Post-COVID-19 olfactory dysfunction persists in many patients and significantly affects quality of life. The primary objective was to evaluate olfactory function using psychophysical tests (BAST-24) and quality-of-life questionnaires. Secondary objectives included analysing correlations between questionnaires and the relationship between subjective and psychophysical measures.
Materials and methodsA prospective study was conducted including 86 adults with olfactory dysfunction ≥ 6 months after SARS-CoV-2 infection. Clinical and demographic data and scores from the Visual Analog Scale for smell (VAS-smell) and the SNOT-22, svQOD-NS, and QVOLF questionnaires were collected. Olfactory function was assessed using the BAST-24 (detection, identification, and olfactory memory). Descriptive statistics, Spearman’s correlation coefficient, and the Mann-Whitney U test were used (p < 0.05).
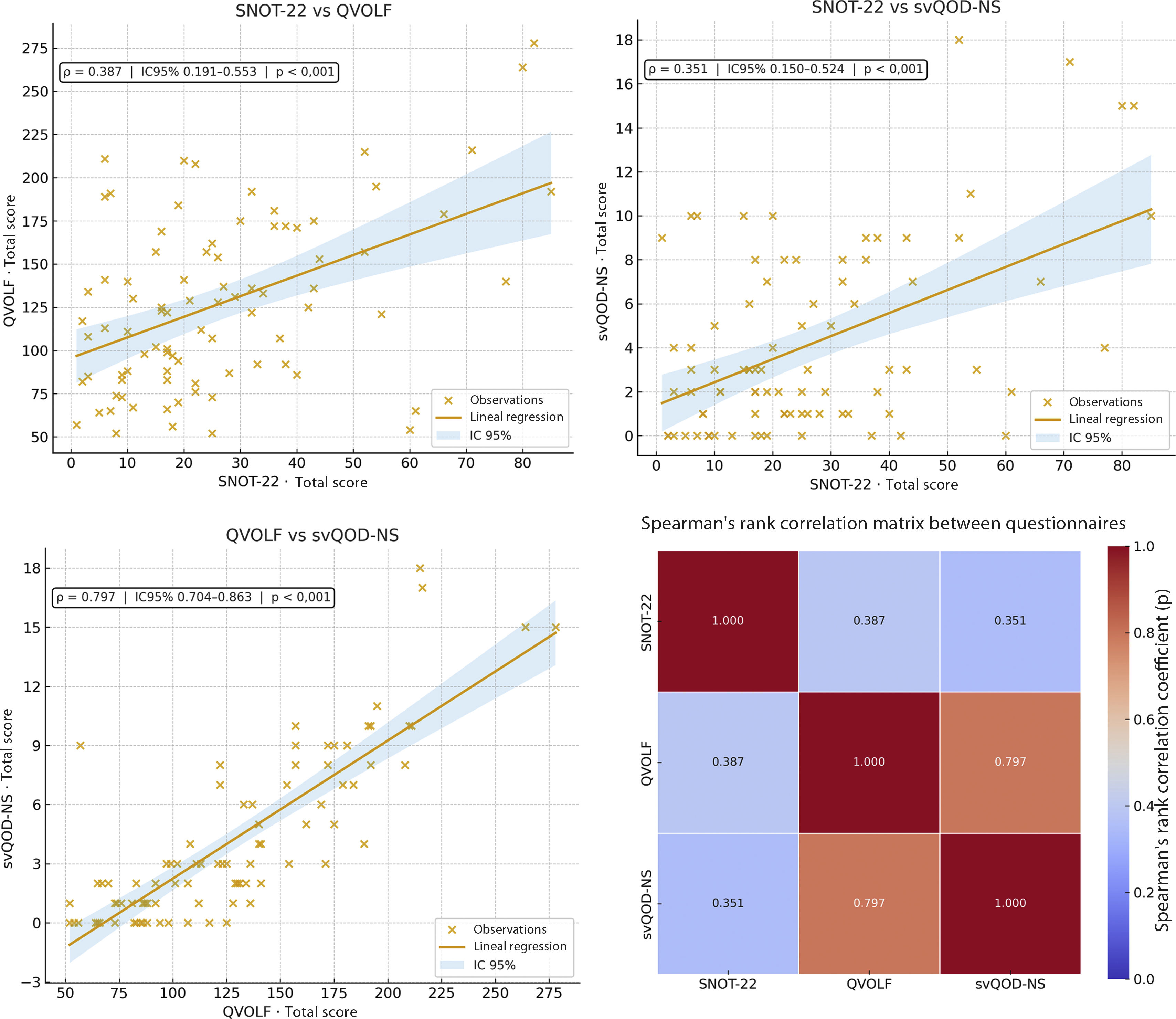
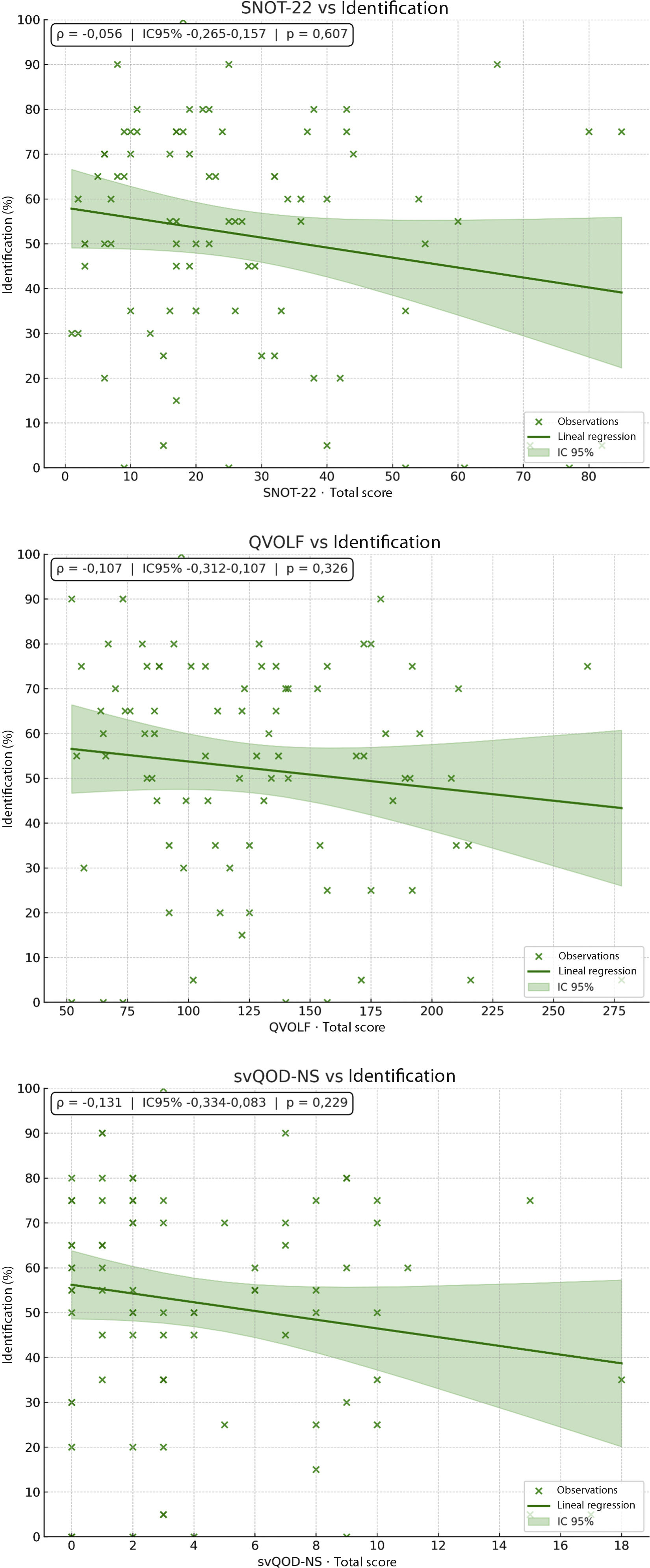
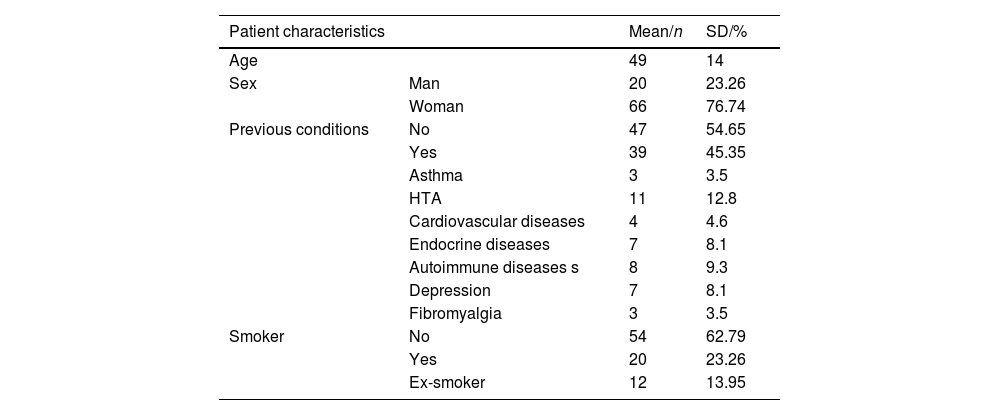
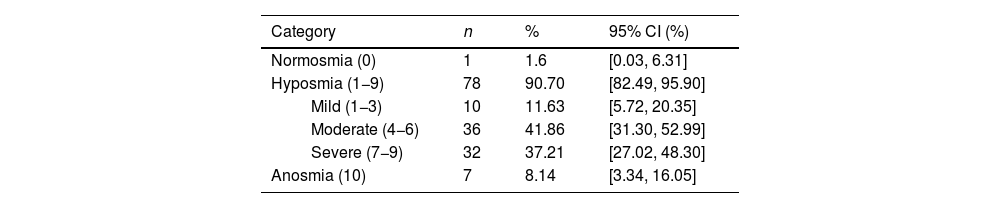
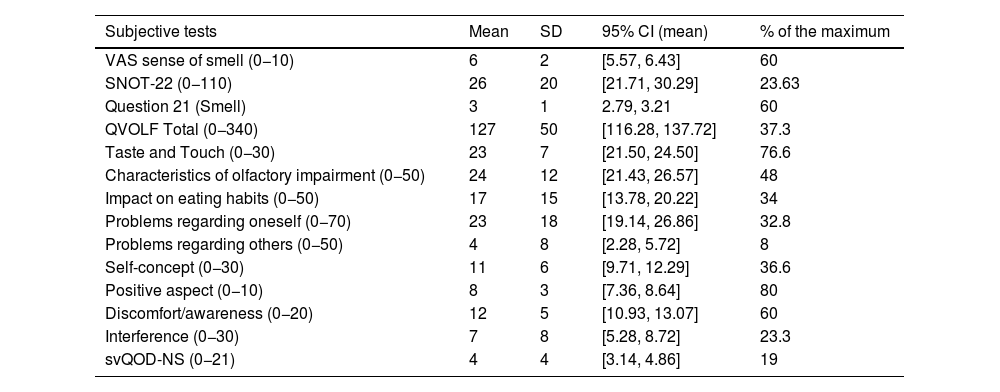
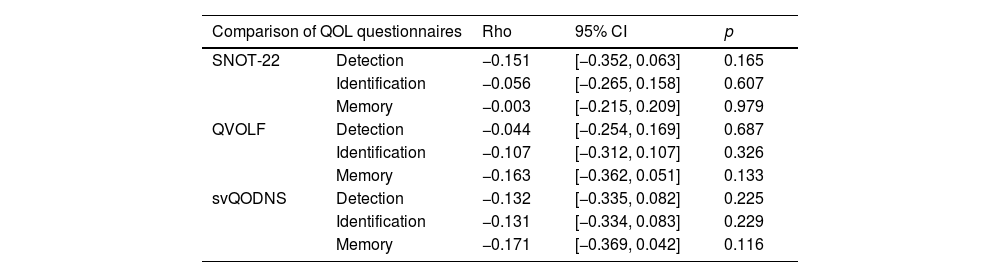
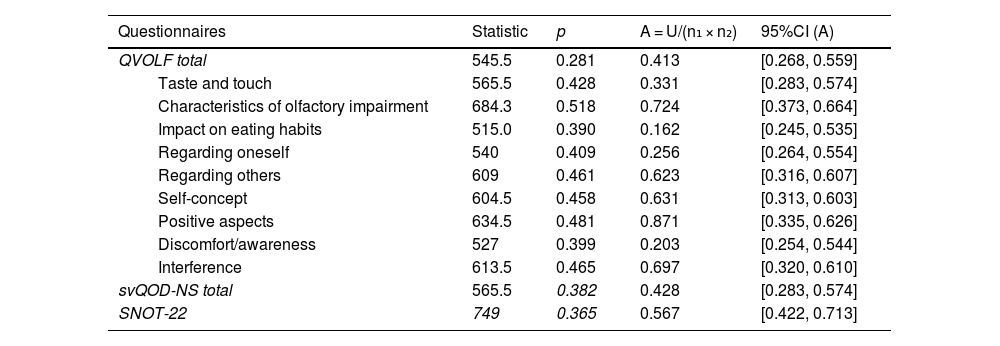
ResultsMean age was 49 years (SD = 14); 76.7% were women. According to the VAS, 90.7% had hyposmia and 8.1% anosmia. Mean scores (SD) were: VAS 6 (2), SNOT-22 26 (20), svQOD-NS 4 (4), and QVOLF 127 (50), suggesting a moderate impact on quality of life. BAST-24 scores: detection 91% (22), identification 52% (25), and memory 35% (20). QVOLF and svQOD-NS showed a strong correlation (ρ = 0.797; p < 0.001), while SNOT-22 correlated moderately with both. No significant correlations were found between any questionnaire and BAST-24.
ConclusionsQVOLF and svQOD-NS questionnaires are more sensitive than SNOT-22 for assessing olfactory dysfunction. The lack of correlation with psychophysical tests highlights the need to combine both approaches fora comprehensive evaluation.
La disfunción olfativa post-COVID-19 persiste en muchos pacientes y afecta significativamente la calidad de vida. El objetivo principal fue evaluar la función olfativa con pruebas psicofísicas (BAST-24) y cuestionarios de calidad de vida. Como objetivos secundarios, se analizó la correlación entre los cuestionarios y explorar la relación entre medidas subjetivas y psicofísicas.
Material y métodosSe realizó un estudio prospectivo en 86 adultos con disfunción olfativa ≥6 meses tras infección por SARS-CoV-2. Se recogieron datos clínico-demográficos, y puntuaciones de la Escala Visual Analógica para el olfato (EVA-olfato) y de los cuestionarios de calidad de vida SNOT-22, svQOD-NS y QVOLF. La función olfativa se evaluó con elBAST-24 (detección, identificación y memoria olfativa). Se usaron estadísticos descriptivos, coeficiente de correlación de Spearman y prueba de Mann-Whitney U (p < 0,05).
ResultadosLa edad media fue 49 años (DE = 14; IC95% 46,0–52,0); el 76,7% eran mujeres. Según la EVA, el 90,7% presentó hiposmia y el 8,1% anosmia. Las puntuaciones medias (DE) fueron: EVA 6 (2), SNOT-22 26 (20), svQOD-NS 4 (4) y QVOLF 127 (50), sugiriendo impacto moderado en la calidad de vida. BAST-24: detección 91%, (22) identificación 52% (25) y memoria 35% (20). QVOLF y svQOD-NS mostraron fuerte correlación (ρ = 0,797; p < 0,001) mientras que SNOT-22 correlacionó moderadamente con ambos. No hubo correlaciones significativas entre cuestionarios y BAST-24.
ConclusionesQVOLF y svQOD-NS son más sensibles que SNOT-22 para evaluarla disfunción olfativa. La ausencia de correlación con pruebas psicofísicas resalta la necesidad de combinar ambas para una evaluación integral.