Imaging biomarkers define objective characteristics extracted from medical images that are related to normal biological processes, diseases, or the response to treatment.
To develop an imaging biomarker, it is necessary to carry out a series of steps to validate its relation with the reality studied and to check its clinical and technical validity. This process includes defining tests for the concepts and mechanisms; obtaining standardized and optimized anatomic, functional, and molecular images; analyzing the data with computer models; displaying data appropriately; obtaining the appropriate statistic measures; and conducting tests on the principle, efficacy, and effectiveness.
In this article, we aim to explain the steps that must be established to enable biomarkers to be correctly applied, from their theoretical conception to their clinical implementation. To this end, we use the evaluation of angiogenesis in articular cartilage as an example.
Los biomarcadores de imagen definen características objetivas extraídas de las imágenes médicas, relacionadas con procesos biológicos normales, enfermedades o respuestas terapéuticas.
Para desarrollar un biomarcador de imagen es necesario realizar una serie de pasos destinados a validar su relación con la realidad estudiada y controlar su validez, tanto clínica como técnica. Este proceso incluye la definición de pruebas de concepto y de mecanismo; la adquisición estandarizada y optimizada de imágenes anatómicas, funcionales y moleculares; el análisis de los datos mediante modelos computacionales; la visualización adecuada de los resultados; la obtención de medidas estadísticas apropiadas; y la realización de pruebas de principio, eficacia y efectividad.
Nuestro objetivo en este trabajo es mostrar los pasos que deben establecerse para aplicar adecuadamente los biomarcadores de imagen, desde su concepción teórica hasta su implantación asistencial, en un entorno hospitalario. Para ello se planteará como ejemplo la valoración de la angiogénesis del cartílago articular.
A radiologist's report indicating the presence of atrophy on a computed tomography (CT) scan is not the same as a report saying that there is a brain volume loss of 24%, considering the patient's age, more marked in the right middle temporal sulcus. Assuring that there is a tumor in the liver is not the same as knowing that its degree of biological aggressiveness is very high and its response to treatment limited. Saying that no bone abnormalities are observed on magnetic resonance (MR) imaging is not the same as saying that there is a reduction of 34% in the elastic index of the trabecular bone and that the patient has osteoporosis and is at risk for bone fracture. Where does this information come from? How can the radiologist attain this knowledge? What should we do to incorporate it into our clinical practice?
The development of all-digital medical imaging techniques produces high-quality images and generates a wide range of information contained in these images. Simultaneously, the massive expansion of computing has further improved the images used to diagnose and guide the treatment of many diseases. In addition, imaging biomarkers are being developed as an expression of this synergy between digital images and their computational processing. These innovative developments extract from the medical images quantitative information that cannot be visually detected or measured in the source images. The use of imaging biomarkers will allow us, radiologists, to change the concept and the process mapping of our work, opening up the traditional medical imaging to other fields such as engineering and physics.
This multidisciplinary interaction, which falls within the field of Biomedical Engineering, is developing new acquisition techniques and is implementing a variety of mathematical models able to simulate the biological and physiological reality, allowing for accurate and reproducible measurements of patients’ condition. Models based on signals or images are symbolic representations of a real process, defined as a series of relations or equations between the different variables that participate in such reality. Because of their huge potential, imaging biomarkers have emerged as one of the most active research fields, allowing for the visualization and measurement of physiological and biological processes using 3D modeling of a region of interest in a specific patient.
An imaging biomarker can be defined as a characteristic extracted from the images of an individual that can be objectively measured and acts as an indicator of a normal biological process, a disease, or a response to a therapeutic intervention.1 From simple size or shape measurements to complex computational models, biomarkers have proved useful to provide complementary information to the traditional radiological diagnosis to determine the presence of a disorder or lesion; measure its biological condition; define its natural history and progression; stratify phenotypic abnormalities and assess the treatment response. In theory, biomarkers may be obtained from any imaging modality but, among all the available techniques, MRI stands out for its great versatility in the study of different types of tissues and processes.
Imaging biomarkers have two major advantages. First, they represent quantitative variables that characterize and measure different parameters, obtained from medical images that are relevant to a specific disease. Second, parametric images allow us to analyze the spatial distribution of the biomarker in the sample observed through its visual representation. These images are generated to provide a graphical representation of the values of each biomarker or parameter calculated on the basis of original image post-processing.
The aim of this work is to present the steps required for an appropriate implementation of imaging biomarkers, from their theoretical conception to their clinical implementation in a hospital setting.
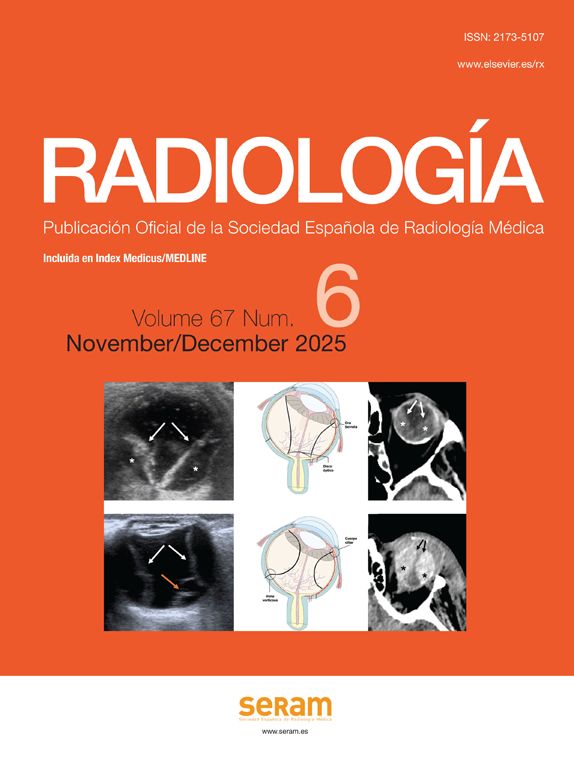
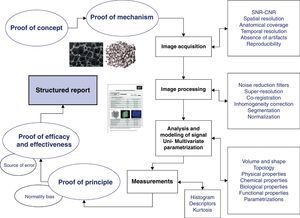
Initial development of an imaging biomarkerThe process required to integrate an imaging biomarker into both the clinical practice and a clinical trial is complex and must meet the criteria of conceptual consistency, technical reproducibility, sensitivity and specificity. The definition of the problem, acquisition of source images, analytical methodology and biomarker extraction using computational models and measurement instruments are essential aspects required to study a specific disease. The development of a biomarker involves validating its relationship with the objective reality (structural, physiological, biological or molecular) and monitoring its technical validity. The path to biomarker development, expansion and subsequent implementation involves a number of consecutive steps described below (Fig. 1).2
The first step is to define the reasons why we want to measure a specific aspect of the disease. This Proof of Concept must demonstrate that a specific biological process, seen as a cause and effect chain, may be studied using the imaging and computational techniques available.
Once the objective has been defined, the relation between the biomarker, the parameter analyzed and the disease under study must be established. In this respect, the Proof of Mechanism has to demonstrate an interaction between the biomarker and the concept, focusing on the effect (in magnitude and direction) that a specific disease or a treatment has on the biomarker. For example, the proof of mechanism serves to determine whether the relation between the severity of a disease and a specific biomarker is directly or inversely proportional, non-linear, exponential, among others.
Acquisition and analysis of an imaging biomarkerAppropriate images are essential for the extraction of useful biomarkers. For this reason, the definition of the method and the technique used for image acquisition are the most important steps. Irrespective of the technique used (radiography, ultrasound, CT, MRI, SPECT−Single Photon Emission Computed Tomography−or PET−Positro Emission Tomography), several issues must be taken into consideration. Image quality, measured in terms of signal-to-noise ratio (SNR) and contrast-to-noise ratio (CNR), must be optimized to detect the parameter under examination. This is a critical step to properly analyze the signal of the object of interest and its changes, facilitating the implementation of the most appropriate computational tools.
As for anatomical coverage, the target organ must be studied with sufficient spatial resolution, defined by voxel size. Both the resolution and anatomical coverage must be adjusted to the problem under investigation and the expected size of the lesion. In order to ensure reproducibility, the images must be free of artifacts that may degrade or disturb the signal and must maintain their quality over time (SNR, CNR) without modifying the acquisition parameters. Moreover, in functional and dynamic studies, it is important to ensure the best compromise between spatial and temporal resolution requirements (frequency of image acquisition of one volume), in order to observe the biological processes in detail.
Regular quality control tests (advisable every two months) are recommended to ensure the stability and proper functioning of the image acquisition equipments. Phantoms are used to verify that the variations in SNR and CNR, frequency tuning, spatial relation and degree of distortion are stable and within the manufacturer's specifications.
Prior to the analysis and modeling of signals, the images must be processed making sure that the acquired data are optimal for the analysis. This image processing must include increasing the SNR as much as possible using filters to reduce scattered noise3; image smoothing or averaging to remove artifactual signals and bright areas of noise4 and maximizing spatial resolution without modifying the signal through super-resolution algorithms5; ensuring an accurate spatial coherence between all the points of an organ during the whole study through coregistration6 to ensure voxel repositioning allowing a specific area of an organ to be represented by the same points in all the images obtained in different series.
Segmentation allows us to classify tissues to facilitate their analysis and visualization.7 Several methods may be used for this purpose and they are generally based on the signal of the tissue to be segmented after optimization, enhancement of contrast and use of filters to remove noise. The highest possible resolution in the final image, which decreases the partial volume effect, is recommended to achieve an appropriate segmentation.
In many processes, the signal and volume normalization of an organ into a standard framework is recommended to allow comparison with other population data. This normalization process is done using spatial transformation algorithms, which determine the geometric operations needed to associate the structure of interest with a reference template allowing for a joint analysis.8
The analysis and modeling of the signal allow us to extract information about the biomarkers from the medical digital images using the appropriate computational processes. Although there are many methods and to define them all in the present paper would be impossible, they can be classified according to the parameters analyzed. In this respect, there are methods to estimate the volume and shape of the tissues (as in lung carcinoma), their typology (as in trabecular bone), and some of their physical (transverse and longitudinal relaxation times), chemical (spectroscopy), biological (angiogenesis and cellularity) and functional (local consumption of oxygen) properties.
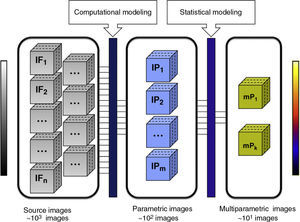
Once these variables are obtained for each voxel of interest, the spatial distribution of the biomarker can be depicted using parametric images (Fig. 2). In parametric images, the pixels represent the value of a specific parameter (morphological or functional) obtained through numerical adjustment of a model and usually represented in a color scale, in contrast with the grayscale used for source images (however, apparent diffusion coefficient maps also show the parametric images in a grayscale). Anomalous pixels may also be represented in a color scale overlaying the source image, in which organs and structures are depicted in a grayscale.
Relevant information extraction process using parametric images. The box on the left represents the number of MR images providing morphological and functional information. The central box represents the parametric images obtained after applying computational models to the source images. Each of these images represents the value of a specific biomarker for each slice of a volumetric study. The box on the right side shows an example of multiparametric images obtained after applying a statistical modeling to the parametric images. This could be used to obtain one or several discriminant functions that indicate which biomarker, and in what proportion, are relevant to statistically characterize biological processes associated with a disease. The reduction in the number of images between boxes is evident. The reason for this is that the implementation of computational and statistical models reduces redundant information, highlighting only those biomarkers statistically associated with the disease.
Multivariate parametric images allow us to reduce redundancy of the acquired data, in other words, to refine the images that must be visualized after optimizing the useful information (Fig. 2). Unlike parametric images, in the images generated by multivariate analysis the color of each voxel is determined by a multivariate statistical function, which is in turn a combination of several parameters or biomarkers. This approach aims to improve the unidimensional statistical analysis and to help the radiologist in the decision-taking process. Some of the multivariate methods include linear regression, discriminant function analysis and the principal component analysis.
Biomarker measurementParametric images, both conventional and multivariate, provide measurements from either the whole tissue or organ being studied or only from those areas considered more representative or abnormal. At this point, we need to define how to work with the histogram that displays the values obtained (a histogram is a graphical representation of the distribution of a given variable in which the vertical axis represents the frequency for the observed value of the biomarker and the horizontal axis represents its different values).
Results such as mean value, standard deviation, and range (parameters used in descriptive statistics) can be obtained from the histogram. One of the problems with this approach is the clear tendency to underestimate the changes in body tissues and organs, since the values indicative of disease, or its most relevant manifestations, are minimized. For this reason, the most extreme values are used in the same settings (the maximum 10% in perfusion maps or the minimum 25% in diffusion coefficient maps) to obtain a better relationship with the most relevant predictive clinical variables. The optimal type of approach chosen must be defined for each problem (complete histogram, partial histogram in quartiles, partial histogram in deciles).
A further approach involves, in a given region (usually a tumor or lesion), the analysis of the heterogeneity in the spatial distribution of a biomarker provided by its parametric image. A classic analysis is to calculate the kurtosis, a statistical parameter that measures the sharpness of the histogram with high kurtosis values meaning narrow distributions, in other words, homogeneous distributions, and vice versa.
Biomarker validationOnce the concept, appropriate images, pertinent analysis, modeling, and the measurement methodology are available, a pilot test on a small sample of subjects, with or without the disease, is conducted to validate the process. The Proof of Principle, or pilot study, is focused on validating the proof of concept and the proof of mechanism, both theoretical, in a small sample, before embarking on large-scale clinical trials. In addition, potential variations related to age, sex or other factors that could lead to misinterpretation must be defined.
If the pilot study succeeds, further work must be done on larger and well-defined series of cases to draw statistically significant conclusions. Within this framework, the Proof of Efficacy is defined as the study that allows us to assess the ability of a biomarker to properly measure (in a reproducible, reliable and accurate manner) the concept. Efficacy is a demonstrative study that defines the strength of healthcare technology in optimal control conditions. The Proof of Effectiveness analyzes, in normal conditions of use, the ability of a biomarker to measure the clinical variable relevant to the disease (endpoint). It is important to remember that the surrogate endpoints, in other words, the imaging biomarkers, are meant to replace clinical endpoints only if they have proved effective and efficient. Only the qualified imaging biomarkers must be used as surrogate endpoints to define a given disease, its state and treatment response.
A bias is a form of systematic error affecting and distorting the measurement process. Since biased studies lack validity, their sources of error must be identified and minimized to avoid misinterpretation of the results.9 Moreover, a good experimental model for method validation is required, as overestimation of the results may bias, in many instances, the measurements of goodness toward the new proposed methodologies against the conventional tests.10
Although the parameters that define an ideal biomarker fulfill this chain of proofs and error control, the reality is that they are more complex in practice. A biomarker must be acquired in technologically stable equipments (to avoid sources of variability in the acquisition process), using non-invasive and safe methods for the patients (in our setting using medical images), must be widely available (to ensure its worldwide distribution and use), reproducible (ability to replicate the obtained value, so that it remains lower than the intended differences, obtaining similar result in institutions with different equipments), standardized in terms of imaging acquisition (technical parameters), preparation and process (filtering, improved signal-to-noise resolution and spatial resolution), analysis and signal modeling (fine tuning of computational models, extraction of variables), and it must be validated in terms of precision and efficacy.
The ideal biomarker must be clinically useful (allowing a measurable clinical improvement), must have a high specificity (high relation between the changes in the biomarker and the effect to be measured) and a high percentage of true positives (to correctly classify as abnormal a true altered finding). In addition, it must have a high specificity (percentage of healthy people who are correctly identified as negative or not having the condition) and being closely associated with the biological process or pathological condition to which is related.
Lastly, the ideal biomarker must be obtained at the lowest possible costs (low-priced) and in the shortest time (fast).
The radiological report with biomarkersTo innovate in clinical practice, the intuitive results produced by the biomarkers need to be conveyed. An appropriate system to convey these results is the DICOM Structured Reporting (DICOM SR). This architecture is designed as a means of encoding documents and exchanging pertinent information through a hierarchical structure. DICOM SR introduces the definitions of DICOM information objects and the systems used for storing and transmitting those structured reports. DICOM SR not only defines the structured data (patient, episode, images, biomarkers and partial reports) but also allows for their storing, search, retrieval, statistical analysis and transfer. Furthermore, DICOM SR has the ability to associate the clinical document of the biomarkers with the patient's episode within a PACS.The structured report must comprise complete and accurate information including the assessment of potential bias and the generalizability of the results. The standard for the presentation of diagnostic reports (of STARD type−Standards for Reporting of Diagnostic Accuracy) consists of a checklist of the main items for the reporting of studies of diagnostic accuracy and the flow diagram required to ensure that all the pertinent information is displayed.11 Among these guidelines, the following are worth mentioning:
- -
Describe data collection.
- -
Describe the study population or the technique used as the reference standard.
- -
Describe the statistical methods and provide information about the test reproducibility.
- -
Include the distribution of the severity of the disease and patient condition in comparison with the reference group.
- -
Discuss the study conclusion based on the clinical applicability to help tackle the relevant clinical endpoints.
The study of the articular cartilage using MRI is based on the qualitative assessment of conventional MR images. In routine practice, a standard classification system12 is used. This system involves the subjective interpretation by the radiologist and may not be sufficiently sensitive to detect early changes or reproducible in follow-up studies. Imaging biomarkers that allow disease staging in a more objective, sensitive and reliable manner (cartilage thickness, T2 and T1 maps) have been recently developed. In the present example, the microvascular properties of the articular cartilage are demonstrated as initial biomarker of degeneration.
Proof of conceptNormal cartilage is resistant to the vascular invasion arising from the subchondral bone. Studies have demonstrated that as articular cartilage degenerates to chondromalacia or osteoarthritis, changes in their antiangiogenic properties occur due to overload and hypoxic conditions, resulting in overexpression of the vascular endothelial growth factor (VEGF) produced by the chondrocytes.13–17
Proof of mechanismPharmacokinetic analysis of dynamic MR images after the administration of a contrast agent is performed to evaluate the microvascular characteristics of the tissues, particularly tumors.18,19 Since we intend to analyze the vascular changes associated with the progression of cartilage degeneration, we propose the utilization of neovascularization biomarkers derived from the implementation of a pharmacokinetic model using contrast enhancement curves to assess early degeneration, progression and vascular response to treatment.
Image acquisitionIn order to extract imaging biomarkers associated with the pharmacokinetic modeling of MRI perfusion, three sequences are required.
First, an echo gradient (EG) sequence with fat suppression (TR=3.4ms, TE=1.9ms, α=10°) is obtained for cartilage segmentation using high spatial resolution and contrast between cartilage, fluid and the rest of tissues.
Then a sequence that allows calculation of the T1 value of the tissue is obtained before contrast agent injection. The T1 value is necessary to convert the signal intensity curves extracted from the perfusion sequence into contrast concentration curves, in order to obtain pharmacokinetic parameters with a physiological meaning. This is a non-linear correlation and must be estimated from shortening of T1 values caused by the contrast agent. Several EG acquisitions with different flip angles are required to obtain T1 (TR=9.2ms, TE=5.4ms, α=2°, 5°, 7°, 10°, 15°, 20°, 25°, 30°, 40°, 60°).20
Dynamic MR perfusion images are obtained using a T1-weighted EG sequence with high temporal resolution for a correct sampling of the arterial phase (TR=7.2ms, TE=3.5ms, α=15°, <5s per dynamic). The acquisition must have a minimum duration of 5min, allowing the analysis of contrast washout and must provide sufficient temporal information of the entire enhancement curve.
Image processingFirst, a spatial filter is used to reduce image noise and then a temporal filter eliminates the noise fluctuations caused by slight movements or by the noise inherent to the MRI process.
Modeling and analysis of signalsThe intensity vs time curves need to be converted into concentration vs time curves before extracting the pharmacokinetic parameters. To this end, the T1 baseline value is obtained using a multi-angle sequence before contrast administration. The T1 vs time curves are then calculated using time-intensity curves extracted from the perfusion sequences. Finally, the values of concentration of contrast are obtained dividing the difference between T1 and baseline T1 by the longitudinal relaxivity of the contrast agent.21
In this work a one-input (popliteal artery) two-compartment (vascular and interstitial) pharmacokinetic model is used. The enhancement curve of each voxel is fitted applying the least square method to the mathematical equations governing the model. This allows us to obtain the modeling parameters including the permeability constant (Ktrans), extraction rate (kep), extravascular extracellular space volume fraction (ve) and the effective intravascular volume fraction (vp). Tofts et al.22 standardized the parameters and units of this pharmacokinetic model; for this reason, it is usually referred to as extended Tofts model.
Uni-multivariate parametrizationThe calculated values may be represented in a parametric map that shows the degenerated areas. Discriminant function analyses allow us to generate new multiparametric maps depicting the tissue degeneration or damage and nosologic maps (Fig. 3). These multivariate techniques allow us to understand which parameters, and in which degree, provide relevant information to characterize the local presence and severity of the disease.
Nosologic map that describes the different degrees of articular cartilage degeneration. On the left, the image shows the coronal view of a normal patellar cartilage. The color scale shows the degree of degeneration. The central and right images depict patellar cartilages with early and advanced degeneration, respectively. These nosologic maps derive from discriminant functions, in which the value of the functions represents the probability of a given pixel being pathological.
The measurements of the different pharmacokinetic parameters are obtained using the regional histogram given the heterogeneous distribution of the areas of neovascularization. This ensures that no relevant regional information is missed, particularly when detecting small degenerated areas that may be masked if calculating the mean of the whole cartilage. In these cases, it is advisable to work with only the maximum deciles or quartiles.
Proof of principleRecent studies have correlated an increase in these parameters with the progression of the articular degeneration in a small group of patients, including healthy patients and patients with chondropathy.23,24 These studies prove that pharmacokinetic parameters can measure the intended effect, with statistically significant differences for Ktrans and ve values between healthy subjects, patients with early degeneration and patients with advanced degeneration.
Proof of efficacy and effectivenessLarge-scale research studies are required to assess the feasibility and clinical use of this biomarker in the staging and follow-up of the articular cartilage.
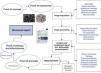
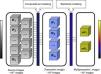
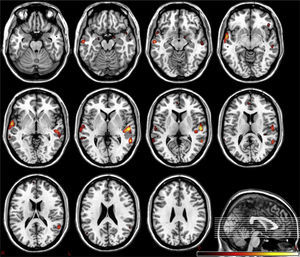
Present and future of imaging biomarkersThe new radiological scenario that emerges after the introduction of biomarkers provides considerable benefits to diagnostic imaging techniques. As with other emerging technologies, they may initially build to a peak of expectations that is very different from reality, but over time, the quantitative information provided by biomarkers will be incorporated into daily clinical practice. For instance, it is already possible to study if structural and functional brain abnormalities coexist in patients with psychiatric and neurodegenerative disorders, allowing for the identification of the most relevant areas for the disease25,26 (Fig. 4). Similarly, it is possible to analyze the elasticity and several topological and mechanical parameters of trabecular bone in order to determine bone resistance and fracture risk in osteoporotic conditions27,28 (Fig. 5).
Left, 3D reconstruction of trabecular bone extracted from high spatial resolution MR images. Center, conversion of the trabecular bone geometry into a finite element model based on small hexahedron elements. In this 3D image, each small cube corresponds to a finite element. Each element has specific physical and chemical properties, corresponding to bone in this example, and is linked to the rest of finite elements forming a mesh. Right, parametric map shows nodal strain caused by simulated compression on the trabecular structure. High values (in red) correspond to high nodal strain, in other words, to regions with higher risk of rupture.
The present and future of these and other biomarker applications involve the integration of the radiological processes, with particular attention to the standardization of the procedures and of the acquisition protocols and the development of the shared instruments for knowledge extraction clearly focused on improving the disease diagnosis and follow-up.
ConclusionDigital medical imaging and computational processing allow for the extraction of parameters considered as functional images29 or imaging biomarkers.30 In the clinical practice, these biomarkers may be of great interest due to the advantages that they provide to the diagnostic, therapeutic and follow-up processes in many pathological conditions. To this end, we must check the integrity of the whole cycle, from the conception to the implementation (Fig. 1). The combination of digital imaging and computing is in many ways a kind of magic, conjuring up the occult and the mysterious, but its ultimate purpose is to achieve professional success understood as excellence in clinical and experimental medicine. All these advantages lie in the multidisciplinary work of professionals who work together to provide better care to patients and better understanding of the disease.
Conflicts of interestThe authors declare not having any conflict of interest.
Please cite this article as: Martí Bonmatí L, et al. Biomarcadores de imagen, imagen cuantitativa y bioingeniería. Radiología. 2012;54:269–78.