Casos Clínicos en Gastroenterología y Hepatología
Más datosMultiple myeloma (MM) is a common hematologic malignancy characterized by abnormal proliferation of plasm cells. Gastrointestinal (GI) involvement is rare and more often detected after the diagnosis of bone marrow disease. Herein, we present a rare case of a patient presenting with GI complaints ultimately diagnosed with MM.
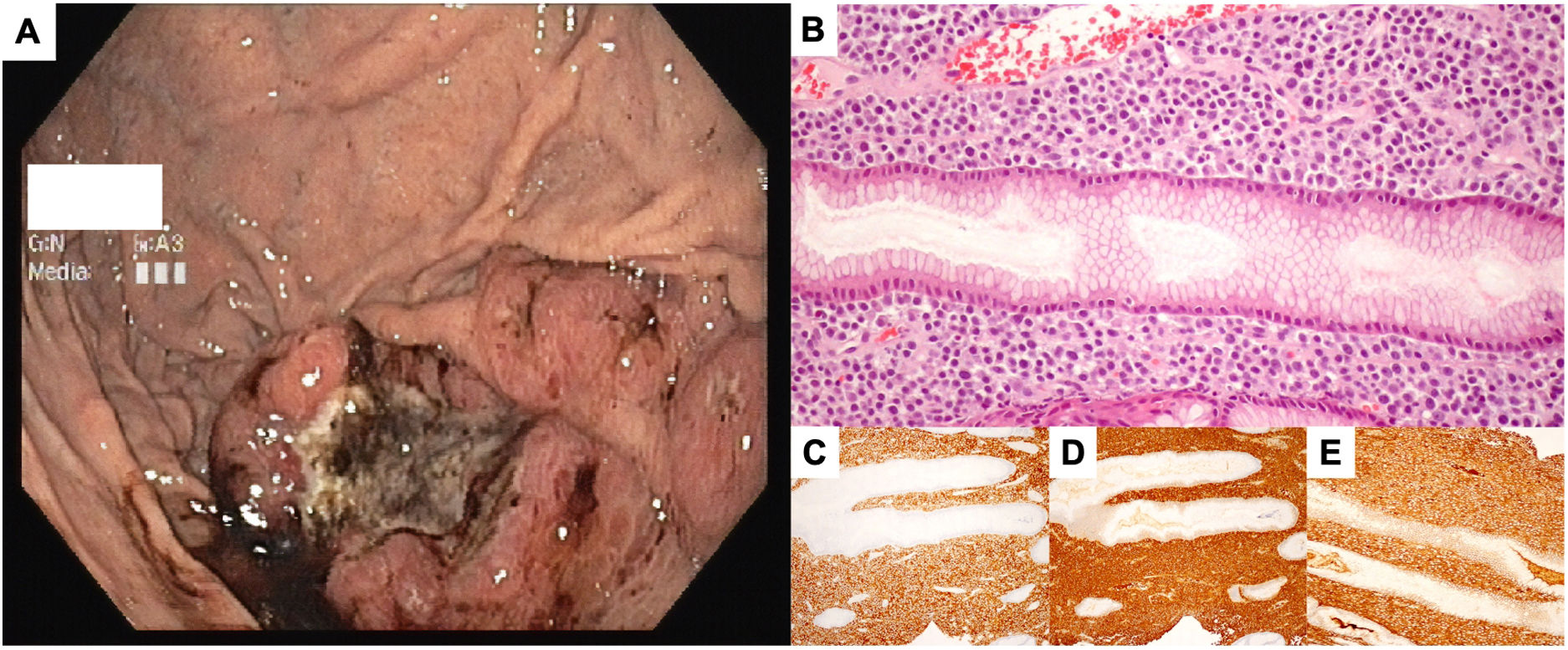
A 66-year-old male patient presented with a 6-month history of heartburn and epigastric pain. He denied other symptoms, including regurgitation, dysphagia, chest pain, and weight loss. His clinical history was remarkable for diabetes and dyslipidemia, for which he was being treated, and Helicobacter pylori-positive gastritis, with proved eradication. A recent lab study was unremarkable. The patient underwent upper esophagogastroduodenoscopy which revealed a gastric ulcer located in the greater curvature of the body with approximately 5cm of diameter and regular borders (Fig. 1A). An erythematous halo extended distally along the greater curvature for 10cm. Biopsies of the lesion revealed gastric body mucosa with infiltration of the lamina propria by intermediate-large neoplastic cells, many with peripheric nuclei, prominent nucleolus, and abundant cytoplasm. Immunohistochemistry revealed CD38-positive neoplastic cells, with expression of immunoglobulin alpha and lambda chains (Fig. 1B–E). These findings were compatible with an IgA lambda gastric plasmacytoma. Further immunofixation was compatible with IgA lambda monoclonal gammopathy. Bone marrow biopsy was compatible with criteria for MM. Further workup revealed an osteolytic lesion in the occipital bone. Lab values revealed had no signs of anemia, hypercalcemia or impaired renal function. The patient was proposed systemic chemotherapy with 4 cycles of Bortezomib plus dexamethasone followed by autologous hematopoietic stem-cell transplant (AHSCT). A gastric biopsy performed after the completion of the chemotherapy revealed no signs of plasm cell infiltration. Similarly, a bone marrow biopsy showed no plasm cells and immunofixation revealed no signs of monoclonal gammopathy, but an abnormal kappa/lambda relation persisted on peripheral blood. The patient was submitted to AHSCT, and complete remission was maintained for the following 9 months. At that point, the monoclonal IgA lambda spike and the gastric lesion reappeared. The patient was treated with 6 cycles of lenalidomide plus dexamethasone, for which the he demonstrated partial response. After the 6 cycles of salvage chemotherapy, the patient presented with upper gastrointestinal bleeding. Endoscopic evaluation at that time revealed a large ulcerated lesion extending from the cardia to the proximal antrum. Biopsies revealed infiltration by the previously diagnosed MM. The patient's clinical status rapidly deteriorated. Further clinical course was complicated by several pathologic fractures and uncontrollable pain, renal failure and multiple infections. The patient died 4 months after the diagnosis of the recurrence.
(A) upper gastrointestinal endoscopy showing an ulcer with 4×5cm with regular borders and hemosiderin pigment, located in the greater curvature of the body; (B) Hematoxylin-eosin (20×) showing infiltration of the lamina propria with intermediate size neoplastic cells, many with eccentric hyperchromatic nuclei and a large cytoplasm; (C)–(E) immunohistochemistry showing positivity for CD38, lambda and alpha chains, respectively, compatible with infiltration of plasma cells expressing IgA lambda.
Gastrointestinal involvement in MM is rare, and it is most often reported as single case reports.1–3 A report of a database including more than 2500 MM patients found a prevalence of 0.9%.4 Sunkara et al. provide a summary of reported cases and respective clinical presentations.3 The clinical picture is extremely variable as it depends on the affected organs. GI involvement may produce nonspecific symptoms, which may not be adequately valued by the patient or the clinician, or even be completely silently. Such cases may be found posthumously at autopsy and, therefore, the reported prevalence of GI involvement may be underestimated.4 Most cases of GI involvement by MM are discovered after the diagnosis of the primary hematologic condition. The occurrence of GI complaints leading to the diagnosis of MM involving the GI tract, as reported in our case, is extremely rare.4 The endoscopic phenotype is nonspecific and may include ulcers, nodules and fold thickening.3,5 Our case highlights the importance of histopathological analysis for a correct diagnosis. Furthermore, immunohistochemistry is crucial for accurate identification of cellular lineage in cases with primary extramedullary involvement, as was the case of our patient.
Patients with MM and GI infiltration often display cytogenetic and phenotypic characteristics that are associated with worse prognosis. In fact, the occurrence of complex karyotype and plasmablastic in the bone marrow plasmacytes is more frequent in patients with concomitant gastric plasmacytoma.4 The prognosis of these patients is poor. Talamo et al. reported a median survival of 7 months after diagnosis.4 Aggressive chemotherapy and stem-cell transplant are often effective in providing complete responses. Unfortunately, a disease relapse almost invariably occurs.4
Ethics statementThe patient's identity was preserved, and all patient data has been anonymized. Informed consent was obtained from the patient for the publication of his case.
Authors’ contributionsMiguel Mascarenhas Saraiva: wrote and revised the manuscript.
Tiago Ribeiro: wrote and revised the manuscript.
Amadeu Corte Real: revised the manuscript.
Elsa Fonseca: obtaining and editing of microscopic images; histologic analysis.
Guilherme Macedo: revision and approval of the final version of the manuscript.
Conflicts of interestNone declared.