There is no current consensus on the most suitable hip approach. However, there is a trend to reduce damage to soft tissue, which may have an influence on early outcomes. The SuperPath approach accesses the capsule maintaining integrity of the external rotators. The purpose of this study was to compare the SuperPath approach with the conventional posterior approach, in terms of early outcomes and radiological results.
Material and methodsA cohort of 30 patients operated using the SuperPath approach was prospectively matched for age, gender, body mass index and hip function with 60 patients operated using a conventional posterior approach. Clinical evaluation was performed by the Harris score, Merle d’Aubigné score, reduced Western Ontario and McMasters Universities (WOMAC), Short-Form 12 (SF12), IHOT-ADV and IHOT-12 questionnaires. Radiological evaluation was also performed.
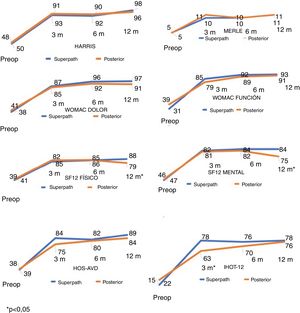
ResultsPreoperatively, no significant differences were detected between cohorts. Skin-to-skin operation time and blood loss was higher in the SuperPath cohort. Length of stay was similar between cohorts. Clinical evaluation improved significantly from the preoperative values to the 1-year follow-up. At 3 months the SuperPath cohort showed better results for IHOT-12, and at 12 months for SF. Radiologically, there were no differences between cohorts.
ConclusionThis prospective randomized study reveals that the learning curve for the SuperPath approach provides similar outcomes to the conventional posterior approach within the first year after surgery. The SuperPath approach was associated with longer skin-to-skin operation time, and greater blood loss.
En la actualidad no existe un consenso sobre cuál es el mejor abordaje de cadera para implantar una prótesis. Sin embargo, sí existe una tendencia a reducir al máximo la lesión sobre los tejidos periarticulares, lo cual puede influir en los resultados a corto plazo. El abordaje Superpath permite el acceso a la articulación manteniendo la integridad de los rotadores externos. El objetivo de nuestro estudio fue comparar los resultados a corto plazo del abordaje Superpath con el abordaje convencional posterior.
Materiales y métodosEstudio prospectivo de cohorte de 30 pacientes intervenidos mediante abordaje Superpath pareados por sexo, edad, índice de masa corporal y valoración clínica, con 60 pacientes intervenidos mediante abordaje convencional posterior. La valoración clínica se realizó mediante la escala de Harris, Merle d’Aubigné, Womac para el dolor y la función, SF-12 físico y mental, HOS-AVD e IHOT-12. Radiológicamente se determinó el ángulo de inclinación acetabular, alineación del vástago femoral e integración de los implantes.
ResultadosNo hubo diferencias significativas entre ambas cohortes con respecto a las variables preoperatorias. El tiempo quirúrgico, el descenso medio de Hb y Hto y la pérdida sanguínea fue mayor en la cohorte Superpath. La estancia hospitalaria fue similar. Los cuestionarios de valoración clínica mostraron incrementos significativos entre los valores preoperatorios y a los 12 meses en ambas cohortes. El cuestionario IHOT-12 a los 3 meses, el cuestionario SF-12 físico y el cuestionario SF-12 mental a los 12 meses, mostró mayores puntuaciones en la cohorte Superpath, siendo las diferencias significativas. Radiológicamente no se detectaron diferencias entre ambas cohortes.
ConclusionesLa curva de aprendizaje del abordaje Superpath presenta unos resultados clínicos y radiológicos similares al abordaje posterior durante el primer año tras la prótesis total de cadera. A destacar la necesidad de un mayor tiempo quirúrgico y un mayor sangrado en los pacientes.
Various approaches are described in the literature for implanting a total hip replacement (THR), without scientific evidence to support the superiority of one type of approach over the others, and with good long-term radiological and clinical results.1 The choice of the surgical approach to use is influenced by the surgeon's experience, patient-related factors and even geographical trends.2 Proper management of soft tissues parts needs to be one of the orthopaedic surgeon's objectives in order to achieve a satisfactory short-term result, regardless of the type of approach used for implanting a THR. Minimally invasive surgery (MIS) approaches were developed to achieve rapid recovery and reincorporation to daily living and work activities.3
In 2011, Dr Chow developed the MIS approach to THR using the supercapsular percutaneously assisted total hip (SuperPath®, MicroPort Orthopedics Inc., Arlington, TN, USA) approach,4 which combines elements of 2 posterior MIS approaches: the percutaneously assisted approach of Path® (MicroPort Orthopedics Inc., Arlington, TN, USA)5 and the superior capsular THR (SuperCap®, MicroPort Orthopedics Inc., Arlington, TN, USA) approach.6 Recent studies have shown satisfactory short-term results, correct radiological position of the prosthetic components, and a low rate of complications.7–10
The objective of our study was to determine the short-term results of a cohort of patients that had undergone a THR intervention using the SuperPath approach, and compare the results with a cohort of patients operated for THR using conventional posterior approach. Our working hypothesis was that the SuperPath approach would make it possible to obtain results similar to those of the posterior approach.
Material and methodsStudy designIn January 2016, the SuperPath approach was introduced in our service. A prospective, observational analytic cohort study was designed, to establish the short-term results of the learning curve for the SuperPath approach.
During the period between January 2016 and December 2017, patients with a diagnosis of hip arthrosis and indication for cementless hip replacement surgery. Minimum follow-up time for inclusion in this study was 6 months. The patients were divided into 2 cohorts depending on the type of approach used: SuperPath cohort or posterior cohort. Patients with femoral or acetabulares defects, acetabular protrusion, femoral fracture or neurological condition with impaired gait were excluded. To compare the results, for each patient in the SuperPath cohort, 2 consecutive patients were selected from the posterior cohort with an age ±5 years, the same sex, BMI±3kg/m2 and Harris Scale score ±5 points. All patients were asked for their informed consent to be included in the study.
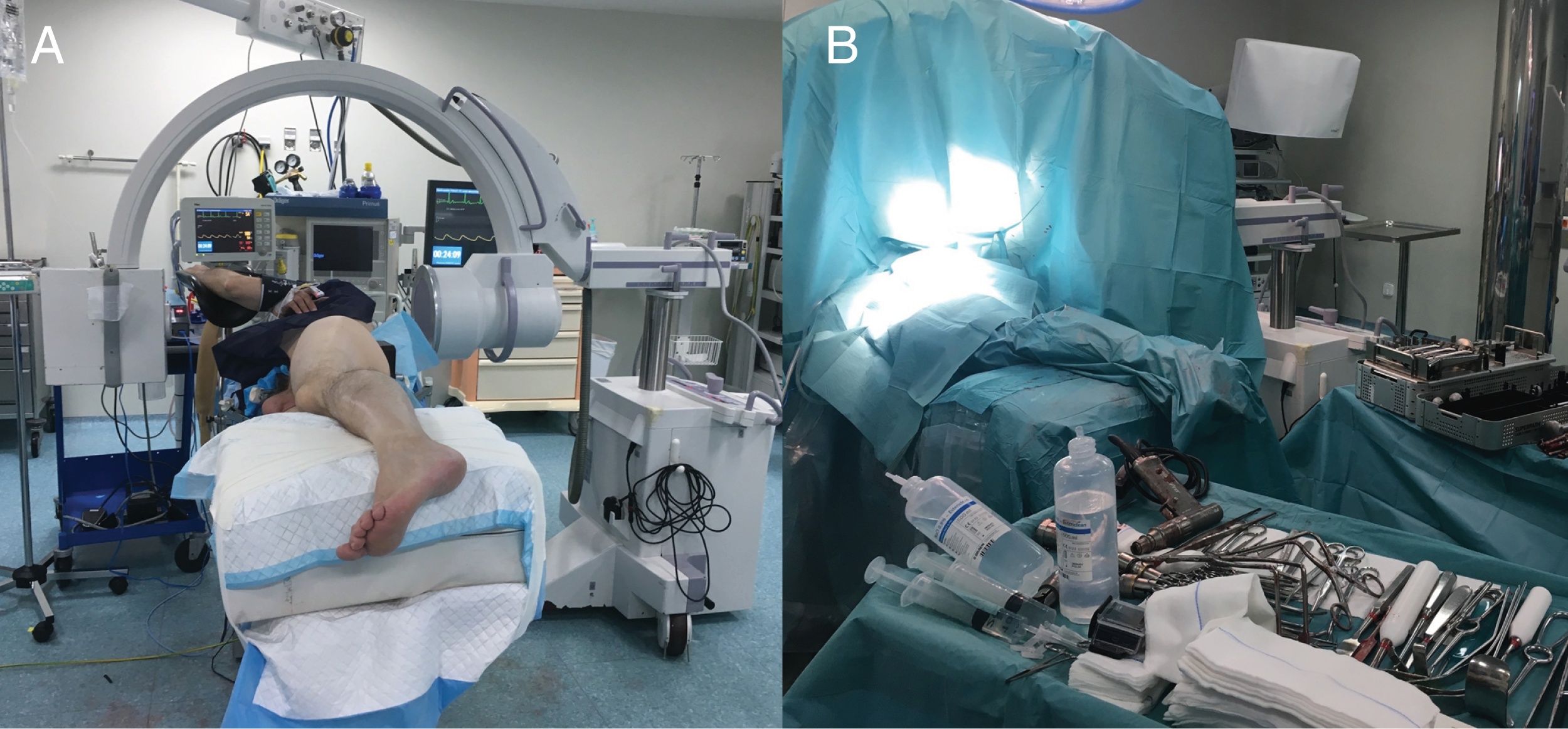
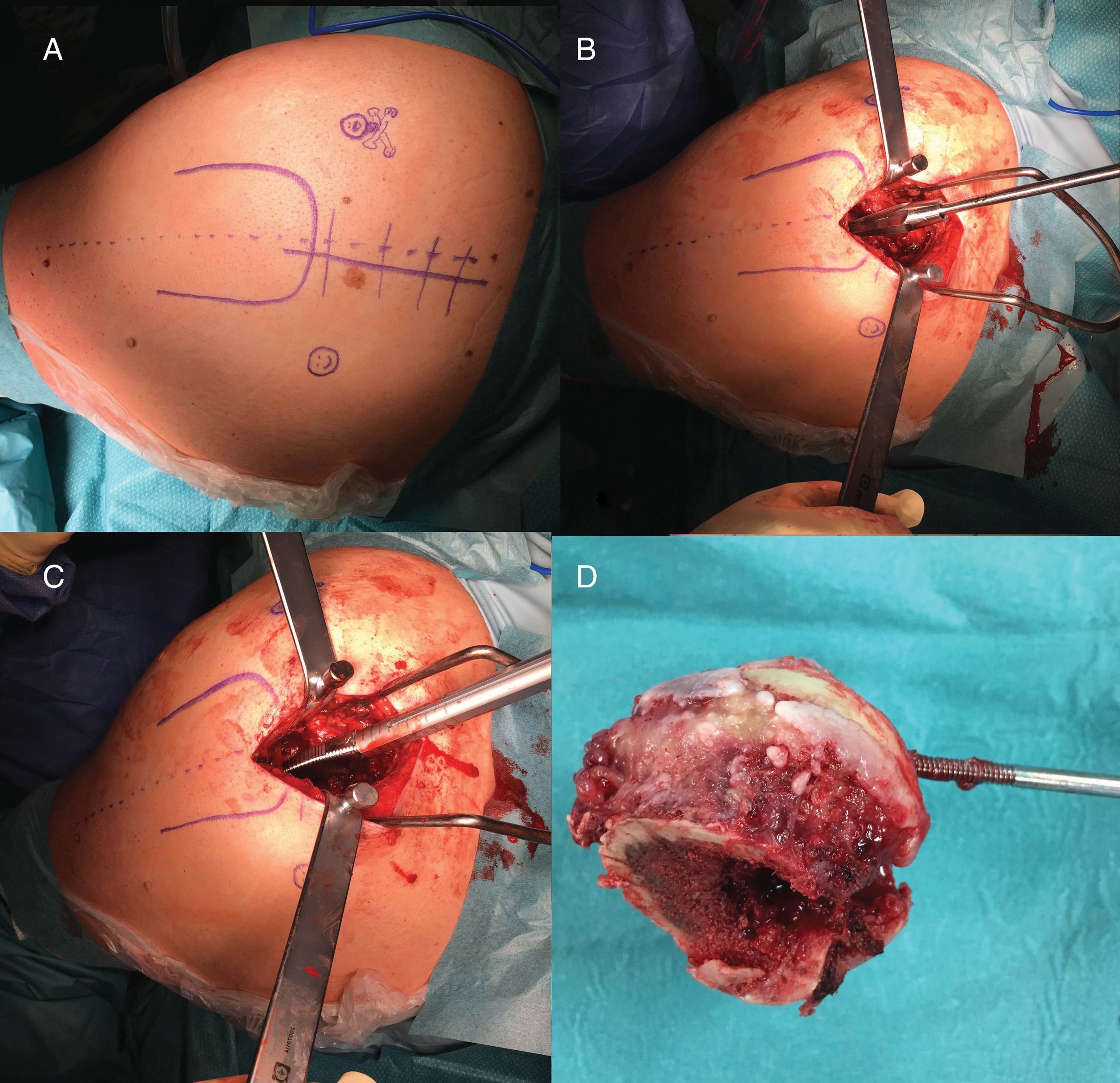
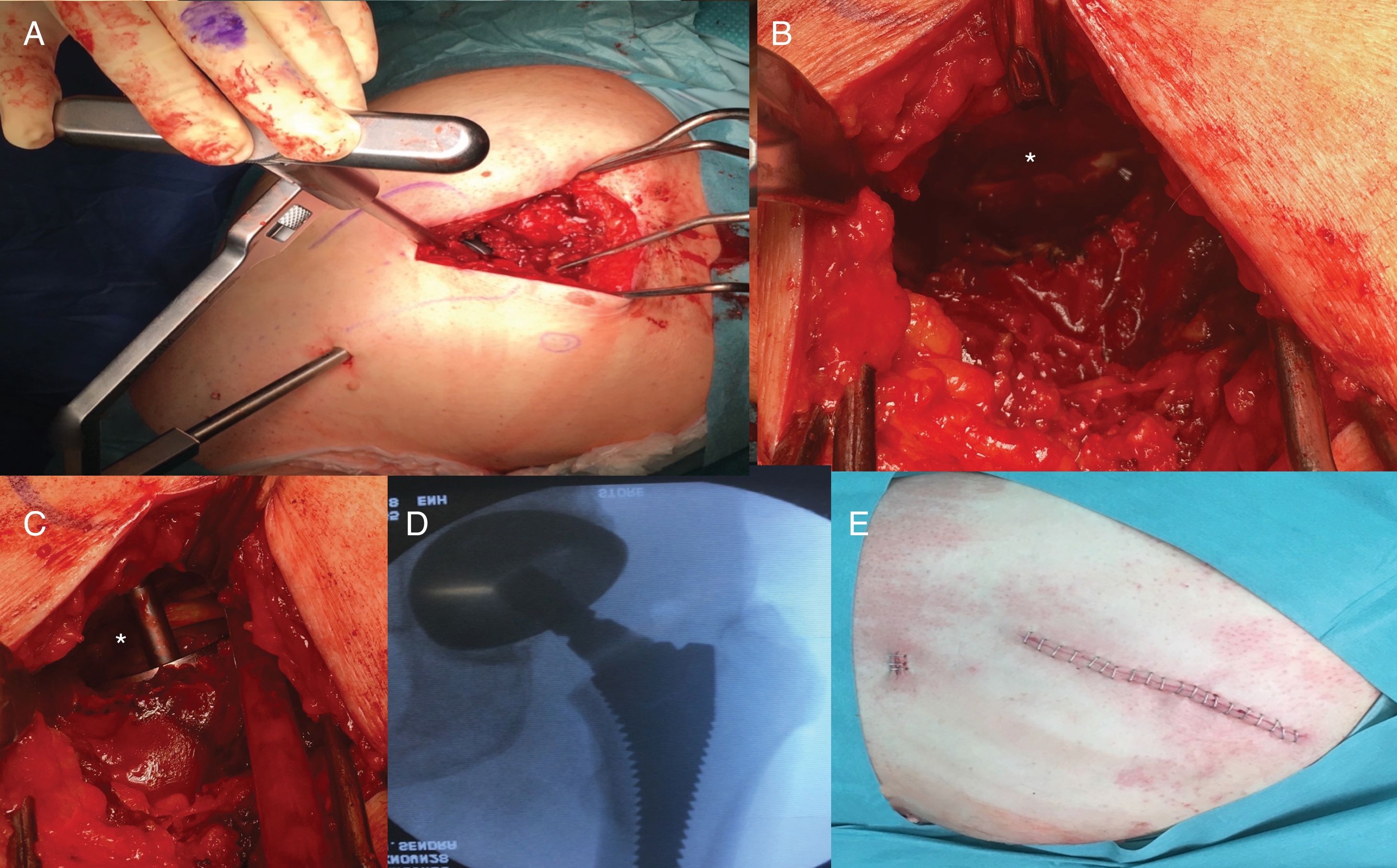
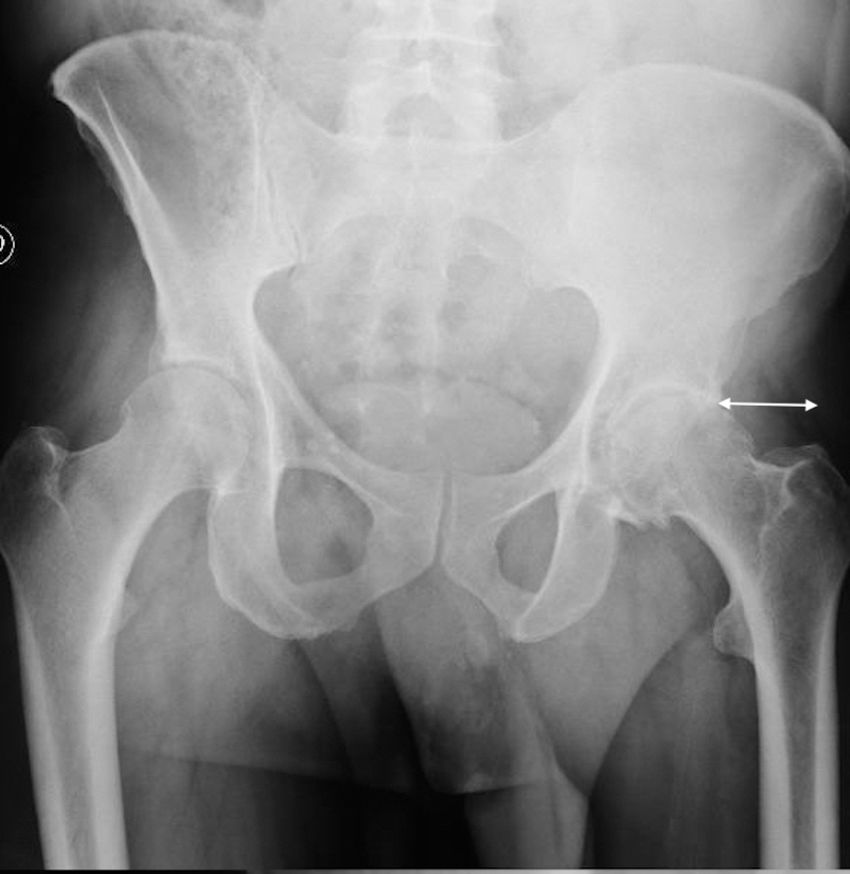
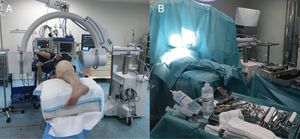
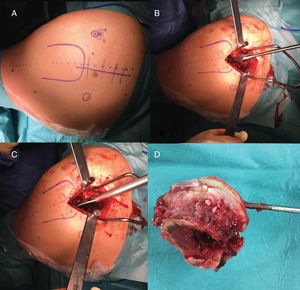
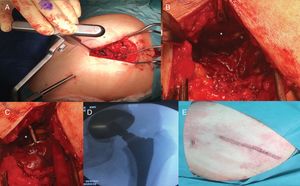
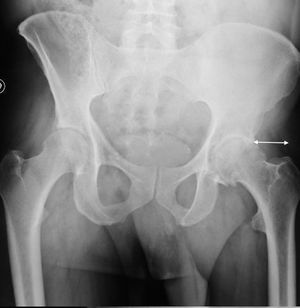
Surgical techniqueThe SuperPath approach was carried out using a conventional operating table for hip replacement surgery, with the patient in a lateral recumbent position with the affected hip having a flexion of 50° and internal rotation of 20°, and the lower limb supported on a cushion to keep the leg elevated above the surgical table (Fig. 1). The incision commenced at the upper border of the greater trochanter and continued proximally in line with the femoral axis. The upper part of the hip joint capsule was accessed through the interval between the gluteus minimum and piriformis muscles, keeping the external rotators intact. Femoral preparation began without dislocating the hip through the base of the femoral neck, first with reamers and then with rasps. Using the cervical part of the rasp as a femoral cut template, the femoral osteotomy was performed and the femoral head was removed (Fig. 2). A distal and posterior portal was made with an external guide to access the acetabulum. Through the portal, acetabular reaming, impaction of the acetabular component and supplementary fixation with screws, if necessary, were performed. The transverse acetabular ligament was used as a guide to orientate the acetabular component. Next, the test insert, head and neck were placed; the prosthesis was reduced and its stability, lack of impact between components, presence of leg length discrepancies and mobility were checked. Intraoperative radioscopy allowed verifying the correct implant position (Fig. 3). The test head and neck required decoupling with a special device. The definitive components were implanted based on the test performed. The capsule was closed and the upper planes, approximated.2 The surgical team chose to indicate the SuperPath as long as the distance between the lateral area of the acetabular rim and the apex of the greater trochanter was greater than 2.5cm (Fig. 4).
Left hip. (A) Establishment of distal and posterior portal for acetabular access. (B) Visualisation of the transverse acetabular ligament (asterisk). (C) Acetabular reaming perpendicular to transverse acetabular ligament (asterisk) for proper cup orientation. (D) Intraoperative radioscopy to verify the optimal implant position. (E) Surgical site wound.
The conventional posterior hip approach was carried out using a conventional operating table for hip prostheses with the patient placed in a lateral recumbent position. A longitudinal incision was made over the greater trochanter, the fascia lata was sectioned, the gluteus maximus fibres were dissected, and muscular-capsular detachment of the external rotators was performed. Next, the femoral head was dislocated, osteotomy of the femoral neck was performed and the hip prosthesis was implanted.11
The surgical team and the epidural anaesthesia procedure were identical in all the cases. The cementless prosthesis model used was the Profemur L stem and Procotyle® cup (MicroPort Orthopedics Inc., Arlington, TN, USA) in the patients operated using the SuperPath approach; and stem Accolade and Trident® cup (Stryker, Kalamazoo, MI, USA) in the patients operated using the conventional posterior approach. For the 46 and 48 cups, 28mm size head was used; for the 50 and 52 cups, a 32mm head; and for the 54 or larger cups, a 36mm head was used. Periarticular analgesic (ketorolac 60mg+ropivacaine 200mg, in 22ml of normal saline [NS]) was injected, and tranexamic acid (2g in 25ml of NS) was administered in an inter-articular fashion after closing the fascia of the gluteus maximus or the fascia lata. No postoperative drains were used.
Postoperative managementParacetamol and nonsteroidal anti-inflammatory drugs (NSAIDS) were administered in pain-relieving continuous infusion pumps on the ward. At 24h, the pump was removed, and oral paracetamol and dexketoprofen and intravenous metamizol were administered every 8h. Walking with crutches or a walker was authorised after assessing the radiological control and postoperative laboratory results. The patient was told not to perform any activities that increased pain in the operated hip. The suture was removed after 2 postoperative weeks.
EvaluationsHip function outcomes were assessed preoperatively and at 3 months, 6 months and 1 year using the Harris Hip Scale,12 the Merle d’Aubigné Hip Score,13 the Western Ontario and MacMaster Universities (WOMAC) osteoarthritis index for pain and for function,14 the 12-item Short Form Health Survey (SF-12) physical and mental scales,15 the Spanish Activities of Daily Living subscale of the Hip Outcome Score (HOS-AVD)16 and the International Hip Outcome Tool, short version (iHOT-12)17 scales.
The variables recorded were as follows: epidemiological, ASA degree, haemoglobin (Hb) and pre- and postoperative hematocrit (Hto) at 24h, operative time, friction torque, size of the femoral head, blood loss,18,19 need for autologous blood transfusion, hospital stay, complications and readmissions. The angle of acetabular inclination, femoral stem alignment, and implant integration were determined by radiological control at 3 months after the surgery.20,21
Statistical analysisStatistical analysis was carried out using the SPSS® version 18.0 program (SPSS Inc., Chicago, USA). Significance was set to P values equal to or less than 0.05. The Kolmogorov–Smirnov test was used to determine the normal distribution. For comparisons, univariate analyses were used for categorical variables by the Chi-square test in variables with normal distribution and the Mantel–Haenszel test for non-parametric data, and Student's t-test or the Mann–Whitney U test in continuous variables.
ResultsGeneral resultsDuring the study period, 172 THR were implanted in our service. Of these, 30 were handled using the SuperPath approach and 113 by the posterior approach. From the posterior approach cohort, 60 prostheses that fulfilled the criteria established were selected for comparing both cohorts.
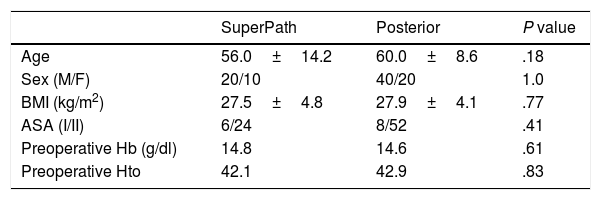
The general preoperative characteristics of the 2 cohorts are presented in Table 1. There are no statistically significant differences between the groups (P>.05).
General data on both cohorts.
| SuperPath | Posterior | P value | |
|---|---|---|---|
| Age | 56.0±14.2 | 60.0±8.6 | .18 |
| Sex (M/F) | 20/10 | 40/20 | 1.0 |
| BMI (kg/m2) | 27.5±4.8 | 27.9±4.1 | .77 |
| ASA (I/II) | 6/24 | 8/52 | .41 |
| Preoperative Hb (g/dl) | 14.8 | 14.6 | .61 |
| Preoperative Hto | 42.1 | 42.9 | .83 |
BMI: body mass index; Hb: haemoglobin; Hto: hematocrit.
Mean±standard deviation.
The friction torque used in the SuperPath cohort was the ceramic-ceramic in 5 cases, ceramic-polyethylene in 17 cases and the metal-polyethylene in 8 cases; and in the posterior cohort, in 5, 44 and 11 cases, respectively, without statistically significant differences (P=.25). The size of the femoral head in the SuperPath cohort was 28mm in 8 cases, 32mm in 15 cases and 36mm in 7 cases; and in the posterior cohort in 10, 28 and 22 cases, respectively, without statistically significant differences (P=.34).
Mean operative time was 69.5±7.1min in the SuperPath cohort and 56.1±5.2min in the posterior cohort. There were no statistically significant differences (P=.001).
Mean postoperative Hb dropped to 11.2±1.3g/dl at 24h in the SuperPath cohort, and to 12.5±1.5g/dl in the posterior cohort, without statistically significant differences (P=.07). However, the mean Hb decrease was greater in the SuperPath cohort, with 3.4±1.0 points, compared with 2.5±0.8 points in the posterior cohort; the differences were statistically significant (P=.04).
Mean postoperative Hto dropped to 32.1±3.3 in the SuperPath cohort and to 35.6±4.8 in the posterior cohort; the differences were significant (P=.03). The mean Hto decrease was also greater in the SuperPath cohort, with 10.3±3.2 points, compared with 7.7±3.0 points in the posterior cohort, with statistically significant differences (P=.04).
Blood loss was greater in the SuperPath cohort, with a mean of 977.85±285.1ml; the mean blood loss was 752.46±299.3ml in the posterior cohort. The differences were also statistically significant (P=.03). None of the patients required autologous blood transfusion.
The mean hospital stay was similar in both cohorts: 2.2 days in the SuperPath cohort and 2.4 in the posterior cohort (P=.23).
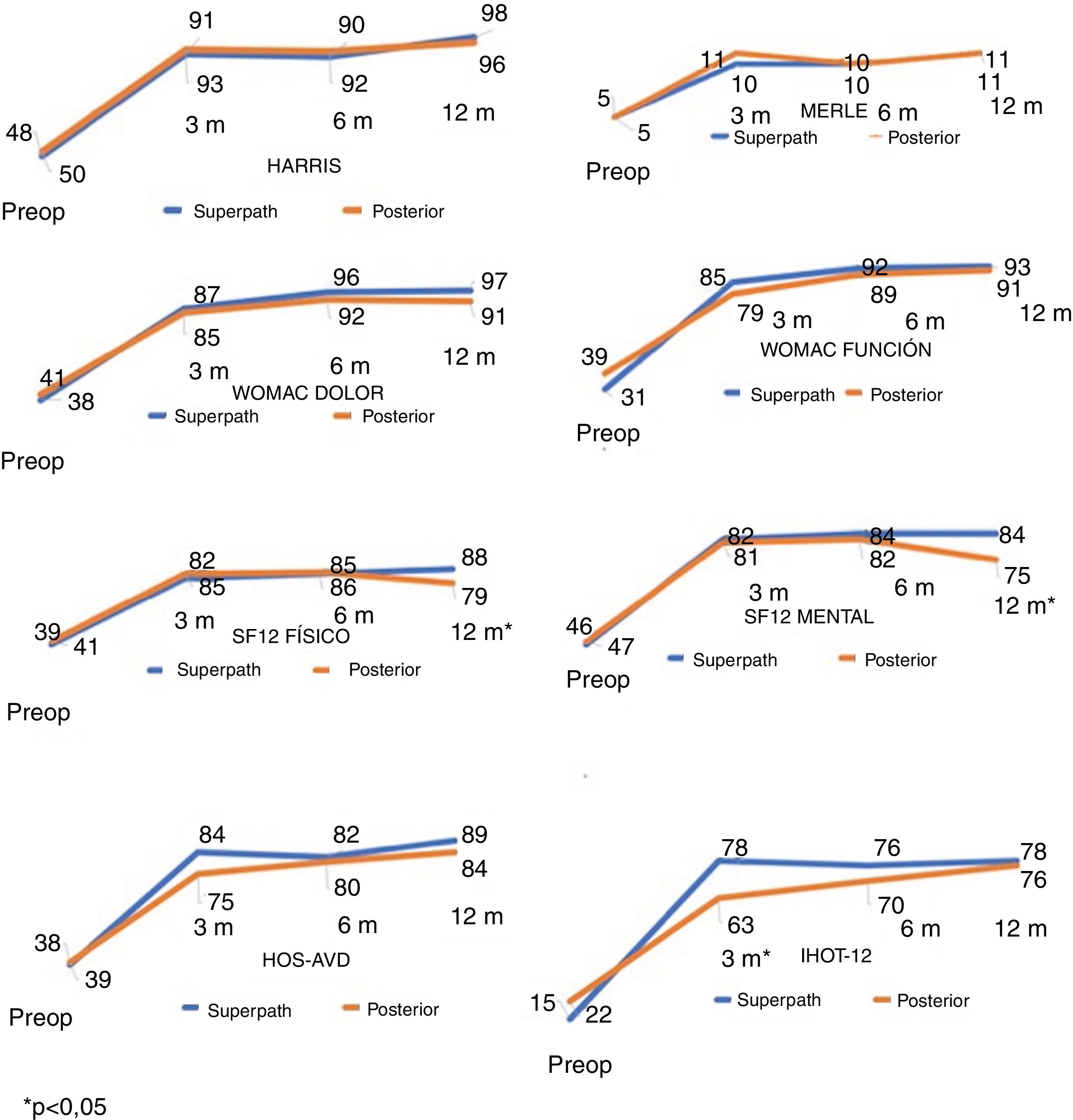
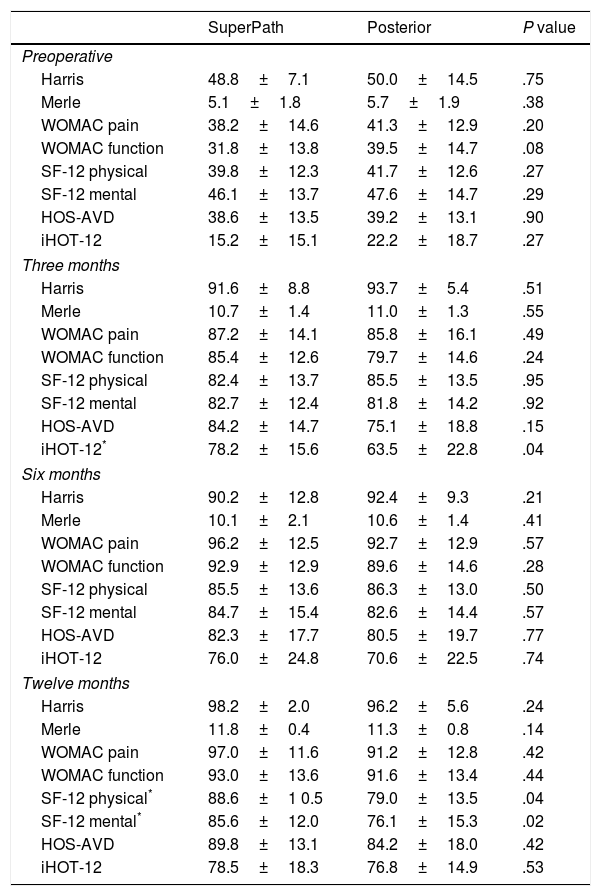
Clinical and radiological assessmentThe hip function surveys showed significant increases between the preoperative values and those at 12 months in both cohorts, with statistically significant differences (P<.001). Patient scores were generally similar between the 2 cohorts during follow-up. However, the exceptions were the iHOT-12 questionnaire at 3 months, the SF-12 physical survey and the SF-12 mental survey at 12 months, with greater scores in the SuperPath cohort; the differences were statistically significant (P<.05) (Table 2 and Fig. 5).
Clinical assessment tool results during the follow-up of the 2 cohorts.
| SuperPath | Posterior | P value | |
|---|---|---|---|
| Preoperative | |||
| Harris | 48.8±7.1 | 50.0±14.5 | .75 |
| Merle | 5.1±1.8 | 5.7±1.9 | .38 |
| WOMAC pain | 38.2±14.6 | 41.3±12.9 | .20 |
| WOMAC function | 31.8±13.8 | 39.5±14.7 | .08 |
| SF-12 physical | 39.8±12.3 | 41.7±12.6 | .27 |
| SF-12 mental | 46.1±13.7 | 47.6±14.7 | .29 |
| HOS-AVD | 38.6±13.5 | 39.2±13.1 | .90 |
| iHOT-12 | 15.2±15.1 | 22.2±18.7 | .27 |
| Three months | |||
| Harris | 91.6±8.8 | 93.7±5.4 | .51 |
| Merle | 10.7±1.4 | 11.0±1.3 | .55 |
| WOMAC pain | 87.2±14.1 | 85.8±16.1 | .49 |
| WOMAC function | 85.4±12.6 | 79.7±14.6 | .24 |
| SF-12 physical | 82.4±13.7 | 85.5±13.5 | .95 |
| SF-12 mental | 82.7±12.4 | 81.8±14.2 | .92 |
| HOS-AVD | 84.2±14.7 | 75.1±18.8 | .15 |
| iHOT-12* | 78.2±15.6 | 63.5±22.8 | .04 |
| Six months | |||
| Harris | 90.2±12.8 | 92.4±9.3 | .21 |
| Merle | 10.1±2.1 | 10.6±1.4 | .41 |
| WOMAC pain | 96.2±12.5 | 92.7±12.9 | .57 |
| WOMAC function | 92.9±12.9 | 89.6±14.6 | .28 |
| SF-12 physical | 85.5±13.6 | 86.3±13.0 | .50 |
| SF-12 mental | 84.7±15.4 | 82.6±14.4 | .57 |
| HOS-AVD | 82.3±17.7 | 80.5±19.7 | .77 |
| iHOT-12 | 76.0±24.8 | 70.6±22.5 | .74 |
| Twelve months | |||
| Harris | 98.2±2.0 | 96.2±5.6 | .24 |
| Merle | 11.8±0.4 | 11.3±0.8 | .14 |
| WOMAC pain | 97.0±11.6 | 91.2±12.8 | .42 |
| WOMAC function | 93.0±13.6 | 91.6±13.4 | .44 |
| SF-12 physical* | 88.6±1 0.5 | 79.0±13.5 | .04 |
| SF-12 mental* | 85.6±12.0 | 76.1±15.3 | .02 |
| HOS-AVD | 89.8±13.1 | 84.2±18.0 | .42 |
| iHOT-12 | 78.5±18.3 | 76.8±14.9 | .53 |
Harris: Harris Hip Scale; Merle d’Aubigné: HOS-AVD: Spanish version of the Hip Outcome Score, Activities of Daily Living subscale; iHOT-12: International Hip Outcome Tool, short version; Merle d’Aubigné Hip Score; SF-12: 12-item Short Form Health Survey scales; WOMAC: Western Ontario and MacMaster Universities Osteoarthritis Index.
Mean±standard deviation.
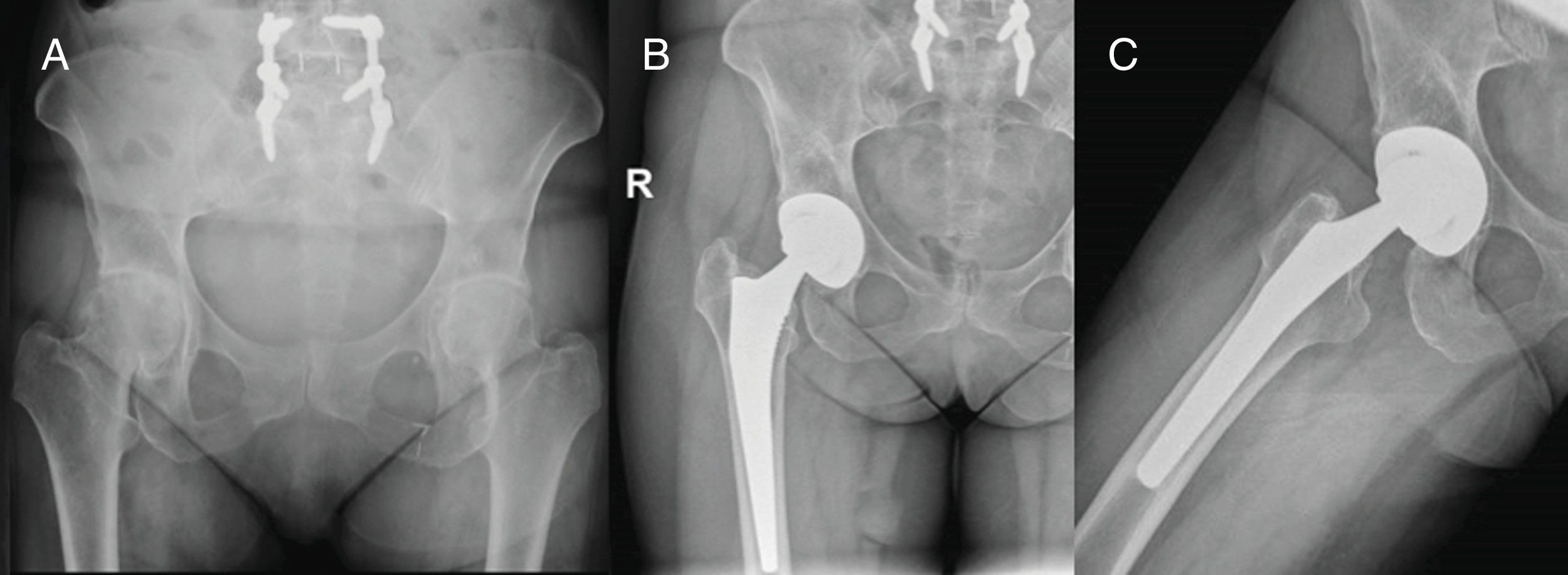
The mean angle of acetabular inclination was 47.6° in the SuperPath cohort and 45.9° in the posterior cohort; these differences were not statistically significant (P=.41). All the femoral stems were implanted in neutral position. There were no recorded cases of mobilisation of prosthetic components (Fig. 6).
ComplicationsA 77-year-old patient in the SuperPath cohort under treatment with teriparatide for osteoporosis presented fracture of the acetabular background during the impaction of the cup component. The external rotators were removed and the approach was converted to a conventional posterior approach. Given that osseous acetabular circumferential stability existed, a cemented stem was implanted. Evolution at 12 months’ of follow-up was satisfactory, with a Harris Scale score of 82 points and Merle scale score of 10 points.
A 68-year-old patient in the posterior cohort was readmitted at 14 days postoperatively for periprosthetic infection, requiring debridement and replacement of mobile components. Evolution was good at 1 year of follow-up, with a Harris Scale score of 84 points and a Merle scale score of 10 points.
A 52-year-old patient in the SuperPath cohort presented progressive pain in the buttock on the affected side at 6 months’ follow-up. Complete radiolucency of the stem without implant mobilisation was detected in the radiological control, and the patient is now pending surgical revision.
There were no cases of prosthetic dislocations or thromboembolic complications during follow-up in either cohort.
DiscussionOur hip function results during the learning curve of the SuperPath approach have been similar to the short-term results of the posterior approach in the surveys used, except for the iHOT-12 questionnaire at 3 months, and the SF-12 physical and SF-12 mental at 12 months, with a higher score and a significant difference in the SuperPath cohort. In another comparative study on 46 hips operated using the SuperPath approach and 46 hips with the posterior approach, Xie et al.10 report significantly greater values at 1 week, 1 month and 3 months in the Harris Scale and Barthel Index; a lower pain level on a visual analogue scale (VAS); shorter time to get up from a chair, walk 3m and return to the chair (Timed Up and Go [TUG] test); and less time to go up and come down 12 steps (Timed Stair Climb [TSC] test); and there were no differences at 12 months. Our findings coincide in that there are statistically significant differences in 1 of the surveys at 3 months, but not in detecting differences at 12 months. Preserving the external rotators might facilitate a quicker functional recovery of the operated hip, as a greater score at 3 months in the iHOT-12 survey shows. However, it would be logical for functional outcomes to be similar with the follow-up. The statistically significant differences in the SF-12 questionnaire at 12 months are produced by a decrease in the values in the posterior cohort, while they remain the same in the SuperPath cohort. A longer patient follow-up would allow us to confirm whether the differences increase or decrease.
The posterior approach is the one most used in prosthetic hip surgery, allowing wide exposure of the joint through muscular-capsular detachment of the external rotators, facilitating implant insertion and preserving the gluteal musculature. The SuperPath approach is close to the posterior approach, allowing preservation of the external rotators, respecting the iliotibial band, and requiring specific instruments; but it is performed on a conventional operating table for orthopaedic surgery and allows changing to the posterior approach if necessary.
Preserving the external rotators should be an advantage in reducing the incidence of prosthetic dislocation. Xie et al.10 report a dislocation in their series of 46 hips operated using the SuperPath approach (2.1%). Gofton et al.7 publish 4 dislocations in 479 hips operated (.8%). Rasuli and Gofton9 indicate a dislocation in 50 hips (2%). In our series, there were no cases of dislocation. A future line of research might be the use of the SuperPath approach in patients with a greater risk of prosthetic dislocation, such as elderly patients with subcapital fracture of the femur. Han et al.22 share the outcomes of 2 cohorts of patients older than 65 years with subcapital fracture of the femur and associated neurological disorder that were operated using the posterior approach preserving the rotators compared with the conventional posterior approach: there are 0% of dislocations in the group in which the rotators are preserved compared with 7.7% in the group in which the conventional approach is used.
Blood loss in prosthetic hip surgery is related to the bleeding at the level of the osteotomy and the medullary cavity. Smaller soft tissue dissection in the SuperPath approach should be linked to less bleeding and fewer transfusions. Gofton et al.7 report a 3.3% need for transfusion in their series of 479 hips; Rasuli and Gofton,9 4% in 50 hips; Chow and Fitch,23 1.9% in 419 hips; and Cardenas-Nylander,2 9.5% in 21 hips. Xie et al.10 indicate 4.3% compared with 11% in the posterior approach cohort, and a blood loss of 303.6ml compared with 326.4ml; the differences were not statistically significant. In our series, the decreases in Hb and Hto, and the blood loss were significantly greater in the SuperPath cohort, possibly because of a greater operative time. In spite of this, no patient required autologous blood transfusion.
The correct position of the prosthetic components is essential for medium- and long-term implant survival. The smaller exposure in el SuperPath approach might, theoretically, be associated with improper implant position. Della Torre et al.8 report an acetabular inclination angle of 40.1° in their series of 66 hips, with properly implanted femoral components. Carlomagno2 indicate an acetabular angle of 44.0°, while that of Rasuli and Gofton9 is 39.0°. Xie et al.10 publish an acetabular angle of 43.6° and 93.4% of neutral femoral alignment, without statistically significant differences with the posterior cohort. In our series, the acetabular angle was 47.6°, with no statistically significant differences with the posterior cohort, and the alignment of the femoral component was neutral in all the cases. To determine acetabular orientation, using the transverse acetabular ligament as a guide is recommended, as well as using the intraoperative radioscopy to align the femoral stem correctly.
Some authors report a shorter hospital stay in patients operated using the SuperPath approach. Gofton et al.7 indicate a mean admission of 1.6 days in their series, with 50% fewer days than the data published by the National Agency for Healthcare Research and Quality. Rasuli and Gofton9 report a mean stay of 2.2 days. Carlomagno2 indicates a mean stay of 4.7 days, with a mean reduction of 1.4 days compared with a posterior approach cohort. And Xie et al.10 publish a mean hospital stay of 8.3 days, as against the 11.4 days for the posterior cohort. In our series, the mean stay in the SuperPath cohort was 2.2 days, with a mean reduction of 0.2 days, although the differences were not statistically significant.
Operative time usually increases in the learning curve for a new surgical approach. Chow and Fitch23 report a mean operative time of 142.7min, compared with the 102.8min indicated by Carlomagno.2 Xie et al.10 relate a mean time of 103.6min, similar to the 106.5min in the posterior cohort. With 69.5min, our operative time was less than that published in the literature, but significantly longer than in the posterior cohort (56.1min). This fact might have played a role in the greater bleeding in the SuperPath cohort, even though it is an approach that preserves the soft tissues better.
Periprosthetic fracture is another problem associated with using the SuperPath approach. Chow and Fitch4 report 3 intraoperative fractures: 2 femoral and 1 acetabular. The 3 occurred in patients with a prior history of osteoporosis. Carlomagno2 indicates 1 intraoperative femoral fracture. Xie et al.10 do not communicate this complication. In our series, we had 1 intraoperative acetabular fracture, likewise in a patient with a history of osteoporosis, and possibly due to excessive acetabular reaming.
The need for hospital readmission during the first month after the THR raises the cost of the healthcare process by up to 40%.7 Two recent studies indicate a lower readmission rate in the patients operated using the SuperPath approach. In their multicentre study on 479 THR implanted using the SuperPath approach, Gofton et al.7 report 2.3% of readmissions during the first 30 days after surgery, which represents a reduction of 1.9% over the national mean in the USA. And Chow and Fitch23 indicate, in a comparative study between 419 THR using the SuperPath approach and 1673 THR implanted using other approaches, 0.4% of readmissions as against 2.9%. In our series, no readmissions were registered during the first month after surgery in the SuperPath cohort.
Our study presents limitations as far as the number of cases in the SuperPath approach group, although it should also be remembered that the learning curve for the technique is considered in the series. There is also the fact that different stem-stem models were used in the 2 cohorts, although we consider that they are similar cementless prosthesis models without any differences in friction torques or femoral head size used. Another limitation is the surgeon-dependent distribution of the patients into the SuperPath or posterior approaches. However, the study also has various strengths. It is a cohort-paired prospective analytical study, with 6 evaluation tools (2 that were objective and 4, subjective) applied to all the patients. In addition, no patients were lost in the follow-up.
ConclusionsThe learning curve for the SuperPath approach presents radiological and clinical results similar to those of the posterior approach during the first year after the THR. It should be pointed out that the SuperPath approach requires a longer skin-to-skin operative time and has greater patient bleeding.
Level of evidenceLevel of evidence III.
Conflict of interestsThe authors have no conflicts of interest to declare.
Please cite this article as: Más Martínez J, Sanz-Reig J, Morales-Santías M, Bustamante Suarez de Puga D, Verdu Roman C, Martinez Gimenez E. Estudio de cohortes comparativo del abordaje Superpath con abordaje convencional posterior en cirugía protésica primaria de cadera no cementada: curva de aprendizaje y resultados a corto plazo. Rev Esp Cir Ortop Traumatol. 2019;63:346–354.