HPV (human pappiloma virus) infection is an etiologic risk factor for esophageal carcinoma with several studies supporting its carcinogenic role. The main strains are HPV16 and 18 and the less frequent strains 11, 6, 31 and 36. We report the case of a 58 year old male, smoker, admitted to our hospital for progressive dysphagia. Previous endoscopies and biopsies were compatible with a hyperkeratotic esophagus, negative for dysplasia. Second endoscopy showed a hyperkeratotic, verrucous, esophagus with two circumferential stenosis which were dilated with TTS (Through the Scope) balloon. Biopsies revealed multiple squamous cell abnormalities, negative for dysplasia and positive for HPV 11. Symptoms recurred, and he was submitted to another upper gastrointestinal endoscopy with dilation of the stenosis and esophageal stent placement. Two months later, esophagectomy was performed, and the histopathological analysis revealed a squamous cell carcinoma of the esophagus (T2, N0, M0, G1). The patient died after surgery due to acute respiratory distress syndrome.
La infección por el virus del papiloma humano (VPH) es un factor de riesgo etiológico en el carcinoma esofágico, y varios estudios apoyan su papel cancerígeno. Las cepas más comunes son el VPH 16 y 18 y menos frecuentes VPH 11, 6, 31 y 36. Se presenta el caso de un varón de 58 años, fumador, que ingresó en nuestro hospital por disfagia progresiva. Las endoscopias y biopsias previas eran compatibles con esófago hiperqueratósico y presentaban resultados negativos para displasia. Una segunda endoscopia mostró esófago hiperqueratósico, verrugoso con dos estenosis circunferenciales que fueron dilatadas endoscópicamente con balón TTS (del inglés Through the Scope). Las biopsias revelaron múltiples anormalidades en células escamosas, fueron negativas para displasia y positivas para el VPH 11. Dada la recurrencia de los síntomas, el paciente fue sometido a otra endoscopia de las vías digestivas superiores para realizar dilatación de la estenosis y colocación de endoprótesis esofágica. Dos meses más tarde se llevó a cabo una esofagectomía y el análisis histopatológico reveló carcinoma escamoso de esófago (T2, N0, M0, G1). El paciente falleció después de la cirugía debido a un síndrome agudo de dificultad respiratoria.
The association between human papillomavirus (HPV) infection and esophageal carcinoma (EC) was first mentioned 1982. HPV DNA has been isolated from esophageal squamous cell carcinomas with prevalence rates of 0–66%1 providing a causative link in at least a fraction of squamous cell carcinomas, particularly in high risk areas. HPV 16 and 18 are the viral types most commonly identified and few reports identify strains HPV 6, 11, 31 and 33.2–5 Verrucous esophageal carcinoma, a rare subtype of esophageal squamous cell carcinoma (ESCC), has also been described in association with HPV.2 The role of HPV in carcinogenesis seems to be through overexpression of p53, mdm2, erc-B2 and loss of p21. These factors have been shown to decrease overall survival at 5 years in esophageal squamous cell carcinoma.3 However this relation is still not firmly established, with contradictory results having been reported in the literature.6 On the other hand, there is scant evidence supporting an etiological relation between HPV and Barrett's esophagus and adenocarcinoma.1 We report a case of an ESCC in a hyperkeratotic, verrucous esophagus with positive HPV 11.
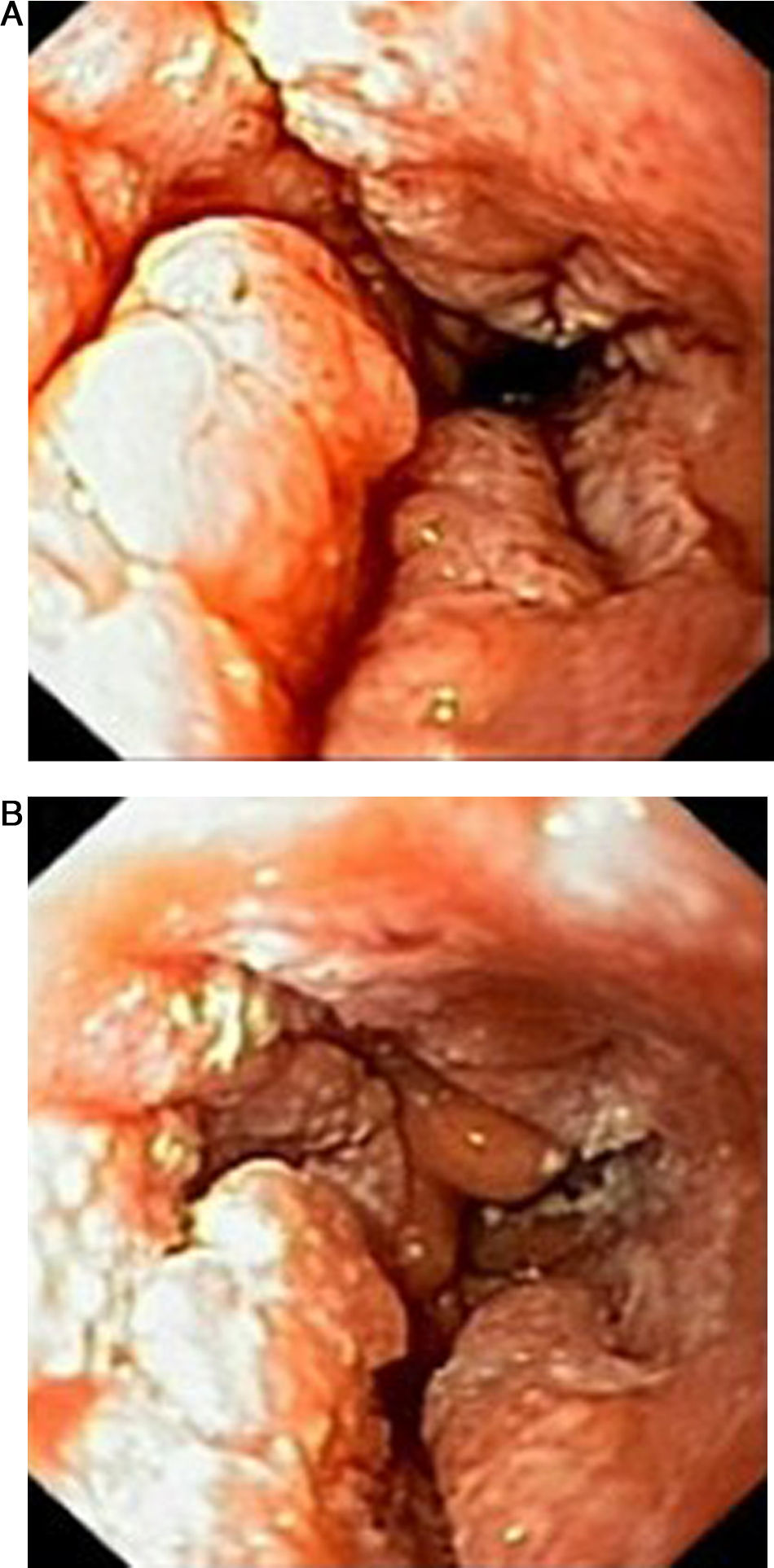
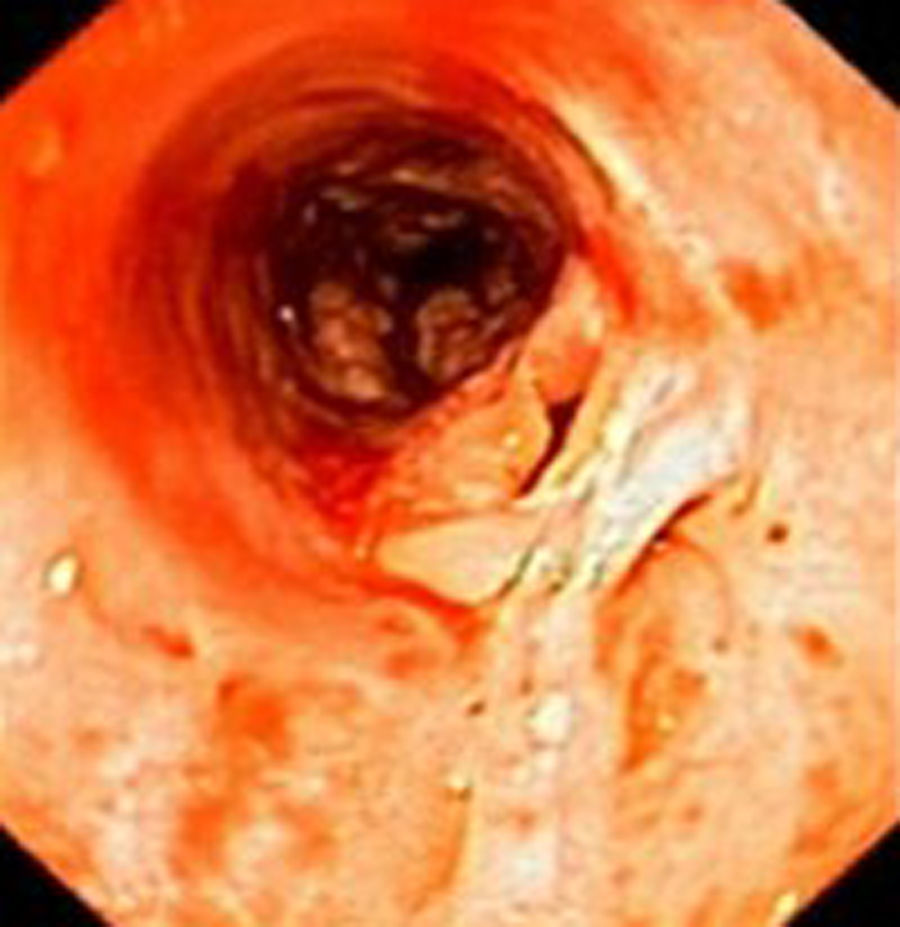
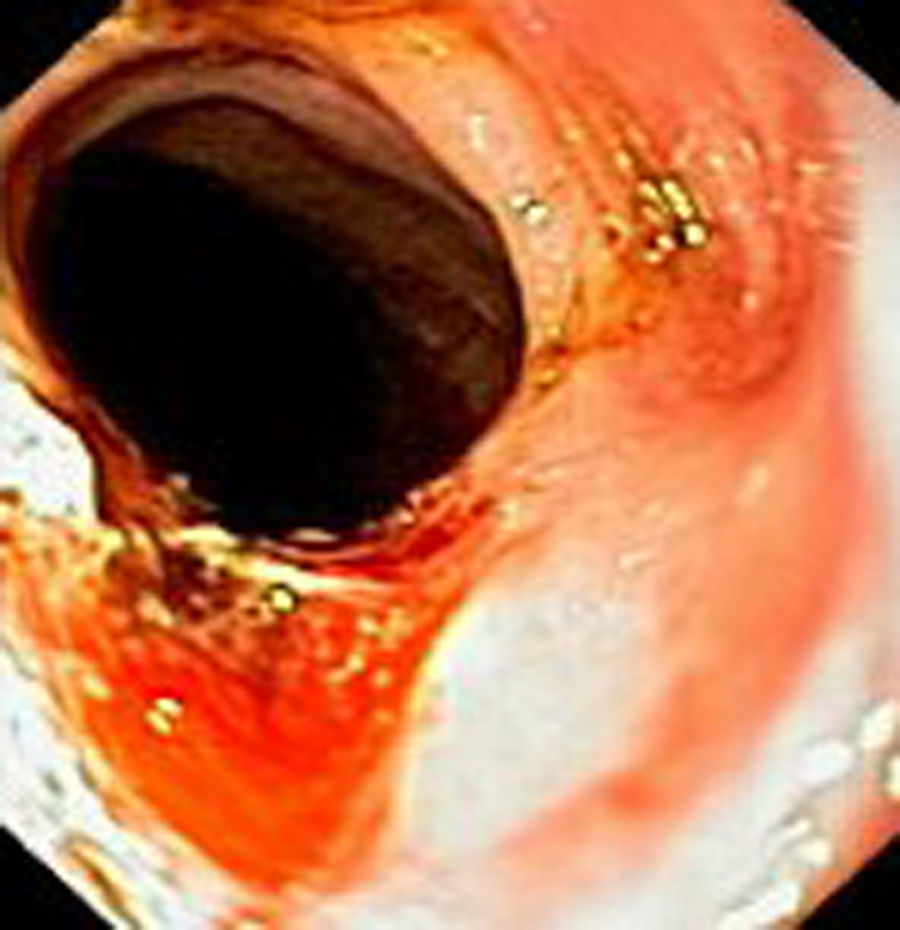
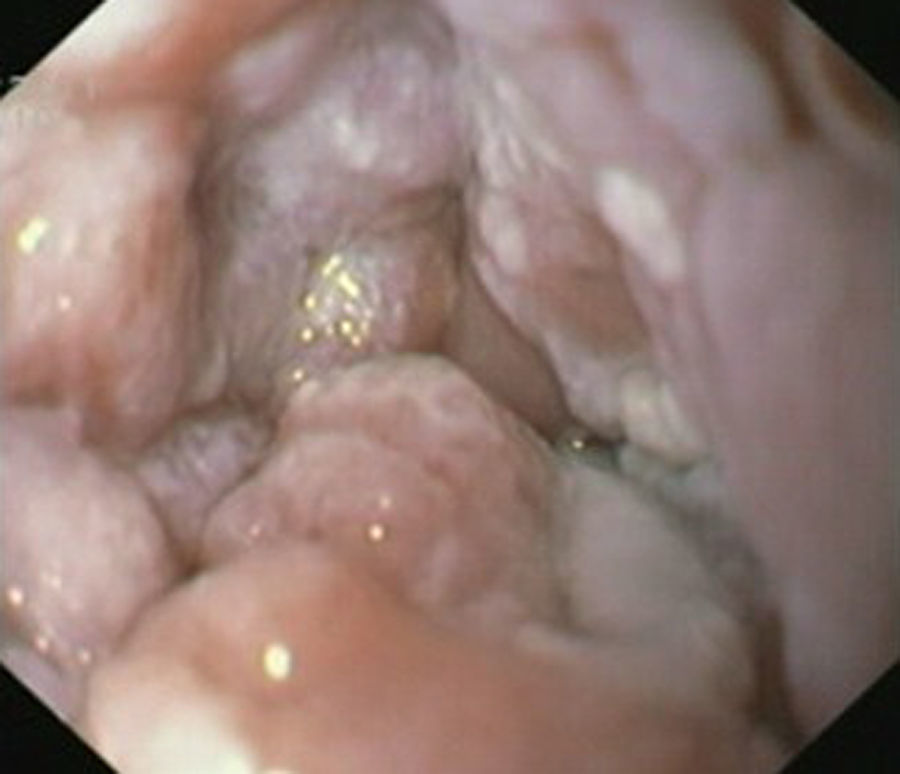
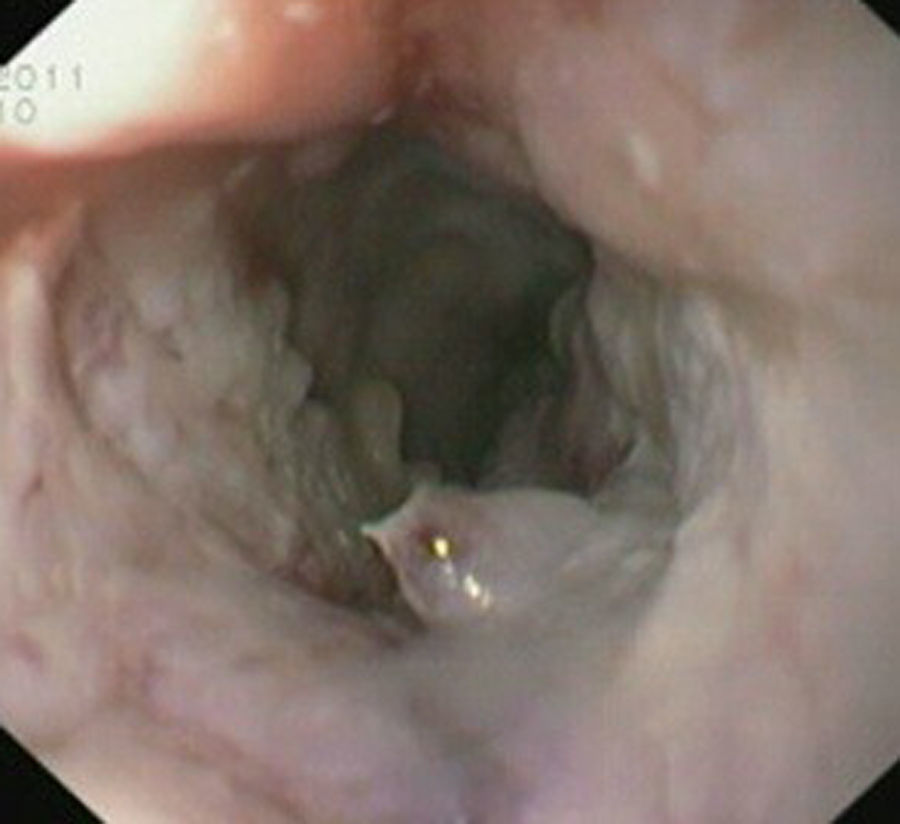
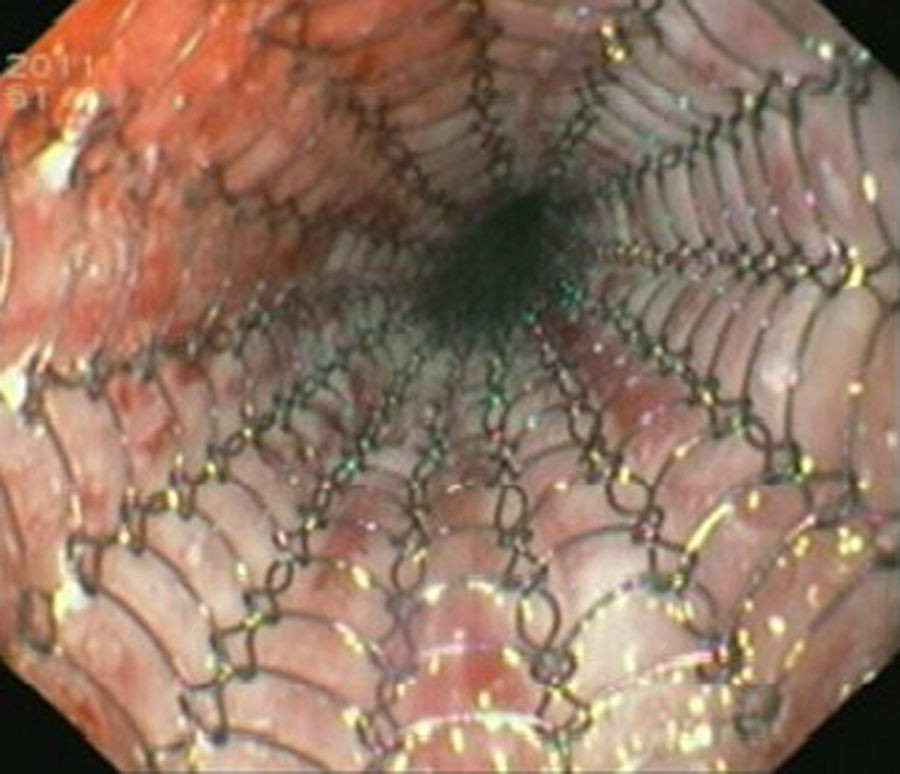
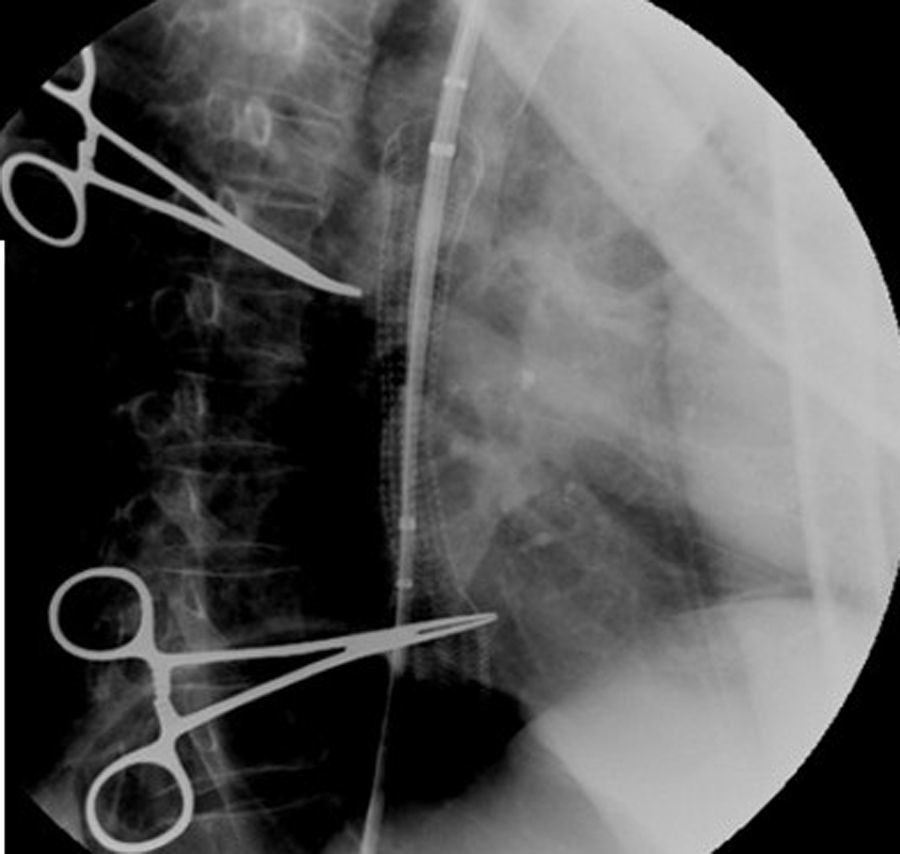
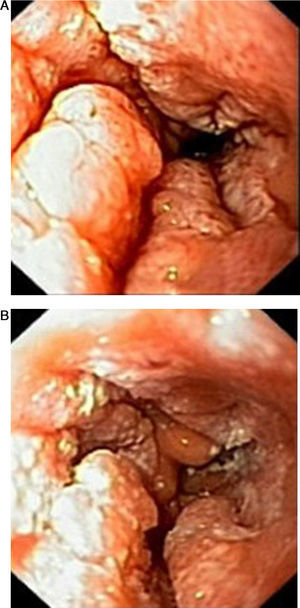
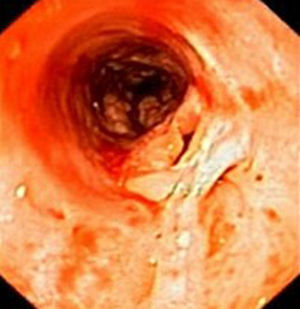
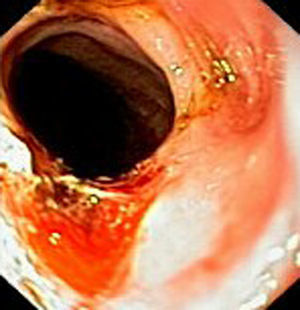
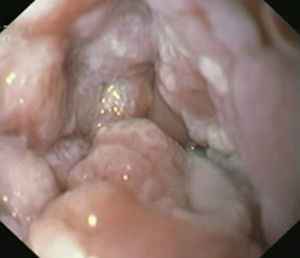
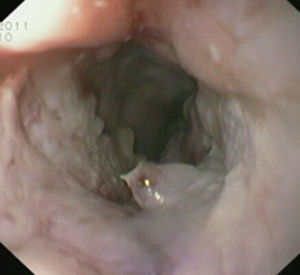
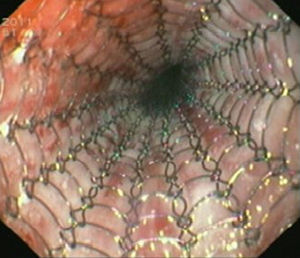
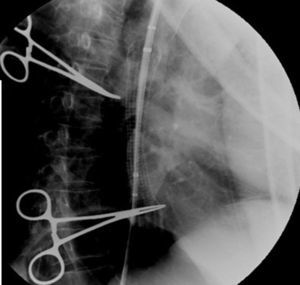
Case reportIn June 2011, a 58 year old man was admitted to our hospital with a four month history of progressive disphagia, for both solids and liquids, and weight loss. He had several previous upper endoscopies, all of which revealed white plaques covering the esophageal mucosa which showed pseudopolipoid smooth mass appearance in some areas protruding into the lumen, causing stenosis at 30cm from the dental arcade margin. Multiple biopsies were obtained and were negative for dysplasia, showing a malpighian epithelium. The patient was a heavy smoker and had had lower extremity amputation due to severe peripheral arterial disease with ischemia. At our hospital, we performed an endoscopy (Figs. 1–3). Esophageal mucosa was normal till 26cm from the dental arcade. Between 26 and 34cm, the mucosa had a hyperkeratotic appearance, with thickened folds, irregular white plaques with pseudopolipoid, verrucous formations. At 30cm, there was a circumferential stenosis, which we could not pass with the scope. TTS Balloon dilation till 36F was performed. Between 34 and 36cm, the mucosa was normal but at 36cm a second stenosis existed and was dilated with a TTS balloon till 36F. Multiple macrobiopsies were performed showing an acanthotic, hyperkeratotic, papillomatoid mucosa with focal koilocytes; it was undefined for neoplasia and negative for fungal organisms. In situ hybridization was positive for HPV 11 and negative for HPV strains 16 and 18. A CT scan was performed and showed no adenopathy in mediastine. Despite not having established accuracy in the setting of verrucous esophageal carcinoma staging, performing an esophageal EUS could have given relevant information suggesting malignancy. However, our EUS scope available was the radial Olympus GF160 UL5, with an outer diameter of 13.8mm, quite superior to the maximum dilatation achieved (12mm, 36F). The patient clinically improved and was discharged. One month later, he was readmitted with complete disphagia. The endoscopy showed significant re-stenosis of the former two esophageal stenosis that had been dilated. We dilated both with TTS balloon and, after discussion with surgery we placed a fully covered metallic stent (Hannarostent, 120mm, 23/28mm) (Figs. 4–7), so as to optimize his nutritional status. The patient developed septic shock 48h after the procedure, with no radiologic findings of pneumomediastinitis on CT scan. Shock resolved under antiobiotic (vancomicin and meropenem) and vasopressor therapy. Despite the absence of neoplasia on biopsies, it was decided to perform surgery. He underwent a laparoscopic esophagectomy, converted to laparotomy and thoracotomy during surgical intervention. Post-surgery, he developed acute respiratory distress syndrome (ARDS) and died two days after the procedure. Histopathological analysis of the surgical esophageal specimen revealed well differentiated squamous cell carcinoma involving the submucosa and focally the muscle layer, with no lymphatic involvement in the 14 ressected lymph nodes. There was no perineural or vascular invasion and both gastric and esophageal margins were free of macro and microscopic disease. There were several findings of acanthosis, papillomatosis and hyperkeratosis with koilocytes, but HPV was negative in the tissue. Therefore, the patient had an ESCC stage IIA (T2, N0, M0, G1).
Our patient had two risk factors for ESCC: smoking and the HPV infection in the esophagus. Although, not from an endemic area, HPV infection has been reported to be present in 2–6.7% of the ESCC patients in Western countries and North America.3,7–9 The strain HPV 11 detected, is not the most commonly described so far.3 In 2005, in a study3 with fifty EC patients, twenty seven (54%) were HPV positive but only three (6%) were of the HPV 11 type (HPV 16–15 (30%) and HPV 18–9 (18%)]. However, no correlation was found between the HPV-DNA and clinical features such as sex, age, EC tumor type, mitotic and keratinization grade. Also in a study conducted between 2002 and 2005 in Australia, gathering 222 patients with ESCC, with only 3.6% prevalence for HPV, no difference was found between patients HPV positive versus negative regarding sex, age, body mass index (BMI), ESCC staging or alcohol consumption. However HPV positive patients tended to be younger and had higher BMI.10 Recently, a study by Gupta et al. demonstrated a tendency of HPV positivity in non keratinizing ESCC type, a conflicting result considering former studies emphasizing the need for larger studies in this area.11 There are several possibilities to explain why HPV was not detected in the tumor itself. A case report of a verrucous carcinoma2 in the setting of hyperkeratotic esophagus with positive HPV-type 51 detected in biopsies, also turned out negative for HPV in the surgical specimen. The absence of HPV DNA detection, could be explained by a possible sampling error, possible DNA destruction in between surgery and pathology analysis, or actual absence of HPV in the tumor, despite its presence in the initial phase with the development of hyperkeratotic squamous cells abnormalities. This case illustrates the possible role for HPV infection in ESCC. Also, in hyperkeratotic esophagus, malignancy may not be detected through several biopsies and surgical resection can be performed if suspicion for malignancy exists. Surgery is the recommended therapy for verrucous carcinoma, in case it does not spread to involving structures, without clear efficacy of radiation and chemotherapy.9
Conflict of interestThe authors have no conflicts of interest to declare.