Rapid transfusion of blood products and the presence of ionic changes as hypocalcaemia and hyperkalaemia are common in liver transplantation. The objective of this paper is to give the reader a clear and practical description of the etiological factors, biochemical mechanisms, diagnosis and treatment of the calcium and potassium plasmatic disorders associated with massive transfusion. The peculiarities that arise in the clinical setting of liver transplant surgery and citrate intoxication are highlighted. A non-systematic review of literature was conducted in MEDLINE, OVID and Cochrane databases. Correct and early anaesthetic management of calcium and potassium disorders prevents serious complications in intraoperative bleeding risk surgeries such as liver transplantation.
La transfusión rápida de hematocomponentes y la presencia de alteraciones iónicas como la hipocalcemia y la hiperpotasemia son frecuentes en el trasplante hepático. El objetivo de este trabajo es brindar al lector una descripción ordenada y práctica de los factores etiológicos, mecanismos bioquímicos, diagnóstico y tratamiento de las alteraciones del calcio y del potasio asociadas a la transfusión masiva. Se destacan las particularidades del contexto clínico de la cirugía de trasplante hepático y se describe la intoxicación por citrato y sus factores predisponentes. Se realizó una revisión no sistemática de la literatura en las bases de datos MEDLINE, OVID y Cochrane. El manejo anestésico correcto y precoz de las alteraciones del calcio y del potasio evita complicaciones graves en el intraoperatorio de las cirugías con riesgo de hemorragia, como el trasplante de hígado.
Liver transplant is one of the surgical procedures that often requires transfusion of large volumes of blood products. This gives rise to complex alterations of the internal milieu that may result in life-threatening intraoperative events. Immediate identification and anticipation of these disorders is a mainstay in anaesthetic management. The objective of this review is to provide an overview of the mechanisms and treatment of the most frequent serum calcium and potassium abnormalities occurring as a result of using large volumes of blood products for replacement. Particular details on citrate intoxication, hypocalcaemia and hyperkalaemia will be provided in the context of acute massive haemorrhage during liver transplant surgery.
MethodologyNon-systematic review of the literature was conducted in the MEDLINE, OVID and Cochrane databases using words such as: “hypocalcaemia”, “hyperkalaemia”, “massive transfusion”, “acidosis” and “liver transplantation”. Relevant articles and associated references that help understand the aetiology, diagnosis and treatment of calcium and potassium disorders during massive haemorrhage were selected. Relevant articles for the interpretation of those disorders during liver transplant surgery were also included.
Massive haemorrhageMassive haemorrhage is one of the main causes of death and intraoperative cardiac arrest in adults as well as children.1–4 It is usually defined in relation to the volume of blood products transfused over 24h by kilograms of body weight: 10 volumes of red blood cells (RBCs) in a patient weighing 60kg,5 replacement of more than 50% of the blood volume in 3h, or transfusion of more than 4 volumes of RBCs in 1h.
However, in cases of acute haemorrhage happening within a period of minutes, immediate identification is required in order not to delay therapeutic action. For that reason, in anaesthesia we prefer to consider millilitres of blood lost in a few minutes: 150ml/min or more than 1.5ml/kg/min over a period of more than 20min.6 Mortality is associated with the presence of acidosis (pH<7.1), hypothermia, coagulopathy, number of concentrated blood products transfused and volume ratios between the different blood products given.7 The acute complications of massive haemorrhage are related with shock and transfusion therapy.8,9 We will focus on two of these, namely, citrate toxicity7,10,11 and hyperkalaemia.4,7,12 Hypomagnesaemia is another frequent ionic abnormality in patients with massive haemorrhage, although it does not seem to be significantly associated with mortality.13
HypocalcaemiaAlthough it is defined as a total serum calcium concentration of less than 8.5mg/dl (4.5mEq/l, 2.10mmol/L),14 clinical hypocalcaemia may occur even with normal total calcium values when serum ionized calcium concentrations are lower than 4.5mg/dl. In surgery, the most common causes of hypocalcaemia are hyperventilation and citrated blood infusion at a rate of more than 1.5ml/kg/min. Acute respiratory alkalosis reduces ionized calcium by lowering hydrogen ion concentrations, freeing albumin binding sites and leading to increased ionized calcium protein binding. The clinical manifestations of hypocalcaemia are due to the lowering of ionized calcium, because it is this free fraction which acts on membrane potentials.15 That is why it manifests in excitable tissues: changes in mental status (central nervous system), tetany (skeletal muscles), hypotension (smooth muscle) and arrhythmias, prolonged QT interval or pulseless electrical activity (myocardium).9,14 Plasma calcium levels are a poor surrogate indicator for total body calcium, accounting for only 0.1–0.2% of the extracellular calcium and 1% of total body calcium. Ionized calcium, in turn, usually represents 40–50% of plasma calcium. Total serum calcium concentrations must be interpreted in relation to serum albumin. In the presence of hypoalbuminemia, there is less substrate for calcium binding, allowing for a larger percentage of free calcium, ionized calcium. In this situation, plasma calcium values may underestimate ionized calcium values.16 Serum calcium concentrations are corrected in relation to a concentration of albumin of 40g/L; for every 1g/L of albumin above or below this value, calcium is adjusted by lowering or increasing it by 0.02mmol/L. This estimate may not be accurate in critically ill patients.17 The use of 5% albumin and of blood products during liver transplant determines protein binding of calcium ions.18 On the other hand, in critically ill inpatients with hypoalbuminemia there may be a lower value of total calcium without lowering of ionized calcium.16 In these cases, there is no need for acute calcaemia correction, but improving nutrition is required.14
During blood transfusion, the degree of hypocalcaemia depends of the patient's volemia, the volume of blood products administered, the transfusion rate, and liver function.2
Hypocalcaemia during massive haemorrhage is a predictor for mortality, and there is a linear relationship between calcaemia values and mortality. Lower plasma calcium is a better indicator of hospital mortality than minimum fribrinogen concentration, acidosis or low platelet counts.19
Citrate intoxicationIt manifests as signs of hypocalcaemia.20 Stored blood is anticoagulated using citrate (3g/unit of RBC), which chelates calcium. In a healthy adult, the liver metabolizes 3g of citrate in 5min. Infusion rates greater than 1 unit of RBC/5min, or liver dysfunction, drive citrate elevation and lower plasma ionized calcium.21 When circulating volume is well maintained, cardiovascular manifestations occur with infusion rates of 150ml/70kg/min of citrated blood. However, when there is hypothermia of 31°C, citrate metabolism rates drop by 50% and toxicity may occur with slower infusion rates. With the new preservatives (lower citrate content), intoxication is less probable.21 A similar situation occurs when sodium bicarbonate (HCO3) is infused at a fast rate; free calcium binds to HCO3, lowering the percentage of ionized calcium.16 In low-flow states (cardiac arrest or haemodynamic arrest with electrical activity) there is ionized calcium lowering22 independent from total concentration. The mechanism is a disrupted distribution of free calcium. Consequently, if citrate-containing blood is administered to a patient with tissue hypoperfusion, ionized calcium reduction will be greater than in stable situations. It is important to improve the patient's haemodynamic state in order to mobilize ionized calcium from body stores. Paradoxically, blood administration may improve plasma levels of ionized calcium as it improves circulatory status. Acidosis and the total volume of transfused fresh plasma are associated with severe hypocalcaemia in patients with massive bleeding.19
Neonates are at risk of developing heart failure due to hypocalcaemia during transfusion, because cardiac function (relaxation and contraction) depends largely on plasma concentrations of ionized calcium.2 When neonates present liver failure leading to lower citrate metabolism and the risk is very high, death may ensue.3 Citrate toxicity may be prevented in these cases if the transfusion rate is kept below 1ml/kg/min2.
During liver transplantation, hypocalcaemia is multifactorial15,23: transfusions, diminished metabolic capacity of the liver,24 and reduced liver blood flow. As a result, there is a need to infuse high calcium doses during liver transplantation (1g/h of calcium gluconate).21
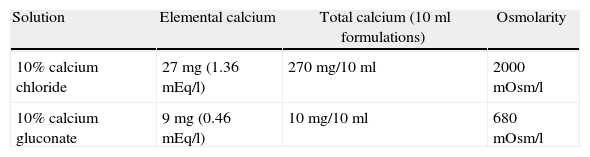
Treatment of hypocalcaemia: calcium gluconate and calcium chlorideIntravenous calcium administration is the appropriate treatment for acute or severe hypocalcaemia, using 10% calcium chloride (1.36mEq/l) or calcium gluconate (0.45mEq/l). The former provides 3 times as much calcium than an equal volume of 10% calcium gluconate, because chloride molecular mass is 147 compared to the molecular mass of gluconate of 448.21Table 1 shows the chemical differences between calcium chloride and calcium gluconate. If an equivalent dose of calcium is administered in either of the two formulations,25 the therapeutic response is similar, there being no significant differences between the dissociation rates for both compounds.25 The two preparations may be used with similar efficacy for the treatment of intraoperative hypocalcaemia.26,27 Calcium gluconate is the preferred form for intravenous use, given that calcium chloride tends to cause local irritation when used through a peripheral line, and has to be administered through a central venous line.14 In the event there are signs of hyperkalaemia or hypocalcaemia, a bolus administration is required under mandatory electrocardiographic monitoring, due to the risk of arrhythmias.28 The recommended dose for treating intraoperative hypocalcaemia in case of bleeding requiring blood product transfusions is 5–10mg/kg of calcium chloride or 15–30mg/kg of calcium gluconate, the requirement in children and neonates being higher.2 This usually results in transient improvement (considering that the two preparations have a short half-life) and there is a need for continuous calcium administration to prevent recurrent hypocalcaemia and dangerous fluctuations of plasma levels.17,29
HyperkalaemiaIt is defined as a serum potassium level greater than 5.5mEq/l. It is usually considered mild up to 6mEq/l, moderate between 6 and 7mEq/l, and severe when greater than 7mEq/l. It may be caused by external or internal balance disruptions.30,31 The most frequent causes include severe renal failure, iatrogenic injury, the use of angiotensin converting enzyme (ACE) inhibitors32 and bank blood transfusions.4,33 In anaesthesia and surgery, perfusion of an extensive, previously ischaemic vascular bed triggers the release into the circulation of large quantities of potassium resulting from the outflow of intracellular potassium, due to disrupted membrane pumps from local acidosis. In liver transplant, the perfusion of the recently placed graft (ischaemic and preserved in a solution with a high potassium content) gives rise to sharp increases of plasma potassium that may provoke the death of the patient.34,35
Hyperkalaemia and acidosisAcidosis increases plasma potassium concentrations by inducing outflow from the cell into the extracellular compartment through hydrogen exchange (altered internal balance).31 In the kidney, H+ increases result in reduced tubular potassium secretion, disrupting the external balance as well.31
Hyperkalaemia and blood transfusionRapid RBC transfusion may result in cardiac arrest.4,33 Plasma potassium concentration increases in stored blood in a manner directly proportional to storage time (0.5 and 1mEq/l per day), reaching values of up to 7–77mEq/l in RBCs.36 The mechanism is potassium outflow from red blood cells due to Na2+−K-ATPase membrane pump as a result of the lack of ATP. Moreover, RBCs contain a CPD (citrate-phosphate-dextrose) or CPDA (citrate-phosphate-dextrose-adenine) solution with a pH of 5.5, which lowers the pH from 7.0 down to 6.6 after 21–35 days of storage. The result is an accumulation of potassium, fixed acids and CO2 that may produce myocardial depression when infused in the context of massive bleeding.37 Potassium concentrations in RBC units increase with radiation and diminish with red blood cell washing.38 Rapid transfusion through a central venous catheter may deliver higher potassium concentrations into the coronary circulation than when given through a peripheral venous line, and this may contribute to the risk of cardiac arrest.9,37 Moreover, some pressurized infusion devices may traumatize red blood cells, giving rise to greater potassium outflows from the cell.39 The new rapid infusion and fluid warming devices do not produce significant cell destruction. The volume of blood products infused per minute appears to be the main factor associated with hyperkalaemia-related arrest in children, the use of peripheral venous catheters being preferred over central lines in the event there is a need for rapid replacement.4
Hyperkalaemia in liver transplantElevated preoperative plasma potassium concentration is the most important predictor for hyperkalaemia during surgery.23 Other factors include the presence of acidosis, osmolarity, insulin and catecholamine treatment, red blood cell transfusion, and the presence of renal failure.23
Controlling plasma potassium values before graft perfusion during liver transplant is a fundamental pillar in preventing intraoperative cardiac arrest. Cardiovascular collapse may be due to many causes hypothermia, acidosis, hypocalcaemia, and pulmonary embolism,40 hyperkalaemia being a frequent, avoidable cause.41,42
Clinical manifestationsPlasma potassium changes drive changes in cell membrane electric potential at rest, which manifest in the form of muscle weakness or paralysis, and cardiac conduction or repolarization abnormalities. The earliest most typical electrographic sign is an altered, more pronounced, T wave. This abnormality may progress to a prolonged PR interval, widening of the QRS complex, ventricular fibrillation and asystole after 7mEq/l.37,43 Treatment includes electric stabilization of the heart, potassium redistribution from plasma into the cells, and potassium clearance from the body.44 Dialysis removes 50–80mEq/l of potassium in 4h and may be used in exceptional cases such as patients with severe renal insufficiency or in simultaneous liver and kidney transplantation.45
ConclusionsThe concomitant presence of bleeding, ionic abnormalities and cardiac complications in the context of liver transplant surgery are frequent and potentially lethal.
Liver transplant patients often have one or several risk factors for developing intraoperative cardiac complications. Firstly, serum calcium, magnesium and potassium disorders are found frequently in cirrhotic patients and/or patients with portal hypertension, and they are more frequent in patients receiving diuretics. Secondly, obesity is associated with two liver diseases usually found in transplanted patients: hepatic steatosis and hepatocarcinoma,46 obesity being a risk factor for coronary vascular disease. Some of the diseases leading to liver transplant may be associated with heart disease, as is the case with haemochromatosis. Thirdly, and as was described in this paper, internal milieu abnormalities produced during bleeding and transfusion create a risk of citrate intoxication,47 with ventricular contractile failure22,48 and cardiac arrhythmias.
The presence of acute haemorrhage during liver transplantation is frequent and usually requires massive blood transfusions resulting in a risk of ion abnormalities. In turn, this results in a predisposition to cardiac failure, rhythm abnormalities, and even death. Preventive strategies are recommended in order to avoid those complications. They include:
- •
Selection of blood product volumes with shorter storage time, and red blood cell washing.4,49
- •
Systematic calcium administration if blood products are required during transplant surgery. It may be used in infusion at a dose of 5mg/kg/h for calcium chloride, or 15mg/kg/h for calcium gluconate, as long as there is significant bleeding requiring transfusion.
- •
Aggressive treatment for hypocalcaemia during transplant surgery: boluses of 5–10mg/kg of calcium chloride or 15–30mg/kg of calcium gluconate.
- •
Immediate correction of factors favouring citrate intoxication, namely, acidosis and hypotension.22,50
- •
Correction of hyperkalaemia (with diuretics, glucose insulin solutions, bicarbonate or beta-agonists), hypercalcaemia and hypomagnesaemia, which may be present in transplant patients already in the immediate preoperative period.
- •
Use of loop diuretics to avoid excess increase of preload when there is a need to administer large plasma volumes for the treatment of coagulopathy.
- •
Evaluate, together with the surgeons, the need for washing the graft with at least 500ml of fluid before reperfusion, in order to avoid rises in serum potassium that may lead to cardiac arrest.42
- •
Avoid reduced oxygen supply and increased oxygen consumption by the myocardium (hypotension, anaemia, tachycardia, and hypertension) in order to minimize ischaemia as an additional etiologic factor of arrhythmias, and heart failure.
Calcium and potassium abnormalities in patients with massive intraoperative haemorrhage during liver transplant surgery must be addressed actively and aggressively with a multidisciplinary approach.
FundingNone.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Rando K, Vázquez M, Cerviño G, Zunini G. Hipocalcemia, hiperpotasemia y hemorragia masiva en el trasplante de hígado. Rev Colomb Anestesiol. 2014;42:214–219.