We present the case of a 52-year-old man with no relevant medical history who was admitted to our centre's intensive care unit due to multiple severe traumatic injuries resulting from a traffic accident. He required orotracheal intubation during transfer to hospital (Glasgow Coma Scale score of 8). A CT study (head, neck, chest, abdomen, and pelvis scans) revealed a small subgaleal haematoma in the left parietal region, with no evident alterations in the brain parenchyma or the base of the skull; orbital blowout fracture; pulmonary contusion leading to right haemopneumothorax; sternal fracture; right posterior sternoclavicular joint dislocation; and flail chest of the right hemithorax. Once haemodynamic stabilisation was achieved, the patient underwent 3 interventions. Firstly, he underwent chest drainage for haemopneumothorax, which progressed favourably. He then underwent surgery to treat the sternoclavicular joint dislocation and sternal fracture (patient in the supine position with a pad placed in the interscapular region). Lastly, a tracheostomy was performed due to prolonged orotracheal intubation (10 days) (patient in the supine position with the neck in hyperextension). A follow-up head CT scan performed at 12 days showed intraparenchymal hypodensities in the right cerebellar hemisphere and in the frontal and parietal subcortical regions bilaterally. An MRI scan and a vascular study revealed infarction of the territory of the right superior cerebellar artery, bilateral frontal and parietal contusion, and no sign of arterial dissection or other parenchymal lesions.
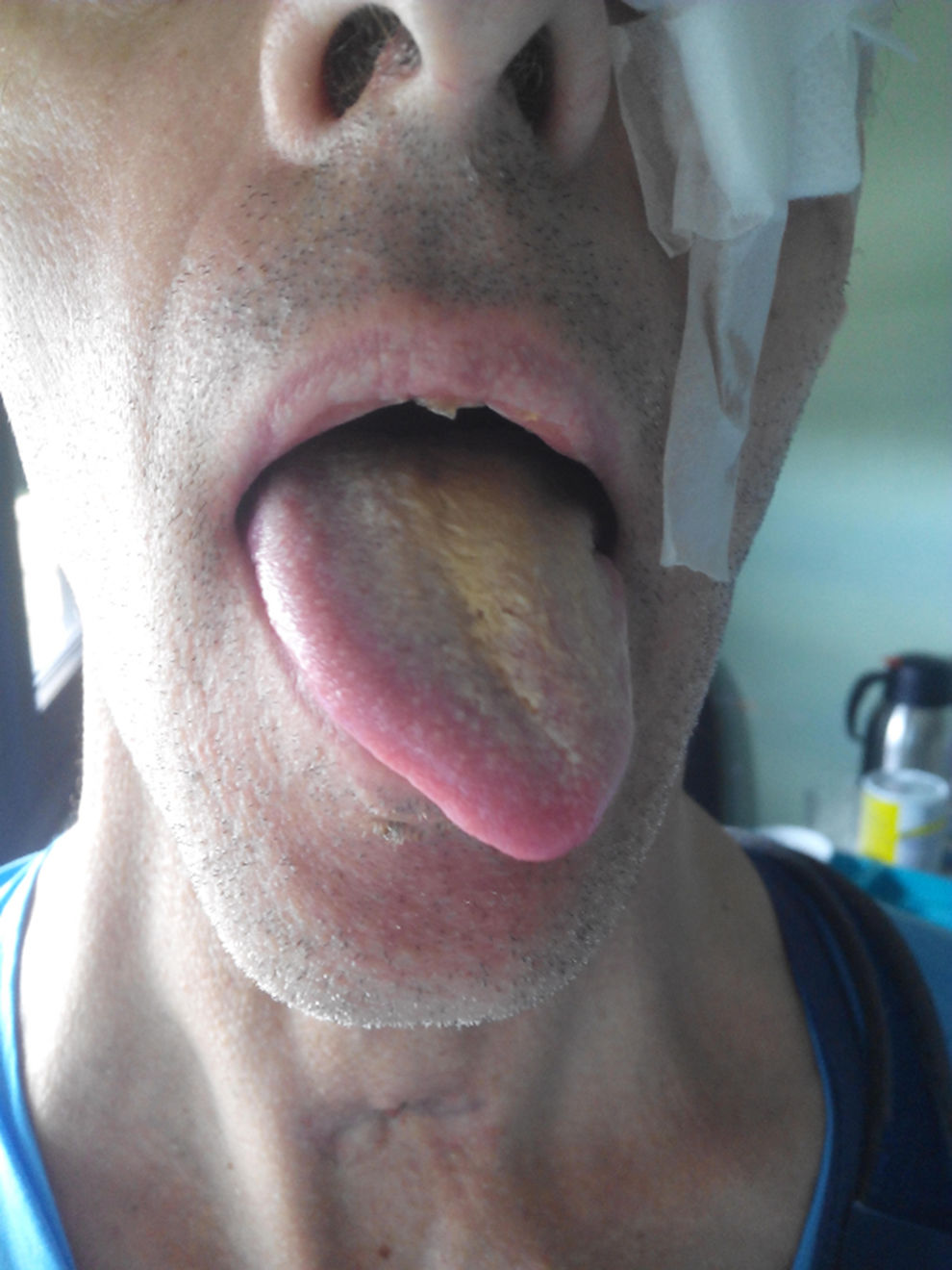
The neurological examination at discharge from the intensive care unit showed that the patient was alert, collaborative, and oriented in time, space, and person. He showed anisocoria with an unreactive mid-sized left pupil; scanning speech; dysphonic, bitonal voice; tongue deviation to the left associated with hypotrophy of the left side of the tongue and no fasciculations (Fig. 1); hypoaesthesia and hypogeusia in the left side of the tongue (2 anterior thirds and posterior third); normal soft palate motility; and pharyngeal reflex upon stimulation of the soft palate and right posterior region of the pharynx but no pharyngeal reflex upon stimulation of the base of the left side of the tongue and pharynx; the patient presented dysphagia secondary to these pharyngeal alterations. The patient also displayed tactile hypoaesthesia in the territories of the right radial and median nerves, mild hypoaesthesia in the left side of the body, and muscle atrophy in the right forearm. The examination revealed no further abnormalities. Fibreoptic laryngoscopy revealed left vocal cord paralysis in the paramedian position. The patient started speech therapy and physiotherapy at the rehabilitation department. Oral and maxillofacial surgery was performed 3 months later: the left orbital floor and wall were reconstructed, with a good outcome. Three months of rehabilitation led to progressive improvements in dysarthria and dysphonia, with near-complete recovery of the patient's ability to swallow solids and liquids.
Polytrauma caused multiple lesions to the central and peripheral nervous systems: parasympathetic lesion to the left oculomotor nerve (orbital fracture); incomplete lesion to the right brachial plexus associated with the right sternoclavicular joint dislocation and sternal fracture; mild left-sided hypoaesthesia secondary to supratentorial haematomas; and, most importantly for the purpose of this case report, a lesion to the left lingual, hypoglossal, and recurrent laryngeal nerves as well as to the tonsillar and lingual branches of the left glossopharyngeal nerve.
Differential diagnoses of lesions to the lower cranial nerves include Tapia syndrome (X and XII), Collet-Sicard syndrome (IX, X, XI, and XII), Villaret syndrome (IX, X, XI, and XII plus Horner syndrome), Vernet syndrome (IX, X, and XI), Schmidt syndrome (X and XI), and Jackson syndrome (X, XI, and XII).1–4 To our knowledge, there have been no previous cases of simultaneous lesion to the nerves damaged in our patient. Our case resembles Tapia syndrome in that cranial nerve XI was intact (unlike in the remaining differential diagnoses), and in the pathogenic mechanisms involved (orotracheal intubation and cervical hyperextension). Unlike in Tapia syndrome, however, our patient showed lesions to the lingual nerve and the tonsillar and lingual branches of the glossopharyngeal nerve. Other researchers have reported injury to the lingual nerve,5,6 and simultaneous damage to the glossopharyngeal, recurrent laryngeal, and hypoglossal nerves3,7 in patients undergoing orotracheal intubation and/or cervical hyperextension.
Tapia syndrome was described in 1904 by the Spanish otorhinolaryngologist Antonio García Tapia, who reported paralysis of the vagus and hypoglossal nerves in a bullfighter following a jaw lesion. The syndrome is characterised by unilateral paralysis of the tongue and ipsilateral vocal cord. This is an infrequent entity caused either by extracranial peripheral nerve injury (hypoglossal nerve and recurrent laryngeal branch of the vagus nerve) or, even more rarely, by damage to such CNS structures as the nucleus ambiguus, hypoglossal nucleus, and pyramidal tract. Peripheral nerve lesions in the syndrome are most frequently attributed to orotracheal intubation (accounting for more than three-quarters of cases).1–4 In view of our patient's symptoms, we may hypothesise that the lesion affected the area between the lateral wall of the lower part of the oropharynx, at the base of the tongue, and the upper part of the hypopharynx, at the piriform fossa. In this area, the recurrent laryngeal and hypoglossal nerves run near the lingual nerve and the common segment of the lingual and tonsillar branches of the glossopharyngeal nerve.1–3
The aetiological study of patients with lesions to cranial nerves IX, X, XI, and XII, or other differential diagnoses, should include a detailed medical history, a thorough examination, head and neck CT and MRI scans (to detect space-occupying lesions, haemorrhage, fractures at the base of the skull), and a vascular study (neurosonology, CT angiography, and/or MRI of the supra-aortic trunks) to rule out lesions to the carotid or vertebral arteries.2,8 Treatment involves speech therapy and swallowing rehabilitation. Other treatments include a 2-week course of oral or intravenous corticosteroids and administration of vitamins B1, B6, and B12 for at least 3 months; the efficacy of this approach is not sufficiently demonstrated, however. A nasogastric tube or percutaneous gastrostomy may be helpful while the patient recovers swallowing function; these options reduce the risk of lower airway foreign body aspiration. As the condition is a neurapraxia in most cases, prognosis is usually very good, with a large proportion of patients achieving complete or near-complete recovery.2,5,8–10
Please cite this article as: Sanabria Sanchinel AA, Navarro Pérez MP, Flores Robles BJ, Tejero Juste C. Tapia Plus: un nuevo síndrome o una variante. Neurología. 2019;34:348–349.