Urinary incontinence remains one of the most significant complications of radical prostatectomy, known as post-prostatectomy incontinence (PPI), can have profound effects on quality of life. The correct diagnosis is critical, and the urodynamic cause of incontinence established as either stress urinary incontinence or detrusor overactivity. Patient evaluation should also include the use of quality of life questionnaires to assess severity of symptoms and a quantitative pad weight or pad usage assessment. Treatment regimes should incorporate conservative measures pelvic floor exercises, and then failing this, a discussion of the more invasive therapies, including bulking injections, the artificial urinary sphincter or the male suburethral sling. All of these options should be discussed carefully with the patient, including success rates of outcomes and potential adverse effects of treatment.
Urinary incontinence remains one of the most significant complications of radical prostatectomy, and is known as post-prostatectomy incontinence (PPI). This clearly has negative quality of life implications for men, and is noted to be a major barrier to social recreation, physical activity and subsequent mental health affects including anxiety and depression (1).
The incidence and severity of the resultant incontinence varies, with no fixed definition. The definition of continence, tends to describe those wearing 1 or less pads over 24 hours. Incontinence can be based on quantity of pad use, to various scores using patient reported outcome measures (PROMs) such as the International Consultation on Urinary Incontinence Short Form series (ICIQ-UI SF) or on Male Lower Urinary Tract Symptoms Long Form (ICIQ-MLUTS LF) (2). Rates of PPI have been quoted to range from 6% to 69% according to the various definitions. Risk factors identified to increase this include age, higher body mass index as well as technical features of the surgery and surgeon experience (3). The Prostate Cancer Outcomes study, followed 1291 men after radical prostatectomy and at 18 months identified 8.4% of men were incontinent, although only 32% had ‘total urinary control’ (4). The rates of continence at best after open prostatectomy are quoted at upto 92%, and up to 96% after laparoscopic or robotic techniques (5).
The pathophysiology of urinary incontinence, tends to be due to intrinsic sphincter deficiency secondary to the loss of function of the external sphincter complex. The external sphincter is understood to have slow twitch striated fibres, which along with smooth muscle fibres provides baseline continence, whilst the fast twitch striated fibres which with rapid recruitment allow voluntary disruption of urinary flow (3). Therefore baseline continence can be impaired, and at times of striated muscle fibre exhaustion (for example during longer physical activity over the day) further incontinence episodes can also ensue.
DiagnosisThe first part of the assessment is with a full medical history and physical examination. Triggers for the urinary incontinence may include coughing, moving into an upright position, sports, and the absence of night-time continence.
Validated assessment tools ICIQ-UISF and ICIQ-MLUTS can be used to provide an objective assessment of the patient's symptom severity (6). A voiding diary should be completed, to document voided volumes and episodes of leakage, and whether there is any element of overactive bladder. The number of pads worn per day correlates well with the 24-pad weight (7), although the 24-pad weight is felt to be the most accurate form of assessment of involuntary urine loss (3). Arbitrary ranges for 24hours pad weight can be used such as <100g as mild, 100-300g as moderate and >300g as severe, however no official cut-off values have been defined.
Basic diagnostic tests should also include a urinalysis to exclude infection or haematuria, and an post void ultrasound of the bladder to determine any residual urine volumes (8). Urethroscopy is also recommended to assess for any evident bladder neck stenosis or urethral stricture that may be sustaining some level of continence, which once dilated may worsen stress urinary incontinence (SUI), which patients must be warned off. During urethroscopy, sphincter assessment can be performed whereby the functional sphincter length (coaptive zone) could if demonstrated suggest suitability for retrourethral transobturator male sling insertion (9).
Urodynamic evaluation is recommended, to distinguish the cause of urinary incontinence and determine whether there is any element of detrusor overactivity (8). The ability of the test to predict outcome of surgery for SUI is uncertain. There has been some small level evidence to suggest that a valsalva leak point pressure (VLPP) of >100cmH2 0 has a high predictability for post-operative success with the AdVance male sling (3). Some tests to assess sphincter function such as urethral pressure profiles or retrograde leak point pressures are also of use to diagnose sphincter incompetence. Urodynamics however should be carried out using the standards laid out by the International Continence Society (ICS) document ‘Good Medical Practice’ (10).
Once a correct diagnosis has been made, and the severity of the patients symptoms and bother has been established, further treatment can be planned.
Conservative managementLifestyle measures include the restriction of bladder irritants such as caffeine, reduced fluid intake when out, timed voiding and the use of penile clamps. Electrical stimulation of the pelvic floor is not currently recommended for male SUI (3). Other lifestyle interventions include supervised pelvic floor muscular training (PFMT), which is recommended for all men post-operatively to hasten recovery of continence (8). PFMT has been shown in large randomised clinical trials to improve continence rates in the short to long term (11). PFMT must be performed to at least over three sets of 10 repetitions of muscle training daily.
With regards to pharmacotherapy, antimuscarinics can be used to treat predominant overactive symptoms (OAB). In mixed urinary incontinence, the most bothersome symptom complex should be treated first. There is low level evidence that drug treatment for male SUI using duloxetine, a serotonin-nor-adrenaline reuptake inhibitor, may be of use with a reduction of incontinence in a small randomised study of 31 patients against placebo (12). However this form of treatment should be considered as a system of temporary symptom relief rather than a cure. There is some evidence to suggest that duloxetine may have synergistic properties when combined with PFMT.
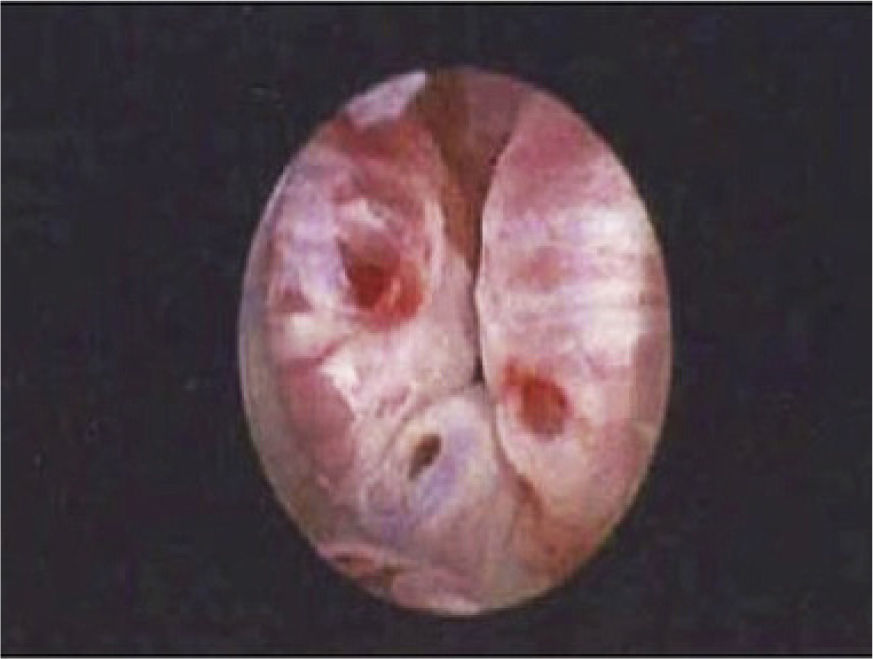
Surgical treatment for male suiBulking agentsThe most minimally invasive option of peri-urethral bulking using collagen, offers patients symptomatic improvement, but is seen more as a short term option, with little durability (See figure 1). A review of 322 men, showed that although having significant symptomatic improvement, men were still wearing upto 3 pads a day and by 40months follow-up, the average number of repeated treatments was 4 (13). Bulking agents should therefore not be offered to patients with severe SUI, but to patients with mild to moderate SUI, with the understanding that this would provide temporary symptomatic relief.
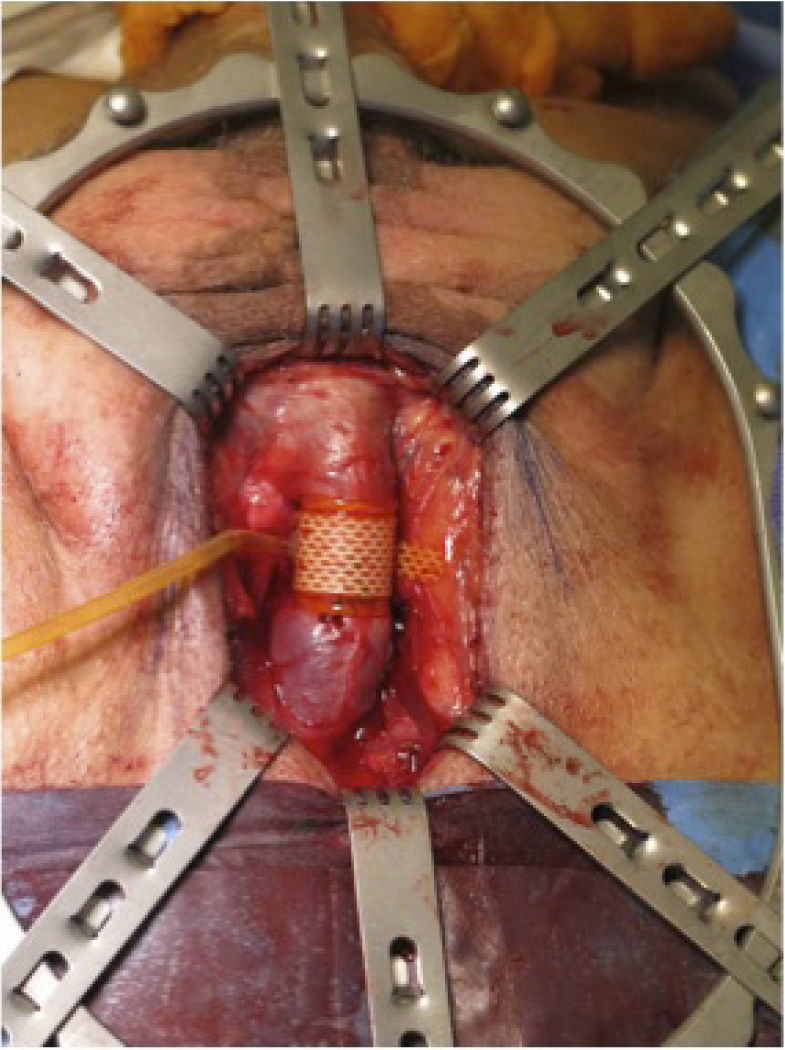
Artificial urinary sphincterThe gold standard surgical treatment for many years has been the Artificial Urinary Sphincter (AUS), for moderate to severe PPI (See Figure 2). The first generation was announced in the 1970s, and since then long term data has suggested its durability. It can also be applied to patients treated with previous radiotherapy, and after failure of other treatment options, a characteristic not applicable to the other treatments. The AMS 800 (Boston Scientific, Marlborough, MA, USA), is the most commonly used device, consisting of 3 parts, a urethral cuff, a pump placed in the scrotum and a reservoir placed in the abdomen (3). The urethral cuff applies pressure around the urethra, providing continence, which can be deflated with compression of the scrotal pump, when the time has come to pass urine. Studies have suggested that dry rates are upto 85.7% (14). With regards to longevity of therapy, a pooled analysis of complications after single-cuff placement, urethral erosion and infection affect upto 8.5% and a global reintervention rate for any reason is at 26% (15). However patient satisfaction has been shown to correlate with the functional outcome and not the number of interventions required. One long-term study demonstrated at 15years. 41% had the native AUS insitu, whereas 47% have a revised AUS, and 12% no longer have an AUS in situ. There is some evidence to suggest that the dry rates are superior when the classic perineal approach to the urethra is taken, over the peno scrotal route (16). There is no evidence to suggest that using a double urethral cuff device is superior, and there is evidence to suggest it may increase complication rates (3).
Although the AUS is the only effective treatment option for PPI, in patients who have received previous radiotherapy, there is evidence that the success rates in irradiated patients (56%) are less than in nonirradiated men (89%) (17).
There are other AUS systems, including the Zephyr Surgical Implants ZSI 375 system, which is 2 part device, preconnected, with no need for an intraperitoneal reservoir balloon hence avoiding the abdominal incision. However the evidence for this system is weak, and only based on small series data, and implantation of this is not recommended outside of the setting of a clinical trial.
Retrourethral transobturator slingsThe use of polypropylene mesh, comes with the advantage of its simplicity, with no scrotal pump, and potentially immediate post-operative continence offered. The most common type of sling is the Advance sling system, which is a non-adjustable mesh, which repositions the bulbar and posterior urethra, increasing the functional sphincter length. Most studies suggest cure rates upto 63% at a follow-up to 40months (18). Most common complications include perineal pain and transient urinary retention. Severe complications and sling explantations are noted to be rare. This tends to be a well-tolerated procedure with a short post-operative stay. The evidence for the efficacy of slings in patients with previous pelvic irradiation is reduced, and therefore use of mesh slings in this group are not recommended (3). The ongoing MASTER trial (Male synthetic sling versus the Artificial urinary sphincter) is comparing the efficacy of these two treatments in a head to head analysis.
ConclusionsLess than 10% of men undergoing radical prostatectomy may end up requiring anti-incontinence surgery. The correct diagnosis is critical, that should also assess the urethra for any anastomotic strictures and urodynamic assessment for underlying bladder dysfunction, especially in the radiotherapy cohort. First line therapy remains PFMT, and bulking agents may provide temporary relief to men with mild SUI. Invasive therapy for moderate to severe SUI, if warranted should involve a discussion with the patient about the AUS, the current gold standard treatment. There is emerging evidence to suggest a role for male slings in the mild to moderate SUI cohort. No randomised trial has as yet investigated the outcome of one surgical treatment versus another, however further prospective series data is encouraged.
The authors declare no conflicts of interest, in relation to this article.