Natalizumab treatment has been shown to be very efficacious in clinical trials and very effective in clinical practice in patients with relapsing-remitting multiple sclerosis, by reducing relapses, slowing disease progression, and improving magnetic resonance imaging patterns. However, the drug has also been associated with a risk of progressive multifocal leukoencephalopathy (PML). The first consensus statement on natalizumab use, published in 2011, has been updated to include new data on diagnostic procedures, monitoring for patients undergoing treatment, PML management, and other topics of interest including the management of patients discontinuing natalizumab.
Materials and methodsThis updated version followed the method used in the first consensus. A group of Spanish experts in multiple sclerosis (the authors of the present document) reviewed all currently available literature on natalizumab and identified the relevant topics that would need updating based on their clinical experience. The initial draft passed through review cycles until the final version was completed.
Results and conclusionsStudies in clinical practice have demonstrated that changing to natalizumab is more effective than switching between immunomodulators. They favour early treatment with natalizumab rather than using natalizumab in a later stage as a rescue therapy. Although the drug is very effective, its potential adverse effects need to be considered, with particular attention to the patient's likelihood of developing PML. The neurologist should carefully explain the risks and benefits of the treatment, comparing them to the risks of multiple sclerosis in terms the patient can understand. Before treatment is started, laboratory tests and magnetic resonance images should be available to permit proper follow-up. The risk of PML should be stratified as high, medium, or low according to presence or absence of anti-JC virus antibodies, history of immunosuppressive therapy, and treatment duration. Although the presence of anti-JC virus antibodies is a significant finding, it should not be considered an absolute contraindication for natalizumab. This update provides general recommendations, but neurologists must use their clinical expertise to provide personalised follow-up for each patient.
Natalizumab es un tratamiento que ha demostrado ser muy eficaz en los ensayos clínicos y muy efectivo en la práctica clínica en los pacientes con esclerosis múltiple recurrente-remitente, en cuanto a la reducción del número de brotes, enlentecimiento de la progresión de la enfermedad y variables de resonancia magnética. Sin embargo, el fármaco se ha asociado con el riesgo de desarrollar leucoencefalopatía multifocal progresiva (LMP). Los nuevos datos aparecidos sobre la monitorización de los pacientes en tratamiento, el diagnóstico, y manejo de la LMP y otros temas de interés como la actuación ante la suspensión del natalizumab se han incorporado en esta actualización del primer consenso sobre el uso del natalizumab publicado en el 2011.
Material y métodosEn esta actualización se procedió con la misma metodología que la del primer consenso. Un grupo de expertos españoles en esclerosis múltiple (los autores de esta actualización) revisaron toda la bibliografía disponible sobre natalizumab hasta la fecha, y basándose en su experiencia clínica definieron los temas relevantes a actualizar. Un primer borrador se sometió a ciclos de revisión hasta llegar a la versión final.
Resultados y conclusionesLos estudios de práctica clínica han demostrado que el cambio a natalizumab es más efectivo que el cambio entre inmunomoduladores, y apoyan una mayor conveniencia del tratamiento temprano con natalizumab frente a una utilización más tardía como terapia de rescate. A pesar de ser un fármaco muy eficaz, se deben tener en cuenta los posibles efectos adversos y, en particular, la probabilidad de desarrollar LMP. El neurólogo debe explicar al paciente en detalle los riesgos y beneficios del tratamiento frente al riesgo de la esclerosis múltiple usando términos comprensibles. Antes de empezar el tratamiento, deben estar disponibles las pruebas de laboratorio y las imágenes de resonancia magnética que permitan hacer un seguimiento adecuado. El riesgo de LMP debe estratificarse en alto, medio y bajo de acuerdo con la presencia o ausencia de anticuerpos frente al virus JC, antecedente de tratamiento inmunosupresor y duración del tratamiento. La presencia de anticuerpos antivirus JC, aunque significativa, no se puede tomar como una contraindicación absoluta para el natalizumab. La presente actualización establece unas recomendaciones generales, pero los neurólogos deben aplicar su experiencia clínica para hacer un seguimiento individualizado de los pacientes.
The first Spanish consensus statement on the use of natalizumab (Tysabri®), a disease-modifying treatment for relapsing-remitting multiple sclerosis (RRMS), was published in 2011.1 Natalizumab is a recombinant humanised monoclonal antibody against the cell adhesion molecule alpha-4-integrin. It selectively inhibits the interaction of integrin alpha-4-beta-1 in mononuclear leukocytes with its analogous receptor, vascular cell adhesion molecule-1, which is present in the endothelium of the blood-brain barrier (BBB).2
It is believed that lesions in multiple sclerosis (MS) are produced when activated T-cells cross the BBB. Lymphocyte migration across the BBB involves interaction between the adhesion molecules in inflammatory cells and the endothelial cells of the vascular wall. The interaction between integrin alpha-4-beta-1 in mononuclear leukocytes and its targets provides a key mechanism for pathological inflammation of the brain.2 Inhibiting this interaction using natalizumab has been proved very effective in patients with RRMS.
Natalizumab was first approved by the European Medicines Agency (EMA) in 2006 for the treatment of adult patients with RRMS (age 18 or older) with disease that remains highly active despite treatment with interferon beta or glatiramer acetate (the latter treatment was recently approved by the EMA). It was also approved for adult patients with severe, rapidly-progressing RRMS (indicated in de novo patients).2
Through the action mechanism described above, natalizumab has demonstrated considerable clinical and radiological efficacy. As a result, ‘absence of disease activity’ is now a treatment objective (no activity such as relapses or disability progression on clinical measures, and no activity on radiological measures). In the AFFIRM trial, this objective was achieved in 37% of the patients treated with natalizumab during 2 years of treatment, compared to 7% in the placebo group.3 Subsequently, multiple studies undertaken in normal clinical practice have confirmed that this treatment objective could be achieved in high percentages of patients treated with natalizumab.4–6
Natalizumab use is associated with an increased probability of developing progressive multifocal leukoencephalopathy (PML).7 When the first reports of PML were published, we gained a better understanding of the risks and benefits of this drug, and continue to do so thanks to the research that has been undertaken by all parties involved. Doctors now have access to an ample body of long-term studies of the drug, and to relevant, highly useful information that will help both patients and neurologists in decision-making.
Since 2011, researchers have published new results on the efficacy of sustained long-term natalizumab treatment,8 the likelihood of keeping patients free from disease activity, and the identification of variables predicting more successful treatment outcomes.5 Stratify JCV (a 2-step immunoenzymatic assay that detects anti-JC virus antibodies) has been shown to detect JC virus seropositivity.2,9 Other risk factors have also been identified.2 These advances provide personalised monitoring tools that are very useful for managing natalizumab in clinical practice. Lastly, the Spanish summary of product characteristics has been updated multiple times since 2011, when the Spanish consensus statement on natalizumab use was published.
Considering the above, we have decided to update the 2011 consensus statement so that it will reflect new evidence gathered since that date. This document, a revised version of the previous consensus statement, summarises advances and new evidence that have been published in the past few years. We also provide neurologists with practical guidelines for starting, continuing, and when necessary, withdrawing natalizumab treatment.
MethodsThe methods used to draft the first Spanish consensus statement were described in that document.1 We used the same methods to complete this revised version. A group of Spanish experts in MS (the authors of this consensus statement) reviewed all the currently available literature on natalizumab. Based on their clinical experiences, they identified relevant topics to be updated as part of the process of revising the consensus for natalizumab use. The coordinator (O.F.) wrote the first draft, which then passed through various cycles of review before appearing in its final form. The group of experts debated the new data and evidence published since the first consensus was written. The most relevant changes with regard to clinical management of natalizumab were included in the draft; this document was reviewed once more by all authors to produce the final statement.
Efficacy and effectiveness of natalizumabThe efficacy of natalizumab as monotherapy for RRMS was demonstrated in the pivotal placebo-controlled clinical trial AFFIRM.10 Over the 2-year study period, natalizumab significantly reduced the annual rate of relapse (ARR) by 68%. It also reduced likelihood of disability progression by 42%, confirmed at 3 months, and by 54%, confirmed at 6 months. Natalizumab was also effective at reducing disease progression as shown by MRI (number of new lesions or growing lesions in T2-weighted images decreased by 83%; gadolinium-enhancing lesions decreased by 92%; and T1-weighted lesions decreased by 76%).11 Natalizumab had a significant effect on the rest of the variables measured in the study (brain atrophy during the second year of treatment,11 number of hospitalisations,12 use of corticosteroids,12 progression of cognitive impairment, and quality of life13). These efficacy results led to a post hoc analysis with new efficacy objectives: ‘percentage of patients free from disease activity (clinical or radiological)’3 and ‘likelihood of sustained improvement on the extended disability status scale (EDSS)’.14 A significant positive effect was shown for treatment with natalizumab vs placebo for both objectives.
The AFFIRM trial studied natalizumab in a population of RRMS patients with no previous treatment (history of treatment with interferon beta or glatiramer acetate for more than 6 months was one of the exclusion criteria).10 However, the drug is indicated for populations showing an unsatisfactory response to prior treatment, as well as in de novo patients with severe and rapidly evolving disease.2 This being the case, effectiveness studies of this drug in clinical practice have included patients whose baseline condition is more severe, and results for effectiveness from these studies continue to confirm and highlight the efficacy observed in pivotal trials.
An example is provided by the results from the study of natalizumab effectiveness in Spanish clinical practice (the GEXNE study).4 That study presented a retrospective analysis of the effect of natalizumab treatment over one year in 825 patients. Median decrease in ARR was 88% compared to the pre-natalizumab state; median score on EDSS dropped from 3.5 to 3.0, and 63% of the patient total remained free from disease activity. Furthermore, 23% of these patients demonstrated clinical improvement. Another Spanish study in 112 patients showed a 99% reduction in the number of gadolinium-enhancing lesions after one year.15 These results coincide with those obtained in studies of other cohorts from different countries.6,16,17 Researchers have also observed improvements in some of the typical symptoms of RRMS, such as fatigue, depression, and drowsiness.18
Another example is the recent study by Prosperini et al.5 proposing not only to describe the effectiveness of that drug in Italian clinical practice, but also to describe the profile of patients most likely to obtain maximum benefits from the drug (that is, remain free from disease activity). This study also confirms that natalizumab is highly effective, especially when treatment is started in early stages. The study concludes that the subpopulation of patients with fewer than 2 relapses in the year prior to treatment, and an EDSS of less than 3.0 at the beginning of the study, were 3 times more likely to experience no disease activity during treatment with natalizumab. In the study's patient subgroup (n=70), 77% responded completely to treatment; this rate was 57% in patients with an EDSS between 3.0 and 4.5, and 44% in those with an EDSS higher than 5.0.
This team's findings for natalizumab5 have been corroborated in an intermediate analysis (n=4434 patients) in the Tysabri Observational Programme for normal clinical practice. These data were presented at the 2013 ENS Congress. The findings show that natalizumab was more effective at reducing ARR in patients who began treatment with a baseline EDSS<3.0 (P<.0001).8 Researchers also observed a significantly greater decrease in ARR in patients who experienced≤1 relapses in the year prior to natalizumab treatment (ARR=0.23) than in patients who had experienced more numerous relapses (ARR=0.28; P<.0001).8 This study also demonstrated that natalizumab was more effective at decreasing ARR in de novo patients than in patients previously treated with one or more disease-modifying drugs.8
These studies, and others indicating that changing to natalizumab is more effective than changing between different immune modulators,19–21 all suggest that early treatment with natalizumab is more beneficial than using the drug as rescue therapy at later stages.
Ideal candidates for natalizumabNatalizumab (Tysabri®) has been approved by the EMA and the Spanish Agency for Medicines and Medical Devices as treatment for RRMS in the 2 groups of patients described below.2
- •
Group 1: adults aged 18 and over with high disease activity despite treatment with an interferon beta or glatiramer acetate.
These patients may be defined as those who have failed to respond to a full and adequate course (normally at least one year of treatment) of interferon beta or glatiramer acetate. Patients should have had at least 1 relapse in the previous year while on therapy, and display at least 9 T2-hyperintense lesions in cranial magnetic resonance image (MRI) or at least 1 gadolinium-enhancing lesion. A ‘non-responder’ could also be defined as a patient with an unchanged or increased relapse rate or ongoing severe relapses, as compared to the previous year.
- •
Group 2: adults aged 18 and over with rapidly evolving severe relapsing-remitting multiple sclerosis defined by 2 or more disabling relapses in one year, and with 1 or more gadolinium-enhancing lesions on brain MRI or a significant increase in T2 lesion load as compared to a previous recent MRI.
Studies of the natural history of the disease22,23 show that relapses have an impact on the progression of disability. Another study23 reveals significant differences in the disability progression rate when patients experience a relapse in the first 2 years of the disease (better prognosis) than when they have multiple relapses (poorer prognosis). Patients in the latter group reach an EDSS of 3.0 almost twice as quickly as those in the former group.
Equally, in the context of the benefits provided by natalizumab, we cannot ignore the implications this has for the public health system in terms of cost-effectiveness; in MS, costs associated with relapses, and with associated disability, are considerable. One of the parameters used in comparing the effectiveness of different drugs in an objective way is number needed to treat (NNT), that is, the number of patients who must be treated in order for an additional patient to receive the desired benefit, for example, avoiding a relapse. The greater the treatment efficacy, the lower the NNT. As we explained in the 2011 consensus statement, the NNT for the objective of avoiding a relapse in the first year of treatment is 2, meaning that only 2 patients need to be treated in order to prevent one relapse. This NNT is lower than for all other current treatments for RRMS. Natalizumab has been shown to be the most effective option for treating RRMS.24–27
The benefits of natalizumab must be weighed against the risk of adverse effects, which can now be more easily managed thanks to the increasing number of tools available. Natalizumab treatment must also be weighed against the risk of disability associated with RRMS itself. As stated previously, in the evaluation of benefits associated with a treatment, low NNT indicates a good treatment response (treatment of just a few patients is sufficient to evidence a good treatment response). Evaluation of the risks associated with treatment is based on the number needed to harm (NNH), which is calculated using concomitant serious adverse events, significant laboratory changes, treatment drop-outs, and the probability of unknown adverse events in the future. Natalizumab is a well-tolerated and safe drug, but it has been associated with increased risk of PML in some patients. Therefore, in the context of a risk/benefit analysis for natalizumab, NNH refers to the probability of developing PML, which varies and is measurable in the case of each individual subject, as described in a later section.
It is necessary to present information about risks in a way that each patient will understand, using specific examples to which the patient can easily relate. In general, patients are more open to running risks than neurologists.28 Evidence shows that patients attach more importance to progression time to disability than to the likelihood of developing PML,29 and natalizumab as a treatment option has demonstrated excellent efficacy results in pivotal trials.4–6
Lastly, risk/benefit analyses of treatment for RRMS should contemplate the risks posed by the disease itself, in both the short term (control over disease activity) and the long term (risk of disability progression, decrease in life expectancy). Each patient's risk of disability caused by MS may be assessed according to the presence or absence of factors associated with poor prognosis. Important factors include residual disability after a relapse,22,23 time between relapses, and lastly, the presence of lipid-specific oligoclonal IgM bands in cerebrospinal fluid (CSF),30 which has been associated with poor long-term prognosis.31 The neurologist has the responsibility of informing the patient of the seriousness of the disease so that the latter is better able to evaluate the risk/benefit profile of treatment in his or her personal case.
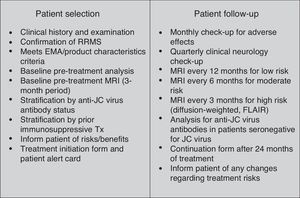
Beginning treatment with natalizumabAfter having defined the ideal patient for starting treatment with natalizumab, we will dedicate this section to the requirements that will make patient selection and treatment onset feasible (Fig. 1).
- •
Confirmation of RRMS based on medical history and examination.
- •
Evaluation of criteria listed by EMA and the summary of product characteristics.
- •
Ruling out any contraindications for treatment specified in the summary of product characteristics. Doctors must confirm that patients are not immunodeficient at the time of treatment onset; this is tested with a laboratory white blood cell count.
- •
Complete blood count and biochemical study. If liver enzymes are abnormal but have caused no symptoms prior to treatment onset, we recommend an in-depth study to determine the origin of the alteration. If doctors opt to start treatment, the patient's liver enzymes must be monitored over the treatment duration.
- •
MRI study in the 3 months prior to treatment onset.2 This MRI image will serve as the baseline study in case it needs to be compared to another MRI image taken during treatment with natalizumab. This radiological method of evaluating the effectiveness of natalizumab can also be used to confirm suspected PML.
- •
Anti-JC virus antibody testing. Assay of anti-JC virus antibodies: performed with Stratify JCV® (2-step ELISA study) and validated for this purpose in patients with MS. The original version of the assay, available in Spain since 2011, has been perfected to create a second-generation test9 that has been used since March 2012. The second-generation assay features a significantly more sensitive technique that delivers a higher negative predictive value for the assay.
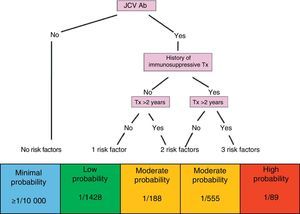
A seronegative patient (no anti-JC virus antibodies) is unlikely to have the virus, and therefore has an extremely low probability of developing PML (≤1/10000).1,32 However, it should be stressed that ‘Anti-JC virus antibody negative patients may still be at risk of PML for reasons such as a new JC virus infection, fluctuating antibody status or a false negative test result’. This statement was recently added to the summary of product characteristics. This probability in a seronegative patient is minimal, but it does exist in the following situations: (i) a false negative test result (the rate of false negatives, estimated from an analysis of 1319 patients, is 2.2%9); and (ii) a seronegative patient who comes into contact with JC virus for the first time and becomes seropositive (seroconversion), or a seronegative patient whose antibody levels fluctuate near the cut-off point for negative and positive values, and who may give a positive result on a later test (intermittent positive). Of the patients in the STRATIFY-1 study who were seronegative at baseline (n=328) and monitored during a 12-month period, 10.7% underwent seroconversion from negative to positive.33,34
If the patient does present anti-JC virus antibodies (seropositive), that patient has been exposed to JC virus.32 The probability of developing PML ranges from low to medium or high depending on the other 2 risk factors: prior immunocompromise and time in treatment.1
- •
Stratification based on prior immunosuppressant treatment. A history of treatment with an immunosuppressant drug is associated with a two- to threefold risk of developing PML.2
- •
The patient must be duly informed of treatment risks and benefits.
- •
We recommend asking the patient to sign a form at the onset of treatment with Tysabri® as suggested by the Physician Information and Management Guidelines for Multiple Sclerosis Patients on Tysabri®. Furthermore, the patient should be provided with a ‘Patient Alert Card’.
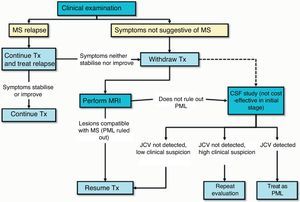
After onset of treatment, patients should be monitored closely to prevent complications, especially appearance of PML (Fig. 1).
- (a)
This includes identifying any of the following factors that may increase the probability of developing PML so as to monitor them appropriately during treatment2:
- 1.
Presence/absence of anti-JC virus antibodies.
- 2.
Presence/absence of history of immunosuppressant treatment.
- 3.
Duration of patient's treatment with natalizumab: the probability of PML in a seropositive patient is low if the patient has been undergoing treatment for less than 2 years, but higher given longer treatment periods.
Although development of anti-JC virus antibodies is unquestionably one of the main factors affecting the risk/benefit assessment for the treatment, this is not the only reason for withdrawing or not administering natalizumab.
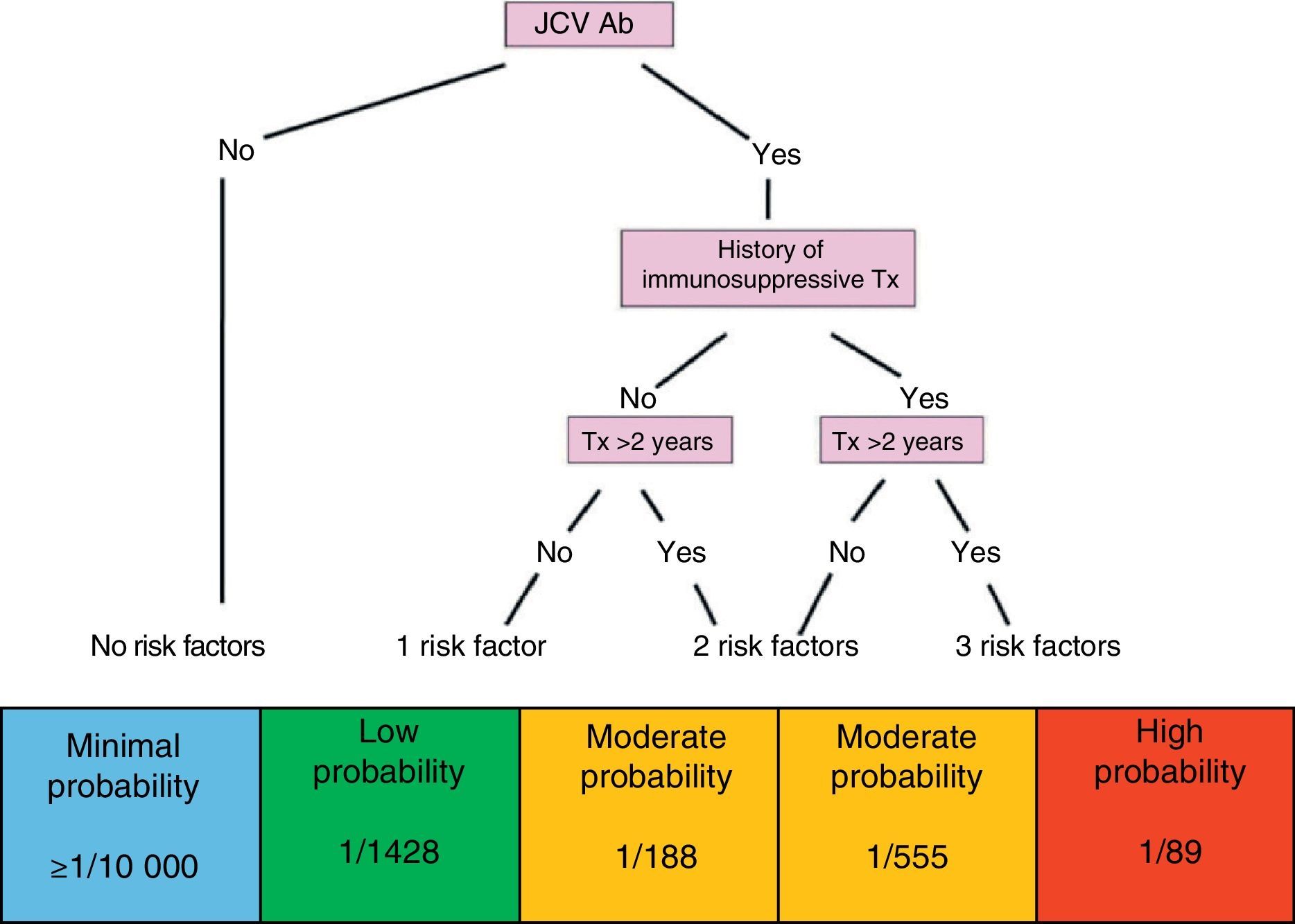
Through identification of all of the above factors, and without losing sight of the risk inherent to the disease itself in each case, patients may be categorised in the risk groups described below (Fig. 2).
- •
Patient without risk factors: seronegative patient with a minimal likelihood of developing PML (≤1/10000).
- •
Patient with one risk factor: seropositive patient; low probability of developing PML (1/1428).
- •
Patient with 2 risk factors: seropositive patient with a history of immunosuppressant treatment; moderate probability of developing PML (1/555).
- •
Patient with 2 risk factors: seropositive patient treated with natalizumab during more than 2 years; moderate probability of developing PML (1/188).
- •
Patient with 3 risk factors: seropositive patient with a history of immunosuppressant treatment and more than 2 years of treatment with natalizumab; high probability of developing PML (1/89).
Classifying patients treated with natalizumab according to their probability of developing PML is an important step in monitoring their situation during treatment.
- (b)
Monitoring patients treated with natalizumab.
- 1.
Hypersensitivity reactions: hypersensitivity may present in 4% of the patients treated with the drug, and reactions are more likely to manifest with the second dose.2 During every infusion, nurses or doctors must watch closely for any reactions to the drug (fatigue, dizziness, headache, asthenia, nausea, etc.) and be able to distinguish them from allergic reactions or hypersensitivity (urticaria with or without systemic repercussions).
- 2.
Development of persistent neutralising antibodies to natalizumab may occur in 6% of all patients; this process occurs most commonly in the first 6 months of treatment. If the drug is not effective, or if doctors observe infusion-related events, checking for neutralising antibodies is recommended. If the result is positive, the analysis should be repeated 4 weeks later. If there is a second positive result (persistent antibodies), discontinuing treatment is recommended. Presence of antibodies is linked to loss of clinical efficacy of natalizumab and increased incidence of hypersensitivity reactions.2,35
- 3.
Monitoring liver function: patients who begin treatment with asymptomatic liver alterations should undergo monthly analyses during the first 3 months, and subsequently every 3 months if values stabilise. Liver alterations rarely appear after the treatment has been started. If an elevation in liver transaminases causes symptoms, we suggest temporarily suspending the drug and investigating the cause. If no symptoms are associated with elevated transaminases, we recommend clinical and analytical monitoring. However, for levels exceeding 3 times the normal level, we recommend temporarily suspending the drug, investigating other causes, and not resuming treatment until laboratory results are normal.
- 4.
Monitoring JC virus serology: to check for seroconversion, seronegative patients should be tested for anti-JC virus antibodies every 6 months.2
- 5.
Informing the patient of changes in the risk/benefit analysis of natalizumab at the 2-year mark: we recommend having the patient sign the informative leaflet for continuing treatment with Tysabri® as suggested by the EMA in its Physician Information and Management Guidelines for Multiple Sclerosis Patients on Tysabri®, as well as informed consent (the latter is optional). Beginning at this moment, this information should be revised annually and any time new evidence about treatment risks is discovered.
- 6.
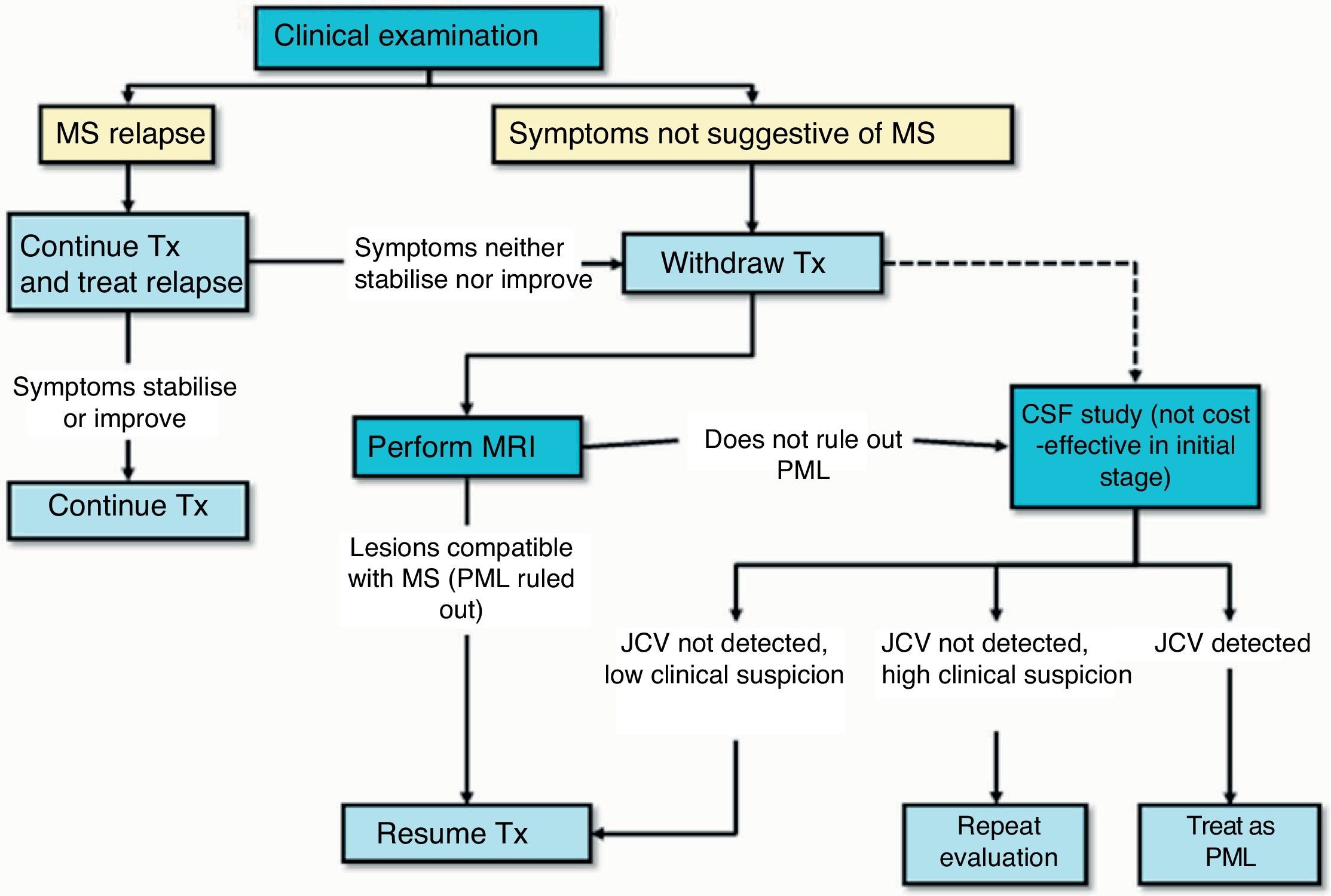
Clinical monitoring: nurses or doctors should be present during every infusion (every 4 weeks) to watch for the appearance of any clinical symptoms or complications related to treatment, especially PML. Clinical monitoring, including a neurological examination, should be performed every 3 months or if new symptoms appear, to determine if those symptoms are compatible with an outbreak of disease or indicative of PML (Fig. 1).2 Clinical monitoring should also continue for at least 6 months after treatment withdrawal since cases of PML have been described in patients showing no findings suggestive of PML at the time treatment was discontinued.2
- 7.
Radiological monitoring: a complete head MRI scan should be performed yearly (T1- and T2-weighted sequences including contrast) on all treated patients to detect the appearance of new MS lesions. This yearly MRI scan also serves as a screening method for PML in patients at low risk for the disease.1 In addition, for patients at moderate to high risk, routine MRI scans must be performed every 6 months (3–4 months in high-risk patients) so as to provide early diagnosis. If a complete head MRI cannot be performed that frequently, check-ups may be performed using diffusion or FLAIR sequences to show any PML lesions. Reports show that patients with an early diagnosis of PML may have a better outlook.36 Lastly, a brain MRI should ideally be performed if any neurological symptoms or signs appear that might suggest either a relapse or symptoms related to onset of PML.
- 8.
Action protocol in cases of suspected PML (Fig. 3): suspected diagnosis is based on clinical and MRI findings, and the disease is confirmed when the JC virus is detected in CSF. Table 1 describes the radiological characteristics that orient us towards a diagnosis of suspected PML.37 If PML is suspected, doctors must immediately inform the therapeutic drug monitoring department at Biogen Idec España (the company that markets the drug) for swift activation of the process to confirm or rule out PML. This process includes analysis of a CSF sample to detect JC virus-specific DNA in the main laboratory certified for that activity. A positive result on the test confirms the diagnosis of PML. The pharmaceutical company also provides a second opinion on the suspicious MRI scan in order to better describe and confirm cases. If PML is suspected but not yet confirmed, the drug must be removed from the body immediately using plasmapheresis or immunoabsorption, as indicated in the Physician Information and Management Guidelines for Multiple Sclerosis Patients on Tysabri®, which was recently revised and updated by the EMA.
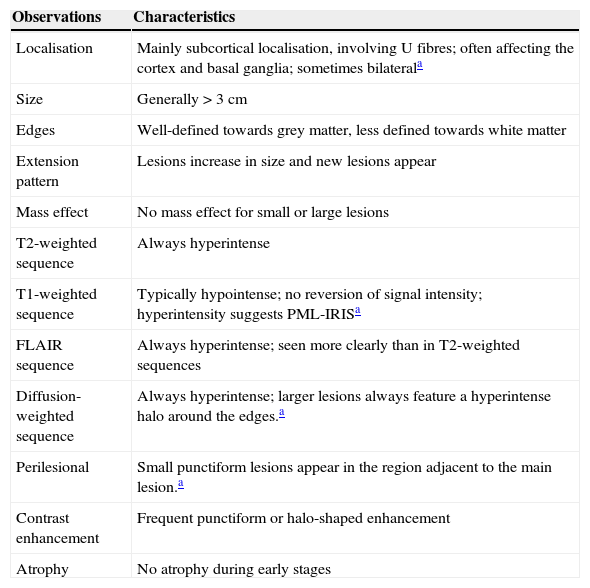
Table 1.Recommendations for early diagnosis of progressive multifocal leukoencephalopathy in multiple sclerosis patients treated with natalizumab
Observations Characteristics Localisation Mainly subcortical localisation, involving U fibres; often affecting the cortex and basal ganglia; sometimes bilaterala Size Generally>3cm Edges Well-defined towards grey matter, less defined towards white matter Extension pattern Lesions increase in size and new lesions appear Mass effect No mass effect for small or large lesions T2-weighted sequence Always hyperintense T1-weighted sequence Typically hypointense; no reversion of signal intensity; hyperintensity suggests PML-IRISa FLAIR sequence Always hyperintense; seen more clearly than in T2-weighted sequences Diffusion-weighted sequence Always hyperintense; larger lesions always feature a hyperintense halo around the edges.a Perilesional Small punctiform lesions appear in the region adjacent to the main lesion.a Contrast enhancement Frequent punctiform or halo-shaped enhancement Atrophy No atrophy during early stages FLAIR: fluid attenuation inversion recovery sequence; PML: progressive multifocal leukoencephalopathy; IRIS: immune reconstitution inflammatory syndrome.
Source: Yousry et al.37
Natalizumab treatment may have to be interrupted for a variety of reasons (for example, hypersensitivity reactions, development of persistent anti-natalizumab antibodies, lack of efficacy, or when risk/benefit ratio is found to be negative). This leads to uncertainty among neurologists for different reasons, many of which have to do with concerns that disease activity may exacerbate.
A review of the different studies published since 1999 indicates that while disease activity that reappears after natalizumab withdrawal is variable, it generally does not exceed the baseline level that the patient experienced prior to treatment with the drug. This is similar to the situation that arises during pregnancy.
The largest study conducted on this topic is a retrospective analysis of patients from the clinical trials AFFIRM, SENTINEL, and GLANCE (n=1866), with a follow-up period of 8 months after withdrawal of the treatment.38 Based on this study, we conclude that most patients will return to their baseline activity level. However, the subanalysis of patients with highly active MS, defined as having one gadolinium-enhancing (Gd+) lesion and ≥2 exacerbations in the year prior to receiving natalizumab treatment, shows that these patients will experience a more rapid and severe increase in activity. One recently published study (n=105) describes serious exacerbations in 10% of its patients, and this is correlated with a higher ARR and elevated activity shown by MRI (Gd+) in the year prior to treatment.39
A serious increase in clinical and radiological activity occurring after treatment has been discontinued, and which is greater than the patient's baseline activity, is known as the ‘rebound effect’. The rebound effect may occur due to an increase in activated lymphocytes and proinflammatory cytokines in the blood that pass through the BBB when natalizumab is withdrawn.40 Baseline activity is understood as the level of activity that the patient experienced while receiving no treatment; many patients who discontinue natalizumab treatment return to the same level of activity. Doctors commonly refer to ‘prenatalizumab activity’, but at this point, the patient is probably under the effects of a prior immunomodulatory treatment that masks real baseline activity; in this way, prenatalizumab activity is probably lower than postnatalizumab activity, when the patient is receiving no treatment at all.
Even so, daily clinical practice shows that some patients experience disease exacerbation, and not only after withdrawal of natalizumab (there are also reports of a similar phenomenon with fingolimod41–43). Further studies are needed to identify the profile of patients most susceptible to the rebound effect, as well as a treatment strategy that will provide effective control. Regarding this last point, none of the treatments used to date have been shown to be effective at controlling disease activity in these cases. This includes the latest treatment to be marketed for RRMS, fingolimod; 4 published studies have shown that it is not fully effective in cases of rebound effect.44–47 Two of these studies compare fingolimod with no treatment in one case46 and with immunomodulatory treatment in another.47 As might be expected, they concluded that treating is better than not treating, that fingolimod may be more effective than the immunomodulator, and that it may be beneficial to start fingolimod treatment before the 3-month wash-out period needed by natalizumab, even though this approach will entail simultaneous exposure to the 2 drugs. Fingolimod's summary of product characteristics anticipates this ‘head start’ by estimating a period of concomitant exposure of 2 to 3 months if fingolimod is begun immediately after suspending natalizumab, and experts recommend using this option with caution.
At present, the summary of product characteristics for natalizumab indicates a wash-out period of 3 months prior to starting another treatment, and this recommendation is justified by the duration of the drug's pharmacodynamic effect. However, the growing tendency is to begin the following treatment before closure of the wash-out period, in an attempt at limiting the reappearance of disease activity and allowing the patient to benefit from the therapeutic effect of the drug. Regarding use of immunomodulatory drugs, interferons, or glatiramer acetate, the overwhelming consensus is that treatment should be started as soon as possible, and some say immediately (provided that drugs have not failed previously). However, this option is not completely supported by the summary of product characteristics, which reads ‘for medicinal products such as interferon and glatiramer acetate, concomitant exposure of this duration was not associated with safety risks in clinical trials. Use of these medicinal products soon after the discontinuation of natalizumab may lead to an additive immunosuppressive effect. This should be carefully considered on a case-by-case basis, and a wash-out period of natalizumab might be appropriate’. This being the case, patients should give written consent for this treatment.
Management of progressive multifocal leukoencephalopathyPML is a severe complication entailing high rates of both mortality (20%) and morbidity. Reports show that cases in younger patients with lower pre-PML EDSS scores are associated with higher probabilities of survival.44
The best treatment for PML involves early diagnosis of that condition. To achieve this end, we recommend extensive monitoring, following currently available recommendations, and ordering as many MRI scans as are necessary.1,48–50
PML associated with natalizumab treatment is aetiologically and clinically distinct from the type of PML associated with HIV. It can be explained by the blocking effect of natalizumab on lymphoid cell access to within the central nervous system. This situation gives rise to a lack of immunological protection in cases of brain infection with JC virus. For this reason, the first step in treating PML associated with natalizumab is rapid elimination of the drug from the blood to restore immune system access to the central nervous system and thereby target the JC virus infection. Treatment approaches include plasmapheresis and immunoabsorption in different dosing regimens.2,51
Other attempts at treating PML have included numerous antiviral drugs (cidofovir), 5HT2A receptor antagonists (mirtazapine), or the antimalarial drug mefloquine. However, no conclusive results about their efficacy are available.
Immune reconstitution inflammatory syndrome (IRIS) is present in most cases of PML and appears several days to few weeks after administration of plasmapheresis or immunoabsorption. It is characterised by the appearance of neurological impairment together with obvious inflammatory signs in neuroimaging. Without proper management, it may cause very severe neurological damage or death. Although there is no consensus regarding IRIS prevention and treatment, abundant clinical evidence, most of which comes from experience with patients with HIV, suggests that high doses of intravenous corticosteroids may be useful in both preventing and treating IRIS.
Latest news on identifying markers for personalised treatment with natalizumab- (a)
Anti-JC virus antibody index: recently published data suggest that the anti-JC virus antibody titre may be useful for determining the probability of developing PML. In these studies, levels of anti-JC virus antibodies were higher in patients who developed PML than in those who did not.52,53 In light of these findings, a study54 is now underway to test the hypothesis that levels of anti-JC virus antibodies might provide more information about the probability of developing PML in seropositive patients: that is, identifying which of those patients who have had contact with JC virus at some point during their lives have the greatest risk of developing PML. Preliminary results from the study have now been presented in international congresses. In the patient population with no previous immunosuppressive treatment, the antibody index (index value delivered by the second-generation assay) was significantly higher in patients who developed PML (n=51) than in patients who did not (n=2242). According to the preliminary results, these differences were not observed in patients who had been treated with immunosuppressive drugs.
Regarding the study population with no prior immunosuppressive treatment, of the 51 PML cases, one had an index ≤0.9, 6 had an index<1.5, and the remaining 44 had an index≥1.5. According to this study, the estimated probability of developing PML in a seropositive patient with no history of immunosuppressive drugs and a 5 to 6-year history of treatment ranges from 0.4 to 1.3/1000 patients (95% CI [0.01–2.15] to [0.41–2.96]) for indexes from ≤0.9 to ≤1.5 respectively. The probability in patients with an index>1.5 would be 8.5/1000 patients (95% CI [6.22–11.38]). In the study population, 42.9% of the seropositive patients who did not develop PML had an index ≤1.5.
If this hypothesis can be validated by a study, it will show that the anti-JC virus antibody index provides information about the change in serological status from negative to positive. This will be helpful for differentiating between changes in serological status due to seroconversion (high index) or to fluctuating antibodies (low index). Furthermore, it may help to better differentiate risk between these 2 types of patients. This study of the utility of the anti-JC virus antibodies index as a tool for stratifying patients positive for anti-JC virus is still underway, and its goal is to confirm results in a larger sample.
If this result is delivered, the antibody index in a seropositive patient will be a key finding and end the assumption that ‘treatment time longer than 2 years’ is a risk factor for seropositive patients with a low index. Likewise, patients positive for anti-JC virus with a low index will need to have their antibody index measured periodically to make sure that the index remains low.
- (b)
Presence of lipid-specific IgM oligoclonal bands (IgM OCBs) in CSF: the presence of immunoglobulin M (IgM) in CSF has been suggested as a factor to examine when estimating the probability of developing PML. The presence of IgM OCBs has already been described as marker of poor prognosis in the disease, and IgM OCBs specific for lipid antigens are particularly important.55 The same authors have researched whether presence of lipid-specific IgMs in CSF could be useful for estimating the patient's probability of developing PML. Data which remain unpublished, but which have been presented in national and international congresses,56 suggest that patients who are seropositive for anti-JC virus antibodies and develop PML did not have lipid-specific IgM OCBs in CSF. If this finding is confirmed in other cohorts and validated clinically, measuring IgM OCBs in CSF may be another factor to consider when estimating probability of developing PML.
- (c)
An additional potential biomarker of PML risk, which remains to be validated, is the percentage of CD4+ cells expressing L-selectin. This percentage has been observed to be significantly lower (9 times lower) in patients who later developed PML.57
In light of the findings described above, the presence of anti-JC virus antibodies should not exclude treatment with natalizumab. The fact that research is still underway is encouraging because increasing numbers of patients may benefit, and more safely, from the effects of natalizumab as researchers refine their understanding of the patient profile associated with a higher risk of PML. In cases of patients treated with natalizumab and showing no disease activity, and who develop anti-JC virus antibodies, experts recommend working closely with the patient and making assessments of disease severity the top priority. It is clear that discontinuing natalizumab must be justified, and that the risk of treatment should exceed the risk of the disease in this case.
As of 31 March 2013, approximately 118100 patients worldwide have been treated with natalizumab since it was first made available in the United States in 2004 (data provided by Biogen Idec).
The multinational Tysabri Observational Programme, in which 13 Spanish hospitals participate, records safety and effectiveness data for long-term treatments in the context of normal clinical practice. Results from intermediate analyses are updated and presented yearly in international congresses. At 1 December 2012, 4821 patients had been included in the study. The median ARR decreased from 1.99 to 0.30 (0.28–0.32) in the first year and remained low (0.24) at 5 years of treatment. In the total population, median EDSS remained stable after 4 years of treatment. Out of 2224 patients in follow up during at least 2 years, 1247 (56%) showed no clinical signs of disease activity. This was also true of 530 patients out of 1000 (53%) monitored for 3 years, and 174 patients out of 355 (49%) monitored for 4 years.8
Another study that evaluated the long-term safety and efficacy of natalizumab was named Safety of Tysabri® Re-dosing and Treatment. This study was carried out in patients from phase-3 drug trials who resumed treatment after the same drug had been withdrawn by the laboratory that developed it; this was due to the appearance of the first case of PML (February 2005–March 2006). Cumulative exposure to the treatment in these patients during the postmarketing period extended to nearly 7 years.58 The drug's safety profile is coherent with data gathered in normal practice with that drug during the post-marketing phase. The resulting ARR is consistent with rates observed in the AFFIRM study, and it remains low during 5-year treatment periods. Furthermore, the EDSS score remained stable or decreased. Once again, researchers observed a lower ARR and an improved EDSS score in those patients randomly selected for the natalizumab arm compared to those in the placebo arm. This underlined the benefits of early versus delayed treatment.
ConclusionsAs we reported in the first consensus statement, natalizumab remains the most effective drug for patients with RRMS, despite the fact that new treatments were authorised by both consensus statements. According to the summary of product characteristics, it is considered the first option not only after treatment failure of a conventional immunomodulatory drug, but also for cases of serious RRMS that progress rapidly from onset. Treatment benefits should be weighed against the risks, and especially those inherent to disease progression and the probability of developing PML. The current situation is encouraging. Results continue to show that natalizumab is highly effective in clinical practice over the short, medium, and long term; also, the associated risks are better understood (referring to risks involved in both treatment and postponing treatment). Research is delivering increasingly accurate measurements of the risk/benefit ratio in each patient.
Conflicts of interestOscar Fernández has received fees as a consultant to committees and as a moderator or speaker at medical congresses and symposia. He has also participated in clinical trials and other research projects promoted by Biogen Idec, Bayer Schering Pharma, Merck Serono, Teva, and Novartis.
Xavier Montalbán has received fees and travel expenses for attending meetings as a speaker. He has acted on the steering committees of clinical trials, and has also recently collaborated with clinical trial committees as a consultant with Bayer Schering Pharma, Biogen Idec, EMD/Merck Serono, Genentech, Genzyme, Novartis, Sanofi-Aventis, Teva, and Almirall.
The other authors have no conflicts of interest to declare.
We would like to thank Dr Greg Morley for his valuable input.
Please cite this article as: Fernández O, García-Merino J, Arroyo R, Álvarez-Cermeño JC, Izquierdo G, Saiz A, et al. Consenso español actualizado sobre el uso del natalizumab (Tysabri®)-2013. Neurología. 2015;30:302–314.