Cannabis and its synthetic analogues are currently the most widely consumed illicit substances worldwide. Multiple alterations have been linked to its use, including cerebrovascular disease (CVD) or stroke, whose association with the substance has been based mainly on the hypothesis of transient vasoconstriction, which explains a large proportion of the cases reported. However, epidemiological studies have not performed an individual analysis of patients without other cardiovascular risk factors, which may limit the estimation of the risk of stroke associated with cannabis use alone.
DevelopmentA systematic literature review was conducted through Medline, EBSCOhost, EMBASE, Lilacs, and Scielo to gather case reports published before 13 May 2016 presenting patients with a diagnosis of CVD or transient ischaemic attack, a history of cannabinoid use, and no other cardiovascular risk factors. Key words such as stroke, cerebrovascular disease, cannabis, and marijuana, among others, were used. A total of 18 case reports were selected from the 566 references found.
ConclusionsThere is a wide variety of reports of stroke associated with cannabis use in patients with no other risk factors. Noteworthy findings were presentation at young age and a strong temporal association, which place cannabis use as a potential risk factor for this population in line with the epidemiological and pathophysiological studies in this area.
El cannabis y sus análogos sintéticos corresponden actualmente a las sustancias ilícitas de mayor consumo a nivel mundial. Resultan múltiples las alteraciones con las cuales se ha relacionado su consumo, y dentro de estas se encuentra la enfermedad cerebrovascular (ECV) o ictus, cuya asociación con esta sustancia se ha fundamentado principalmente a través de la hipótesis de vasoconstricción transitoria, la cual explica una amplia proporción de los casos descritos. Sin embargo, los estudios epidemiológicos realizados no han desarrollado un análisis individual en los pacientes sin otros factores de riesgo cardiovascular, lo que puede limitar la estimación del riesgo que confiere por sí mismo el consumo de esta droga.
DesarrolloSe realizó una revisión sistemática de la literatura a través de Medline, EBSCOhost, EMBASE, Lilacs y Scielo hasta el 13 de mayo de 2016 considerando estudios de tipo «Caso clínico» que presentaran pacientes con diagnóstico de ECV o ataque isquémico transitorio e historia de consumo de cannabinoides sin otros factores de riesgo cardiovascular. Se utilizaron palabras clave como ictus; enfermedad cerebrovascular; cannabis; marihuana, entre otras. Finalmente, de las 566 referencias encontradas fueron seleccionados 18 casos clínicos.
ConclusionesExiste una amplia variedad de publicaciones de ictus asociado a consumo concomitante de cannabis en pacientes sin otros factores de riesgo, destacando una presentación a edad joven y una alta temporalidad, lo que, en concordancia con los estudios epidemiológicos y fisiopatológicos en esta área, ubica a esta sustancia como un potencial factor de riesgo para esta población.
Cannabis sativa, also known as hashish, hemp, pot, weed, and marijuana, is a plant from the Cannabaceae family that has become the most widely used illegal substance among the young population worldwide; this is mainly due to the popular perception that this is a “safe” substance with minimal side effects.1 However, multiple pathological conditions have been related to its consumption, including psychiatric disorders and diseases of the respiratory and cardiovascular systems; the latter group includes transient vasoconstriction and ischaemia, mainly involving the brain parenchyma. This association has been widely discussed in recent years, after the publication of a significant number of clinical cases and other studies that have established a link between consumption of the substance and brain ischaemia.2,3
Many of these analyses include patients with a diagnosis of stroke and reported cannabis use associated with concomitant cardiovascular risk factors; furthermore, no review article focusing on the assessment of patients with no other apparent risk factors has been published to date. Our systematic review aimed to gather and discuss the findings of all the cases published over the past 20 years and reporting a cerebrovascular event with consumption of cannabis in patients with no cardiovascular risk factors. We assess the time between substance consumption and the cerebrovascular event, and discuss the symptoms; we found cannabis use to be a significant risk factor for stroke and underscore the need to continue researching this association.
DevelopmentEligibility criteriaWe reviewed case reports and case series presenting one or more patients with a diagnosis of stroke or transient ischaemic attack (TIA) and a history of consumption of cannabinoids, with consumption reported in the 24hours before stroke onset. We also selected abstracts from communications presented at congresses. Furthermore, we ruled out studies reporting other stroke risk factors, such as the consumption of such other psychoactive substances as cocaine, smoking, dyslipidaemia, arterial hypertension, diabetes mellitus, chronic obstructive pulmonary disease, obesity, family or personal history of stroke, patent foramen ovale, cardiac arrhythmias, and coagulation disorders. We also excluded studies not including diagnostic imaging or reporting outcomes, and those in which a definitive diagnosis of stroke was not established. Finally, we excluded studies in animal models, those published more than 20 years ago, and those published in a language other than English, Spanish, or Portuguese.
Search strategyWith a search end date of 13 May 2016, we performed a search in the Medline, EBSCOhost, EMBASE, Lilacs, and Scielo databases using the following search algorithm:
- 1.
Cerebrovascular disorders or brain ischemia or ictus or cerebrovascular disease or cerebral artery or cerebral infarction or cerebral hemorrhage or intracranial embolism or intracranial thrombosis
- 2.
Cannabis or marijuana or hashish or hemp or bhang
- 3.
We selected studies fulfilling criteria 1 and 2
- 4.
We removed studies not published in Spanish, English, or Portuguese
- 5.
We removed studies in animal models
- 6.
We removed repeated studies
We performed a general review of the articles by assessing their titles and selecting those related to the aims of our study. We then reviewed the abstracts individually using the inclusion and exclusion criteria established. Finally, we manually assessed the references of the articles to include any other relevant studies not identified in our search.
ResultsWe obtained a total of 566 references. We reviewed the abstracts and found that 498 references provided no useful information for the main aim of the study; we then downloaded the selected articles for assessment of the full texts, and ruled out another 50 studies which met other exclusion criteria. Fig. 1 shows the selection process.
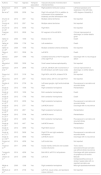
We finally selected 18 articles (Table 1), including a total of 34 eligible patients.4–21 Patients’ mean age was 30 years (range, 15-64 years); they were predominantly men (82.3%).
Clinical cases of stroke in cannabis users with no other cardiovascular risk factors.
| Authors | Year | Age/sex | Temporal association | Vascular structures involved and/or infarcted territory | Outcome |
|---|---|---|---|---|---|
| Baharnoori et al.5 | 2014 | 22/M | Yes | Right posterior lenticular nucleus | Left facial paralysis and weakness in the left limbs |
| Bal et al.6 | 2009 | 22/M | Yes | Right VA and both PCA in addition to involvement of the cerebellum bilaterally and the left temporal lobe | mRS degree of disability 2 |
| Drumm et al. (1)7 | 2012 | 34/F | Yes | Multiple arterial territories | Not reported |
| Drumm et al. (2)7 | 2012 | 29/F | Yes | Multiple arterial territories | Not reported |
| Drumm et al. (3)7 | 2012 | 64/M | Yes | Right MCA | Not reported |
| Freeman et al.8 | 2013 | 26/M | Yes | M1 segment of the left MCA | Clinical improvement at discharge; no further details given |
| Geller et al. (1)9 | 2004 | 16/M | Yes | Bilateral SCA | Death |
| Geller et al. (2)9 | 2004 | 17/M | Yes | Left cerebellum | Death |
| Geller et al. (3)9 | 2004 | 15/M | Yes | Multiple cerebellar arteries bilaterally | Not reported |
| Giray et al.10 | 2011 | 35/M | No | Left MCA | Death |
| Gómez-Ochoa et al.11 | 2007 | 19/M | Yes | Collateral branches of the P3 segment of the right PCA | Discharge with no neurological deficit |
| Mouzak et al.12 | 2000 | 30/M | Yes | Small vessel leukoencephalopathy | Not reported |
| Oyinloye et al.13 | 2014 | 26/M | Yes | Left ICA, left MCA with involvement of the left striatum and left insular cortex | Clinical improvement at discharge; no further details given |
| Rose et al. (1)14 | 2015 | 31/M | Yes | Right MCA, left ACA, bilateral PICA | Not reported |
| Rose et al. (2)14 | 2015 | 25/F | Yes | Basilar artery, left VA, and right PCA | Not reported |
| Santos et al.15 | 2014 | 27/M | No | Left basal ganglia, right lenticulostriate area | Re-exposure to cannabis and recurrent stroke |
| Singh et al. (1)16 | 2012 | 15/M | Yes | Right cerebellar hemisphere | Rehabilitation |
| Singh et al. (2)16 | 2012 | 16/M | Yes | Both cerebellar hemispheres | Death |
| Singh et al. (3)16 | 2012 | 17/M | Yes | Both cerebellar hemispheres | Death |
| Singh et al. (4)16 | 2012 | 22/M | Yes | Right cerebellar hemisphere | Re-exposure to cannabis and recurrent stroke |
| Singh et al. (5)16 | 2012 | 27/F | Yes | Left MCA branch | Discharge with no recurrence |
| Singh et al. (6)16 | 2012 | 28/F | Yes | Right cerebellar hemisphere | Rehabilitation |
| Singh et al. (7)16 | 2012 | 37/M | Yes | Left MCA branch | Rehabilitation |
| Singh et al. (8)16 | 2012 | 44/M | Yes | Both cerebellar hemispheres | Re-exposure to cannabis and recurrent stroke |
| Singh et al. (9)16 | 2012 | 44/F | Yes | Left MCA branch | Rehabilitation |
| Singh et al. (10)16 | 2012 | 49/M | Yes | Right MCA branch | Rehabilitation |
| Singh et al. (11)16 | 2012 | 56/M | Yes | Right PCA and right cerebellar hemisphere | Re-exposure to cannabis and recurrent stroke |
| Singh et al. (12)16 | 2012 | 58/M | Yes | Left MCA branch | Discharge with no recurrence |
| Smaoui et al.17 | 2014 | 42/M | Yes | Left frontal lobe | Not reported |
| Trojak et al.18 | 2011 | 24/M | Yes | Insular mantle, lenticular and caudate nuclear structures | Tonic–clonic seizuresContinued cannabis use without stroke recurrence |
| Tsivgoulis et al.19 | 2014 | 42/M | Yes | Both MCA, left PCA, left putamen | Discharge with total remission of symptoms at 8 months |
| Urbano et al.4 | 2015 | 23/M | Yes | Left SCA | Mild left hemiparesisRecurrence of stroke 2 weeks later with no reported cannabis use |
| White et al.20 | 2000 | 15/M | Yes | Right cerebellar hemisphere | Mild right dysdiadochokinesia |
| Whitlock et al.21 | 2015 | 21/M | Yes | Left PCA, visual cortex, and ipsilateral corpus callosum | Not reported |
ACA: anterior cerebral artery; F: female; ICA: internal carotid artery; MCA: middle cerebral artery; M: male; mRS: modified Rankin Scale; PCA: posterior cerebral artery; PICA: posterior inferior cerebellar artery; SCA: superior cerebellar artery; VA: vertebral artery.
We found 2 cases (5.8%) of consumption of synthetic cannabinoids, such as “K-2”8,14; furthermore, temporal association between consumption and symptom onset was not reported in only 2 cases (5.8%), with both patients reporting chronic consumption of high doses of cannabis; results of the urine and serum toxicological screening tests were positive for cannabis only. Toxicological drug screening tests were performed in 12 cases (35.2%) to detect psychoactive substances; 10 patients tested positive (83.3%), all of them for cannabis only.4,9–12,14–16,18,21
Imaging findings were varied, with predominantly stenosing/obstructive involvement of supratentorial regions; involvement of the middle cerebral artery was reported in 15 cases (44.1%), with the left middle cerebral artery affected in 9 (26.4%); 7 patients (20.5%) presented posterior cerebral artery involvement, and the anterior cerebral artery was affected in 3 cases (8.8%). Some type of cerebellar involvement was reported in 14 patients (41.1%). Other vascular structures were less frequently affected, including the basilar artery (2.9%), the vertebral arteries (5.8%), and the internal carotid artery (2.9%).
No mention was made of final outcome or recurrence of stroke in 9 cases (26.4%). In 4 out of 5 patients with reported recurrence, this was associated with recurrent consumption of cannabis. Five patients died (14.7%).
DiscussionThe results of this review suggest a possible association between cannabis consumption and the development of cerebrovascular events; this is explained firstly by the absence of other relevant risk factors in the cases analysed and its development predominantly in young patients, and secondly by the strong temporal association between substance consumption and stroke symptom onset, both in the cases reported and in subsequent recurrences.
However, despite the wide range of cerebrovascular, cardiovascular, and peripheral vascular events related to cannabis consumption, the mechanism involved in this association is little understood.22 Multiple hypotheses have been proposed to explain this association, including the cardiac overload hypothesis, which attributes these events to increased heart rate and arterial pressure associated with electrocardiographic changes induced by cannabis consumption.23 Furthermore, reduced coronary flow has also been identified as a possible trigger factor.24 This is also reported to be caused by a process of systemic hypotension, with animal studies showing that high doses of this substance may favour inhibition of sympathetic tone.25 These theories include a vasoconstriction or vasospasm-type vascular hypothesis; this is consistent with the findings of Ducros et al.,26 who described 9 cases of reversible vasoconstriction syndrome associated with cannabis consumption; MRI scans revealed diffuse or multifocal segmental arterial constriction. Furthermore, Herning et al.27 reported similar findings using transcranial Doppler ultrasound, observing increased vascular resistance and flow velocity in cannabis users compared to non-users; a similar tendency was observed even during abstinence. Finally, Wolff et al.28 supported this hypothesis in a cohort study of 48 young patients with stroke: the authors found that 21% of the patients presented an angiographic pattern of multifocal intracranial stenosis involving multiple arteries associated with consumption of cannabis, which was reversible with cannabis discontinuation. For this reason, increases in brain vascular resistance and the subsequent changes shown in diagnostic images might be one of the main mechanisms associated with the development of TIA and stroke in cannabis users.22
Other indirectly related factors described in the literature include thromboembolisms of cardiac origin due to patient foramen ovale, or atrial fibrillation or flutter, which have been reported after cannabis consumption.29,30 Finally, increased calorie intake has been reported in patients with chronic consumption of this substance, in addition to a greater likelihood of using tobacco or such illegal drugs as cocaine; together with the risk inherent to the use of cannabis, this exposes patients to a significant risk of more frequent cerebrovascular events.31
Addressing the biologically plausible hypothesis described above, several studies have assessed this relationship, with very contradictory results. In a population study conducted in Texas (USA), Westover et al.32 reported that cannabis consumption was associated with a higher risk of developing an ischaemic stroke even after adjusting for associated variables such as tobacco and alcohol use (OR: 1.76; 95% CI, 1.15-2.71). In a prospective study of young patients admitted due to stroke with a positive urine toxicological drug screen, Barber et al.33 found that cannabis use was associated with a higher risk of ischaemic stroke and TIA (OR: 2.30; 95% CI, 1.08-5.08). However, after adjusting for tobacco use, an independent association was not observed (OR: 1.59; 95% CI, 0.71-3.70).
Subsequently, the study conducted by Hemachandra et al.34 in Australia, gathering patients from the prospective cohort of the Personality and Total Health (PATH) study, found that patients reporting cannabis use in the previous year were 3.3 times more likely to present a stroke/TIA (95% CI, 1.8-6.3, P<.001), with the risk persisting even after adjusting for covariates (IRR: 2.3; 95% CI, 1.1-4.5); the risk was greater in patients using the drug at least weekly (IRR: 4.7; 95% CI, 2.1-10.7; P<.001), whereas patients using the substance less frequently showed no significant association (IRR: 1.2; 95% CI, 0.4-3.4; P=.77).
Finally, Falkstedt et al.35 analysed the association between cannabis use and development of stroke/TIA using data from a national survey of Swedish men conscripted into military service in 1969 and 1970 (including data on alcohol, tobacco, and psychoactive substance use) and the National Hospital Discharge Register to identify those with a cerebrovascular event. In general, they observed no association in patients younger than 45, regardless of the level of consumption (HR: 0.93; 95% CI, 0.34-2.57), although a non-significant increase in the risk of ischaemic stroke was observed in patients reporting cannabis use more than 50 times in the past (HR: 1.47; 95% CI, 0.83-2.56).
The results of the studies identified are clearly contradictory, with no general consensus established on the potential risk attributable to cannabis use. Furthermore, it is well known that most patients with stroke are not cannabis users; therefore, the implications of frequency of use, dose, percentage of tetrahydrocannabinol, coingestants, and genetic susceptibility factors play an important role in the development of a cerebrovascular event, making this condition difficult to analyse.36
However, as mentioned previously, these studies did not exclusively select patients with no other cardiovascular risk factors, and depend on mathematical models for estimating risk. Further prospective cohort studies, including an analysis of the subgroup of patients with no other risk factors, would enable more accurate assessment of the risk inherent to cannabis use. Such information may guide clinicians in the assessment of stroke risk attributable to cannabis use, including this factor in their questions regarding cerebrovascular risk factors in clinical history interviews, especially in young adults with no other apparent risk factors; there may even be justification for the inclusion of this substance in the routine analytical studies in this population.4
FundingThis study required no funding.
Conflicts of interestThere is no conflict of interest to declare.
Please cite this article as: Gómez Ochoa SA. Ictus y consumo de cannabis en pacientes sin factores de riesgo cardiovascular: revisión sistemática de casos clínicos. Neurología. 2021;36:222–228.