Introduction and aim. Excessive alcohol consumption is a public health concern worldwide and has been associated with high mortality rates. This study aimed to determine the prevalence of alcohol consumption and its influence on the prognosis of hospitalized cirrhotic patients in a tertiary care hospital.
Material and methods. We reviewed the medical records of all patients with hepatic cirrhosis admitted between January 2009 and December 2014, in a referral center for liver disease in southern Brazil. Data on clinical outcomes, associated conditions, infections, and mortality were collected and compared between alcoholic and nonalcoholic patients.
Results. The sample consisted of 388 patients; 259 (66.7%) were men. One hundred fifty-two (39.2%) were classified as heavy use of alcohol. Most alcoholic patients were men (n = 144; 94.7%). Mean age was 55.6 ± 8.9 years. Hepatic decompensations and infections were more prevalent in alcoholic patient. Spontaneous bacterial peritonitis and respiratory tract infection accounted for most of the infections. Excessive alcohol consumption was associated with mortality (P = 0.009) in multivariate analysis.
Conclusion. On the present study, the prevalence of heavy use of alcohol was high and associated with a poorer prognosis in hospitalized cirrhotic patients, increasing the risk of infection and death.
Excessive alcohol consumption is a public health concern worldwide. An estimated 4.9% of the world’s adult population suffer from alcohol use disorder.1 In the United States, alcohol abuse led to approximately 88,000 deaths each year.2 In a study in Brazil, 8.9% of males and 3.6% of females had diagnosis of alcohol use disorders.3
Excessive alcohol intake is a major risk factor for chronic liver disease and decompensation.4–6 In addition, patients with compensated cirrhosis may develop acute-on-chronic liver failure (ACLF) that is associated with a high mortality rate.7,8 Moreau, et al., in a multicenter study involving 1,343 patients showed that alcohol and infection were the main precipitant factor of ACLF.9
Alcoholic liver disease (ALD) accounts for 40% of all deaths from cirrhosis and is a major preventable cause of morbidity and mortality worldwide.10–12 In Brazil, a previous study in our center assess the national impact of liver diseases on hospital admissions and mortality and found that cirrhosis was the leading cause of hospitalization and death in patients with liver disease. Alcoholism was the second most common cause of cirrhosis in the country.13
Despite the importance of this issue, studies investigating the influence of alcohol abuse on the outcome and prognosis of patients admitted to hepatology wards are scarce. Therefore, the aim of the present study was to determine the prevalence of alcohol consumption among hospitalized cirrhotic patients in a tertiary care hospital and its influence on clinical outcomes and mortality.
Material and MethodsWe conducted a cross-sectional study with retrospective medical record review of all patients with liver cirrhosis who were admitted between January 2009 and December 2014 to the hepatology ward of Irmandade Santa Casa de Misericórdia, a tertiary care hospital and referral center for liver disease located in Porto Alegre, in southern Brazil.
For comparison purposes, patients were categorized according to their alcohol consumption as alcoholic (alcohol consumption of more than 20 g/day for women and 40 g/day for men) and nonalcoholic patients. Data on daily and weekly alcohol intake (dose/g) and type of beverage consumed (sugarcane liquor, beer, wine, vodka, brandy, and whiskey) were collected from alcoholic patients.
The following data were collected from all patients: sex and age, number of hospitalizations in the past 6 years, associated conditions, smoking status, diabetes mellitus, high blood pressure, HCV infection, human immunodeficiency virus (HIV) infection, clinical outcomes (ascites, hepatic encephalopathy [HE], upper gastrointestinal bleeding [UGIB], jaundice, hepatocellular carcinoma [HCC], alcoholic hepatitis, renal failure, and infection) and death. If present, infection was identified as spontaneous bacterial peritonitis (SBP), respiratory tract infection, urinary tract infection (UTI), positive blood cultures, and cutaneous or subcutaneous infections.
The study was approved by the institutional review board (IRB No. 1.351.101).
Continuous variables were expressed as mean (SD), and categorical variables as absolute frequencies and percentages. Mean values were compared between groups using Student’s t test, and percentages were compared using Pearson’s chi-square test. Multivariate Poisson regression was used to control for potential confounders. Prevalence ratios (PR) with 95% confidence intervals were used as effect measures. The level of significance was set at 5% (P ≤ 0.05), and all analyses were performed using SPSS, version 21.0.
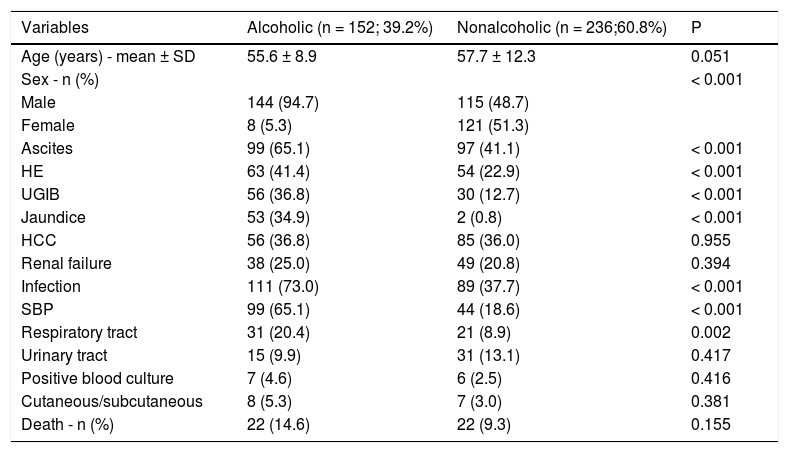
ResultsThe sample consisted of388 patients; 259 (66.7%) men and 129 (33.3%) women. The characteristics of the sample and their clinical outcomes, stratified based on alcohol consumption are shown table 1. Of the total sample, 152 (39.2%) patients were classified as alcoholic and 236 (60.8%) as nonalcoholic. The mean (SD) age of alcoholic patients was 55.6 ± 8.9 years. Most alcoholic patients were men (n = 144; 94.7%). Alcoholic patients were readmitted more frequently than nonalcoholic patients (P < 0.001).
Patient characteristics and clinical outcomes.
| Variables | Alcoholic (n = 152; 39.2%) | Nonalcoholic (n = 236;60.8%) | P |
|---|---|---|---|
| Age (years) - mean ± SD | 55.6 ± 8.9 | 57.7 ± 12.3 | 0.051 |
| Sex - n (%) | < 0.001 | ||
| Male | 144 (94.7) | 115 (48.7) | |
| Female | 8 (5.3) | 121 (51.3) | |
| Ascites | 99 (65.1) | 97 (41.1) | < 0.001 |
| HE | 63 (41.4) | 54 (22.9) | < 0.001 |
| UGIB | 56 (36.8) | 30 (12.7) | < 0.001 |
| Jaundice | 53 (34.9) | 2 (0.8) | < 0.001 |
| HCC | 56 (36.8) | 85 (36.0) | 0.955 |
| Renal failure | 38 (25.0) | 49 (20.8) | 0.394 |
| Infection | 111 (73.0) | 89 (37.7) | < 0.001 |
| SBP | 99 (65.1) | 44 (18.6) | < 0.001 |
| Respiratory tract | 31 (20.4) | 21 (8.9) | 0.002 |
| Urinary tract | 15 (9.9) | 31 (13.1) | 0.417 |
| Positive blood culture | 7 (4.6) | 6 (2.5) | 0.416 |
| Cutaneous/subcutaneous | 8 (5.3) | 7 (3.0) | 0.381 |
| Death - n (%) | 22 (14.6) | 22 (9.3) | 0.155 |
HCC: hepatocellular carcinoma. HE: hepatic encephalopathy. SBP: spontaneous bacterial peritonitis. UGIB: upper gastrointestinal bleeding.
The alcoholic group consumed a mean of 187 g of alcohol per day (SD, 165 g; median, 140 g; range, 26 to 992 g). Most of these patients consumed sugarcane liquor (57.0%), followed by beer (31.0%), vodka (1.6%), wine (5.8%), brandy (3.0%), and whiskey (1.6%).
Regarding associated conditions, smoking (26.0% vs. 3.7%; p < 0.001) and high blood pressure (37.4% vs. 24.1%; p = 0.003) were more prevalent in alcoholic than in nonalcoholic patients. Diabetes mellitus, HCV infection and HIV infection did not differ statistically between groups.
Twenty-two alcoholic patients had alcoholic hepatitis (17.3%). Complications associated with cirrhosis were also more frequent in alcoholic patients. Ascites was the most common complication followed by HE, UGIB and HCC. Infections occurred in 200 (51.5%) patients and were also more frequent in alcoholic; however, the analysis of infection sites revealed that only respiratory tract infections and SBP were significantly more frequent in the alcoholic group.
The overall in-hospital mortality rate was 11.3% (44 of 388 patients). Twenty-two patients (14.6%) died in the alcoholic group, while 22 patients (9.3%) died in the nonalcoholic group (P = 0.155). Among alcoholic patients, 11 died of sepsis (50.0%), 5 of advanced HCC (22.8%), 2 of UGIB (9.0%), 3 of liver failure (13.7%), and 1 of acute pulmonary edema (4.5%). Among alcoholic patients who died, 3 had alcoholic hepatitis (2 died of liver failure and 1 of sepsis).
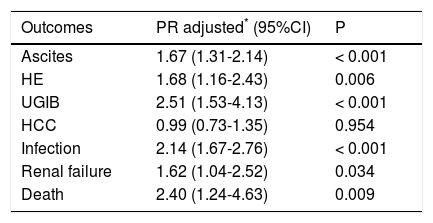
Table 2 shows the results of the multivariate Poisson regression analysis for the influence of alcohol consumption on outcomes. The regression model was adjusted for sex, age, smoking and HCV infection. All outcomes (ascites, HE, UGIB, infection, renal failure and death) except HCC were significantly associated with alcohol consumption. Alcoholic patients were 1.67 times more likely to develop ascites (P < 0.001), 1.68 times more likely to develop HE (P = 0.006), 2.51 times more likely to have UGIB (P < 0.001), 2.14 times more likely to develop infections (P < 0.001), 1.62 times more likely to develop renal failure (P = 0.034), and 2.40 times more likely to die (P = 0.009). We also did a multivariate Poisson regression analysis to investigate the influence of HCV infection on clinical outcomes in the alcoholic cirrhotic patients, and demonstrated that only HCC was different between the groups, being more frequent in alcoholic patients with HCV (47.9% vs. 19.0%); OR 2.47 (1.39-4.39); P = 0.002.
Results of multivariate Poisson regression analysis for the influence of alcohol consumption on clinical outcomes.
| Outcomes | PR adjusted* (95%CI) | P |
|---|---|---|
| Ascites | 1.67 (1.31-2.14) | < 0.001 |
| HE | 1.68 (1.16-2.43) | 0.006 |
| UGIB | 2.51 (1.53-4.13) | < 0.001 |
| HCC | 0.99 (0.73-1.35) | 0.954 |
| Infection | 2.14 (1.67-2.76) | < 0.001 |
| Renal failure | 1.62 (1.04-2.52) | 0.034 |
| Death | 2.40 (1.24-4.63) | 0.009 |
HCC: hepatocellular carcinoma. HE: hepatic encephalopathy. PR: prevalence ratio. UGIB: upper gastrointestinal bleeding.
Excessive alcohol consumption is a major cause of liver disease worldwide.5,6,14 In a recent European study, alcoholic cirrhosis was the leading cause of hospitalization and liver cirrhosis was the main cause of death in patients with liver disease.15 Similarly, in a population-based study of 16 countries across the Americas, alcohol was an important risk factor for mortality.16 In the present study, of a total sample of 388 patients, 39.2% were alcoholic. This rate is probably a reflection of our selected sample, which consisted of patients admitted to a referral center for liver disease. In a cohort study performed at 21 centers in Spain, of a total sample of 1,039 patients hospitalized in internal medicine wards, 12% had a history of alcohol abuse.17 The excessive alcohol consumption associated with males is consistent with the literature.4,18–20
Regarding hospital readmission rates, Berman, et al.21 analyzed 554 admissions of patients with advanced liver disease and found, similarly to the present study, that the hospitalization rate was higher in patients with ALD than in nonalcoholic patients.
In the present study, mean daily alcohol consumption was 187 g in alcoholic patients and may have contributed to the higher prevalence of liver cirrhosis decompensation, including mortality, in this group of patients. Studies showed that alcohol consumption is associated with an increased risk of death due to liver cirrhosis. The most commonly consumed alcoholic beverage in the present study was sugarcane liquor (“cachaça”). Due to its low production cost, sugarcane liquor is the most widely produced liquor in Brazil, thus being consumed in large quantities by individuals of low socioeconomic status.22–24
In addition to chronic liver disease, alcohol consumption may also cause alcoholic hepatitis, a severe complication with an acute onset and poor prognosis.25–27 In the present study, 22 patients (13.7%) had alcoholic hepatitis.
Pessione, et al., evaluating 5-year predictive factors in hospitalized patients with excessive alcohol intake and cirrhosis, found an association between smoking and mortality.29 In the present study, smoking was more common in alcoholic patients, which is consistent with previous studies.19,28 Mortality remained associated with excessive alcohol consumption even after controlling for smoking in the multivariate analysis. Excessive alcohol consumption was also associated with high blood pressure in hospitalized patients. A similar association was also reported in the literatura.30
The prevalence of HCV infection is higher in alcoholic patients than in the general population, ranging from 2 to 51%.31–34 In a systematic review, Veleiro, et al. reported a prevalence of 16.32% of HCV infection in patients with excessive alcohol use,33 while, in the general population, the prevalence was approximately 2%.32,34–36 Alcohol has a negative impact on the progression of chronic HCV infection due to a synergistic effect that increases viral replication and alters the immune response.34,37 In this study the occurrence of HCV infection was similar between groups.
In the present study, we only considered patients with liver cirrhosis. The literature shows that individuals who consume more than 25 g of alcohol per day have an increased relative risk of cirrosis.38 The risk of developing alcoholic cirrhosis was higher in daily drinkers than in those drinking 2-4 days/week.39
Portal hypertension and its manifestations (ascites, HE, and UGIB) are common complications of liver cirrosis.40,41 In the present study, ascites, HE and UGIB were more frequent in alcoholic patients. Jepsen, et al. examined the clinical course of alcoholic cirrhosis in 466 patients; as in the present study, ascites was the most common complication of cirrhosis.42
HCC is another common complication of liver cirrosis.43–47 Mancebo, et al., investigating 450 patients with alcoholic cirrhosis reported an annual incidence of HCC of 2.6%.46 Alcohol consumption and HCV infection have a synergistic effect on the predisposition to HCC.34,37 In the present study, 141 patients (36.3%) had a diagnosis of HCC however its occurrence was similar between alcoholics and non-alcoholics. On the other hand, when we analyzed only the alcoholic cirrhotic patients, we demonstrated that the presence of HCV in this population was associated with HCC.
Despite recent advances in the treatment of cirrhosis, bacterial infections remain an important complication that affects approximately 30% of patients, leading to increased mortality. The mechanism of infection is multifactorial and the incidence of resistant bacteria has increased, especially in hospitalized patients.48,49 In the present study, the overall infection rate was 51.5%. Infections were more frequent in alcoholic (73.0%) than in nonalcoholic (37.7%) patients, and the most frequent infections are SBP, respiratory tract infections, and UTI, which is consistent with previous studies.48,49
In the present study, the overall mortality rate was 11.3% and excessive alcohol use increased the risk of death by 2.4 times. Regarding mortality in Brazil, Garcia, et al. reviewed the data from the Mortality Information System of the Brazilian Ministry of Health between 2010 and 2012 and identified 55,380 deaths attributable to alcohol-related conditions. Liver disease was the main cause of death (55.3%) and the overall mortality rate was 8.7 times higher in males than in females.50
ConclusionThis study showed that alcohol consumption was a frequent finding among patients admitted to a referral center for liver disease. Excessive alcohol use was also a predictor of severe liver disease and its complications, increasing the risk of infection and death in hospitalized patients.
Abbreviations- •
ACLF: acute-on-chronic liver failure.
- •
ALD: alcoholic liver disease.
- •
HCV: hepatitis C virus.
- •
HCC: hepatocellular carcinoma.
- •
HE: hepatic encephalopathy.
- •
HIV: human immunodeficiency virus.
- •
SBP: spontaneous bacterial peritonitis.
- •
UGIB: upper gastrointestinal bleeding.
- •
UTI: urinary tract infection
The authors have no financial relationships to this article to disclose.
Conflict of InterestThe authors have no conflicts of interest to disclose.