of published cases of HA which have become malignant is large (10.5cm), but the relationship between the size of the tumour and traditionally accepted malignant change could not be confirmed statistically.13,28 The smallest published case of a tumour which became malignant only measured 4cm, but only 4.4% of HA which become malignant measure less than 5cm.10,13
It would be of the greatest interest to be able to define which HA are at a higher risk of malignisation in order to be able to remove them immediately.2,7 The following have been cited as possible factors: the consumption of androgens or anabolic steroids, male gender, patients with glycogenosis, patients with polyposis, the presence of dysplasia in percutaneous biopsy, the expression of high levels of β-catenin, non-steatosic HA and those larger than 5cm.10,2,6,13,23,24
Rupture or haemorrhage is another serious complication of HA.3,4,6 A systematic review of the literature undertaken in 2012 set the incidence of haemorrhage at 27% and of intraperitoneal rupture at 17.5%.3 Our series presents an incidence of 10%. Previous ingestion of OC and a size larger than 7cm have been associated with rupture, although the rupture of an HA of 1cm2 has been described. No relationship has been observed between the number of HA and the appearance of this complication.3 In our study, the patients who presented rupture and haemorrhage had larger tumours (8.4 vs 11.9cm) and a greater percentage of adenomatosis (44% vs 7%). Rupture is more common in inflammatory or telangiectasic HA.
When an HA ruptures, it is a serious situation which requires urgent action. Patients are usually (70% of cases) unaware that they have an HA, and suffer extreme abdominal pain and haemodynamic instability.4,6 After initial stabilisation, transarterial embolisation of the HA has been shown to be a very valuable therapeutic tool, as it avoids emergency surgery which has very high morbidity, 81% in our series, and embolisation can even avoid resection in certain cases (small tumours, patients with comorbidities…).0,5,25,28,29 But the major disadvantage is that embolisation is not always available, or, in a few cases, is ineffective. In these cases, emergency laparotomy is the treatment of choice, which includes resection or packing and excision at a second stage.7,25,28,29 Of our 8 cases which started with haemorrhage or rupture, 7 were operated on, and only one was embolised and underwent elective surgery. We believe that this is due to a combination of reasons: the low availability of emergency embolisation in Spanish hospitals and because, in the earlier cases, it was not yet considered.
Hepatic adenomatosis is a very uncommon entity of unknown aetiology and there are barely a hundred published cases. It was described by Flejou in 1985 and defined as the presence of more than 10 adenomas in a healthy liver parenchyma; it occurs more commonly in women.3–6,12,16–18,30 It has been suggested that glucose metabolism disorders, obesity, HNF1-α mutations and steatohepatitis can be aetiological factors.4,5,18 There are 2 forms: multifocal and massive.6,9 Eight of our patients presented the multifocal form and one the massive form. The appearance of complications (bleeding, malignant change…) or symptoms (abdominal pain) with adenomatosis seem to be more common than with single adenomas.5,6,16–18,30 There is controversy over their treatment, although currently, it is increasingly conservative for patients with adenomatosis.18 Treatment varies between monitoring and liver transplant, although resection of lesions greater than 5cm, especially if the β-catenin is positive, and monitoring of the other lesions is currently more common, as complete removal of all lesions is difficult because bilobar involvement is frequent.6,12,17,18,30 Our series includes 9 patients with adenomatosis who were treated by resection of the predominant lesions. One of the first patients was treated with a major hepatectomy and then transplant. Liver transplant is hardly ever used for this entity nowadays.1
Radiofrequency ablation and transarterial embolisation are new therapeutic strategies which are used but have not yet been validated in the treatment of HA.1,4,6,10,13,21,31 The indications which are most accepted for this technique involve central or multiple lesions which require complex hepatectomies, steatosic livers, patients who are not suitable for surgery or women who wish to become pregnant.6,31 Radiofrequency ablation is particularly useful in lesions of less than 4cm which should not be treated but simply monitored initially.6,31
Conflict of InterestThe authors have no conflict of interests to declare.
Please cite this article as: Ramia JM, Bernardo C, Valdivieso A, Dopazo C, Jover JM, Albiol MT, et al. Estudio multicéntrico sobre adenomas hepáticos. Cir Esp. 2014;92:120–125.