Myofibroblastoma (MFB) is a rare spindle stromal tumour of the breast that predominates in 60–80 years-old adult males. Its imaging features are nonspecific, leading to misdiagnosis. Thus, core biopsy is needed for definitive diagnosis.
Macroscopically, MFB is usually a well-circumscribed, firm and rubbery, unencapsulated, pale white to grey round mass. Microscopically, it consists on spindle cells arranged in haphazardly intersecting fascicles or clusters, thick hyalinized collagen bundles and low mitotic activity with a lack of myoepithelial component and necrosis. Immunohistochemistry shows consistently positive immunoreactivity to vimentin and CD34, while expression of desmin, SMA, bcl-2 and CD99 varies. Oestrogen, progesterone and androgen receptors are usually expressed. They are constantly negative to cytokeratins, EMA, S100 protein, HMB-45 and c-kit (CD117). These differentiate them from fibroadenoma, phyllodes tumour, round pattern gynecomastia, carcinoma and sarcoma, since they present infiltrative growth and are negative to CD34.
Wide local excision is curative, with no need of sentinel lymph node biopsy, since local recurrence is extremely low and has been reported to be less than 1.5%. No distant metastases have been described on the literature.
We report a rare case MFB on a 73-year-old male attended at our institution presenting with a nodule on the right breast.
El miofibroblastoma (MFB) es un tumour estromal de células fusiformes que aparece en varones de 60-80 años. Las características radiológicas son inespecíficas, por lo que es necesaria la realización de biopsia para el diagnóstico definitivo. Macroscópicamente se trata de una lesión bien circunscrita, firme, no encapsulada. Microscópicamente consiste en células fusiformes organizadas en fascículos entremezclados con bandas de colágeno hialino, con baja actividad mitótica y ausencia de componente mioepitelial y necrosis. La inmunohistoquímica muestra la expresión constante de vimentina y CD34, con expresión variable de desmina, AML, bcl-2 y CD99. Los receptores de estrógenos, progesterona y andrógenos normalmente son positivos, mientras que la expresión de citoqueratinas, EMA, S100, HMB-45 y c-kit (CD117) es negativa. Estas características lo diferencian del fibroadenoma, tumour filodes, ginecomastia, carcinoma y sarcoma, ya que la mayoría de ellos se caracterizan por ser negativos para CD34 y presentar crecimiento infiltrativo. La tumorectomía es considerada curativa, sin necesidad de realizar biopsia selectiva de ganglio centinela, dado que la recurrencia local es baja (menos del 1,5%). No se ha descrito la presencia de metástasis a distancia en la literatura. Presentamos el caso de MFB en un varón de 73 años que debutó con un nódulo en la mama derecha.
Breast myofibroblastoma (MFB) is a rare benign mesenchymal tumour, derived from mammary stromal fibro-myofibroblasts, described in 1987 by Weiss et al., as a benign breast tumour contained in a pseudocapsule with spindle cells and abundant collagen.1 Characteristically MFB are positive to vimentin, desmin and CD34, whereas they are consistently negative to cytokeratins, EMA, S100, HMB-45 and c-kit (CD117). Classically these tumours have been described in males on the 6–8th decades, representing less than 1% of breast tumours.2,3 Nevertheless, currently is believed that MFB mainly occurs in older men and postmenopausal women.2 Most cases present as movable, nontender palpable masses, which may demonstrate slow, steady growth over months to years.4
Case reportWe report the case of a 73-year-old male presenting with a palpable mass on the right breast. He had a history of atrial fibrillation and hypertension, without family history of breast cancer. He presented with a nodule on the upper outer quadrant measuring 2cm×2cm, well circumscribed, and freely movable. He had no skin or nipple retraction, and no palpable axillar lymph nodes.
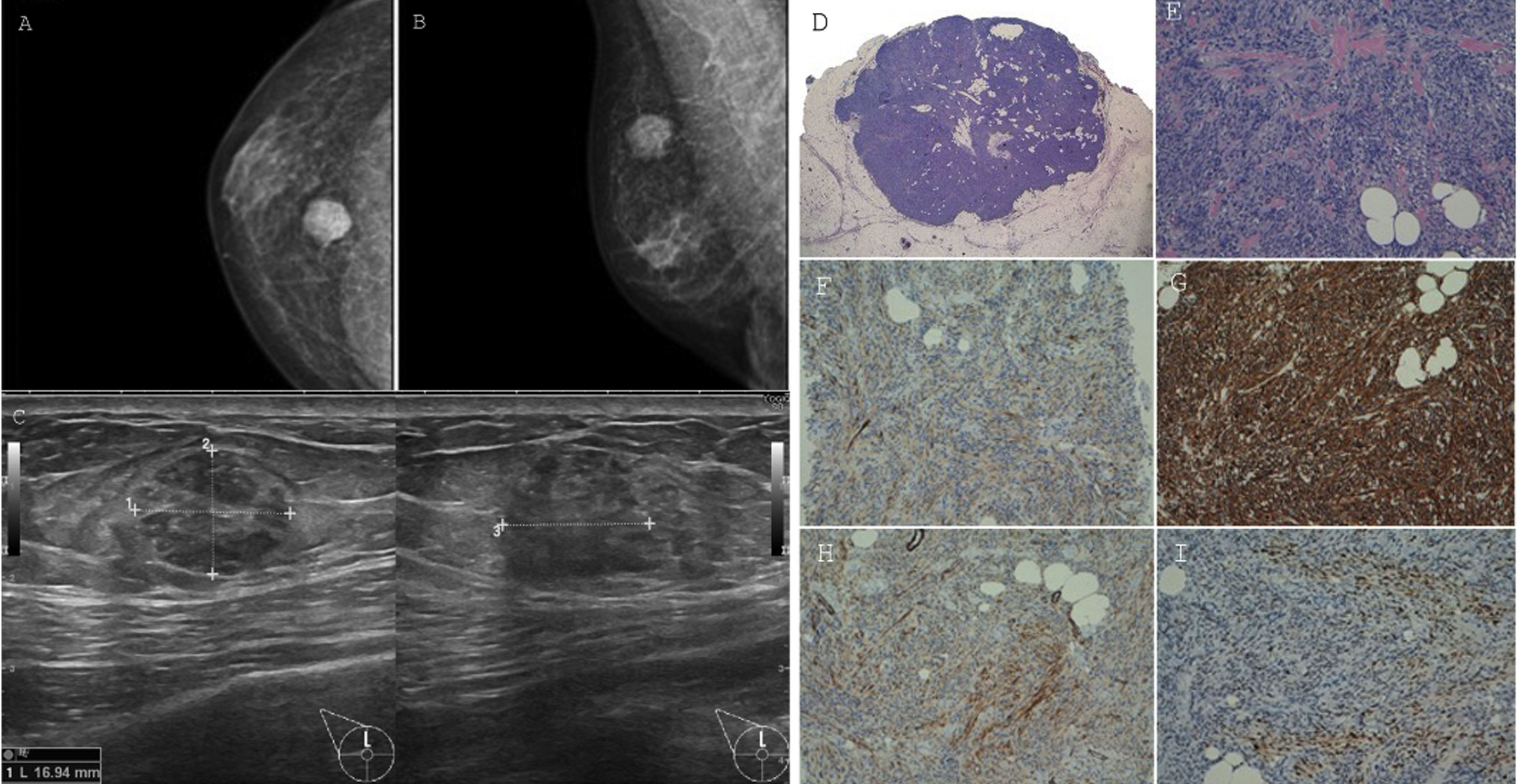
Mammography (Fig. 1) revealed a solid, lobulated nodular lesion of suspicious appearance (BIRADS 4B/C). Neither microcalcifications nor pathological axillar lymph nodes were described.
Right breast mammography (A, B) and ultrasound (C). Myofibroblastoma. Panoramic view showing a circumscribed non encapsulated tumour (D). High-power view demonstrates short fascicles of cytologically bland spindle cells admixed with bands of hyalinized collagen and entrapped mature adipose tissue (E). Inmunohistochemical study. This lesion shows patched expression of smooth muscle actin and desmin (F, G). CD34 is strongly positive (H). Progesterone receptor is also positive (I).
Ultrasound (US) (Fig. 1) confirmed the presence of a heterogeneous nodule on the UOQ that measured 17x13x16mm with ill-defined margins (BIRADS 4C). No pathological axillary lymphadenopathies were found.
According to the US results a core biopsy was performed, showing a spindle-cell lesion highly suggestive of MFB. Immunohistochemistry showed a positive expression of CD34, smooth muscle actin, desmin, and oestrogen and progesterone receptors, whereas cytokeratin 5/6 and p63 was negative.
Considering the radiological examination and the benign pathological findings a lumpectomy was performed. The nodule was well defined and did not infiltrate the mammary gland nor the pectoral muscle. It consisted on a well demarcated, firm, reddish-brown mass.
Microscopically, a well-circumscribed, non-encapsulated, benign mesenquimal neoplasm was observed (Fig. 1). The spindle cells comprising the lesion were uniform in appearance, with large bland nuclei and no evident nucleoli. These cells were arranged as variable size fascicles oriented in different directions and admixed with bands of hyalinized, brightly eosinophylic collagen. There were also some foci of entrapped mature adipose tissue. Mitotic figures and necrosis were absent.
This lesion showed expression of CD34 (expressed on haematopoietic cells, or in mesenchymal stem cells), actin and desmin (patched). Oestrogen, progesterone and androgen receptors were positive, distinguishing it from other with similar immunostaining. CK5/6, S-100 (excluding neuroendocrine origin), and STAT-6 were negative. Proliferation index (ki-67) was 5%.
DiscussionMFB belongs to the family of benign spindle stromal tumours of the breast. MFB predominates in 60–80 years-old males.1,2 MFB does not usually exceed 4cm, although it can grow up to 16cm.4 Several lesions have been identified in patients presenting with gynecomastia, but it has also been associated with postinjury or postsurgical sites, although only few such cases have been reported.5,6
MFB presents as unilateral, firm, mobile, painless mass with slow, steady growth. Imaging features of MFB are nonspecific. Mammography shows a well-circumscribed, round to oval, dense mass with rare course calcifications.5 US findings may vary from homogeneously hypoecoic to heterogeneous mass with or without posterior shadowing. Computed tomography typically shows a circumscribed soft-tissue density mass. MRI demonstrates T1 hypointensity to isointensity with positive early enhancement and nonenhancing septations, whereas they are typically T2 hyperintense.7,8 According to these nonspecific-imaging features, tissue sampling for pathologic diagnosis is needed.
Fine needle aspiration cytology (FNAC) might misdiagnose MFB, as it can be mistaken with gynecomastia, phyllodes tumour or malignancy, while core biopsy increases the chance of a correct preoperative diagnosis.2 Nevertheless, surgical excision retains a pivotal diagnostic and therapeutic role, since the pleomorphism of MFB can lead to diagnostic pitfalls.6
Macroscopically, MFB is usually a well-circumscribed, firm and rubbery, unencapsulated, round to oval mass. The cut surface reveals a solid lesion with a smooth or lobulated surface, pale white to grey, with a variably whirling appearance. Cystic degeneration, necrosis and haemorrhage are absent.2,9
Microscopically, there has been described several histologic variants, such as cellular, infiltative, epithelioid, deciduoid-like, collagenized-fibrous, lipomatous and myxoid. Their recognition is important to avoid confusion with other benign or malignant breast tumours.2 Typically, spindle cells are arranged in short haphazardly intersecting fascicles or clusters, interrupted by collagen bundles, with a lack of ducts and lobules. Essential elements for diagnosis are the presence of thick hyalinized collagen bundles and low mitotic activity as well as the lack of myoepithelial component and necrosis.6,7
Immunohistochemistry is crucial for the diagnosis of MFB, showing consistently positive immunoreactivity to vimentin and CD34, while the expression of desmin, SMA, bcl-2 and CD99 varies. Oestrogen and progesterone receptors are usually expressed, as well as androgen receptors. They are constantly negative to cytokeratins, EMA, S100 protein, HMB-45 and c-kit (CD117).2,3,6,9
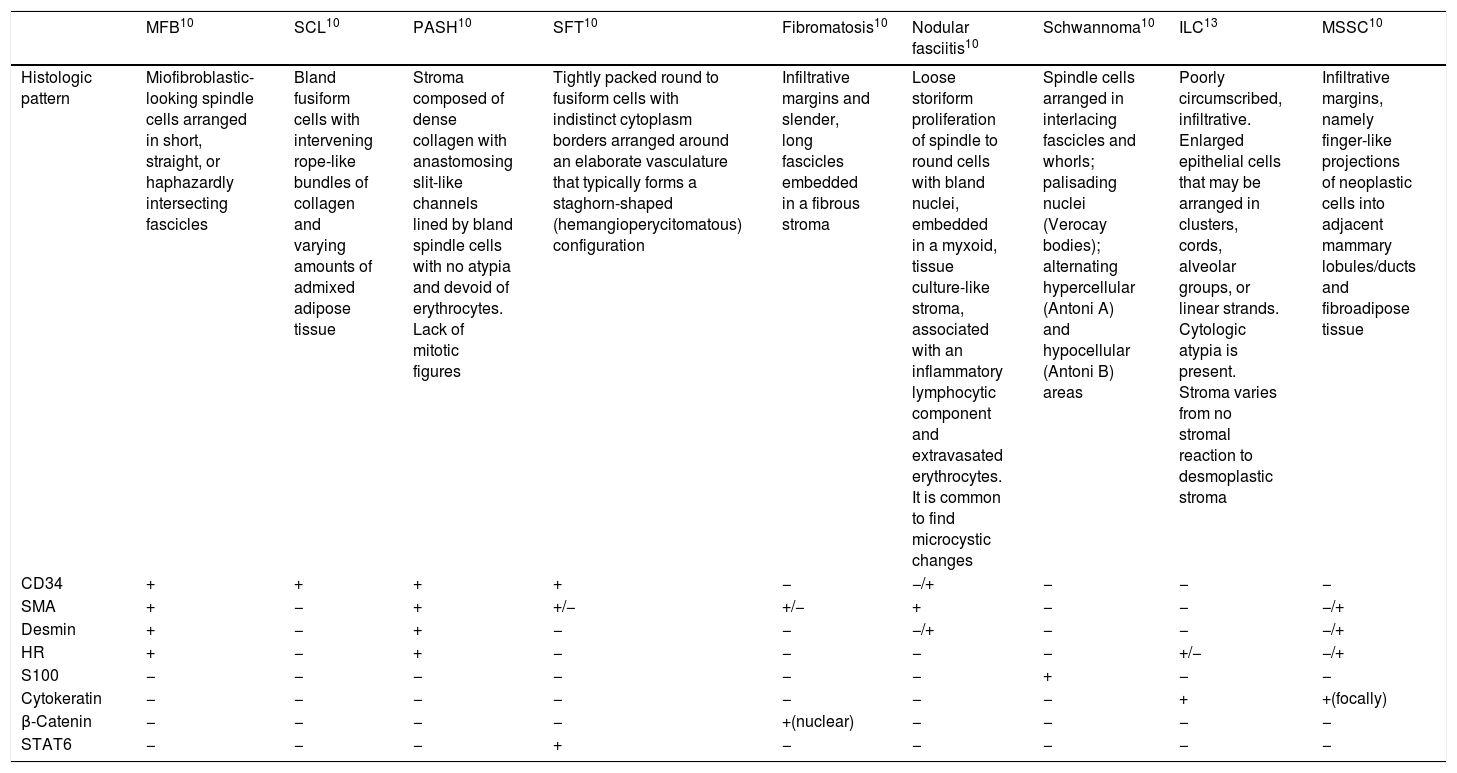
MFB should be distinguished from benign lesions, including fibromatosis, nodular fasciitis, spindle cell lipoma, pseudoangiomatous stromal hyperplasia (PASH), solitary fibrous tumour and schwannoma. Also, several malignant lesions should be included in the differential diagnosis such as invasive lobular carcinoma, metaplastic spindle cell carcinoma and low-grade sarcomas (Table 1).10 It might be challenging, especially if one is facing unusual variants. Epithelioid MFB may be confused with invasive lobular carcinoma due to its growth pattern in single cell files. Deciduoic-like MFB may mimic apocrine carcinoma, whereas lipomatous variant is usually mistaken with desmoid-like fibromatosis, fibromatosis-like low-grade carcinoma or low-grade sacoma.2 Mixoid MFB can be misdiagnosed with other malignant mixoid lesions such as mixofibrosarcoma or mixoid liposarcoma.3 Immunochemistry plays a crucial role on the diagnosis of MFB, since most of these mesenchimal/soft tissue lesions, unlike MFB, are characterized by infiltrative growth and are negative to CD34.11
Differential diagnosis of MFB. Histologic features and immunohistochemistry.
| MFB10 | SCL10 | PASH10 | SFT10 | Fibromatosis10 | Nodular fasciitis10 | Schwannoma10 | ILC13 | MSSC10 | |
|---|---|---|---|---|---|---|---|---|---|
| Histologic pattern | Miofibroblastic-looking spindle cells arranged in short, straight, or haphazardly intersecting fascicles | Bland fusiform cells with intervening rope-like bundles of collagen and varying amounts of admixed adipose tissue | Stroma composed of dense collagen with anastomosing slit-like channels lined by bland spindle cells with no atypia and devoid of erythrocytes. Lack of mitotic figures | Tightly packed round to fusiform cells with indistinct cytoplasm borders arranged around an elaborate vasculature that typically forms a staghorn-shaped (hemangioperycitomatous) configuration | Infiltrative margins and slender, long fascicles embedded in a fibrous stroma | Loose storiform proliferation of spindle to round cells with bland nuclei, embedded in a myxoid, tissue culture-like stroma, associated with an inflammatory lymphocytic component and extravasated erythrocytes. It is common to find microcystic changes | Spindle cells arranged in interlacing fascicles and whorls; palisading nuclei (Verocay bodies); alternating hypercellular (Antoni A) and hypocellular (Antoni B) areas | Poorly circumscribed, infiltrative. Enlarged epithelial cells that may be arranged in clusters, cords, alveolar groups, or linear strands. Cytologic atypia is present. Stroma varies from no stromal reaction to desmoplastic stroma | Infiltrative margins, namely finger-like projections of neoplastic cells into adjacent mammary lobules/ducts and fibroadipose tissue |
| CD34 | + | + | + | + | − | −/+ | − | − | − |
| SMA | + | − | + | +/− | +/− | + | − | − | −/+ |
| Desmin | + | − | + | − | − | −/+ | − | − | −/+ |
| HR | + | − | + | − | − | − | − | +/− | −/+ |
| S100 | − | − | − | − | − | − | + | − | − |
| Cytokeratin | − | − | − | − | − | − | − | + | +(focally) |
| β-Catenin | − | − | − | − | +(nuclear) | − | − | − | − |
| STAT6 | − | − | − | + | − | − | − | − | − |
MFB and SCL: loss of nuclear RB1 protein expression.
HR: hormone receptors; ILC: invasive lobular carcinoma; MFB: myofibroblastoma; MSSC: metaplastic spindle cell carcinoma; PASH: pseudoangiomatous stromal hyperplasia; SCL: spindle cell lipoma; SFT: solitary fibrous tumour.
MFB is managed with wide local excision, without need of sentinel lymph node biopsy. Mastectomy may also be advocated either cases of male patients with coexisting gynecomastia or in females with large tumours.6,9 Wide local excision is considered curative, since local recurrence has only been reported to be less than 1.5% after 20-year follow up.9,12 No distant metastases have been described on the literature.
Confidentiality of dataThe authors declare that they have followed the protocols of their Centre on the publication of patient data.
FundingThe authors declare that they did not receive any funding for the elaboration of this paper.
Conflict of interestSantana, González González, Bueno, García Chiloeches, Vaello, Muñoz Muñoz, Rivas, Cabañas and Fernández Cebrián, state that there are no conflicts of interest to disclose.