Chronic neuropathic pain can be highly refractory to both conventional and percutaneous analgesic treatment. Clinical management of these patients is a challenge due to the limiting nature of their pain, loss of confidence after successive consultations with different specialists, and low expectations of improvement given the apparent lack of effective treatment options. We should note the availability of an alternative treatment which merits greater recognition than it currently receives, despite advances made in recent years.
We present the case of a 43-year-old patient diagnosed with the Samter triad with severe nasal polyps, complicated by recurrent sinusitis, which she had experienced since childhood. The patient had undergone 17 procedures at her local hospital's otorhinolaryngology department, including endoscopic procedures and frontal osteoplasty, which was reviewed several times by the surgical team following onset of inflammation and local pain. She was referred to our clinic due to a 2-year history of chronic, disabling pain involving the supraorbital notch and the right lateral frontal region. The patient reported constant, burning pain. The physical examination revealed the Tinel sign and areas of primary mechanical hyperalgesia and allodynia; the patient was diagnosed with secondary supraorbital neuralgia. The patient's symptoms were refractory to both pharmacological and percutaneous treatment (radiofrequency supraorbital nerve block). The patient scored the maximum possible on the pain disability index (PDI) and visual analogue scale (VAS). The presurgical psychiatric evaluation only indicated an adaptive emotional response to pain. A cylindrical electrode (Precision® SC-2352-50, Boston Scientific) was implanted into the right frontal region under local anaesthesia (Fig. 1). During the procedure, the patient reported paraesthesia in the area receiving stimulation; 7 days later, pain had improved significantly (PDI, 32; VAS, 4). A generator (Precision® SC-1110-02, Boston Scientific) was subsequently implanted under general anaesthesia. Pain had resolved completely at 6 months (PDI, 0; VAS, 0) and continued to be absent at one year of follow-up.
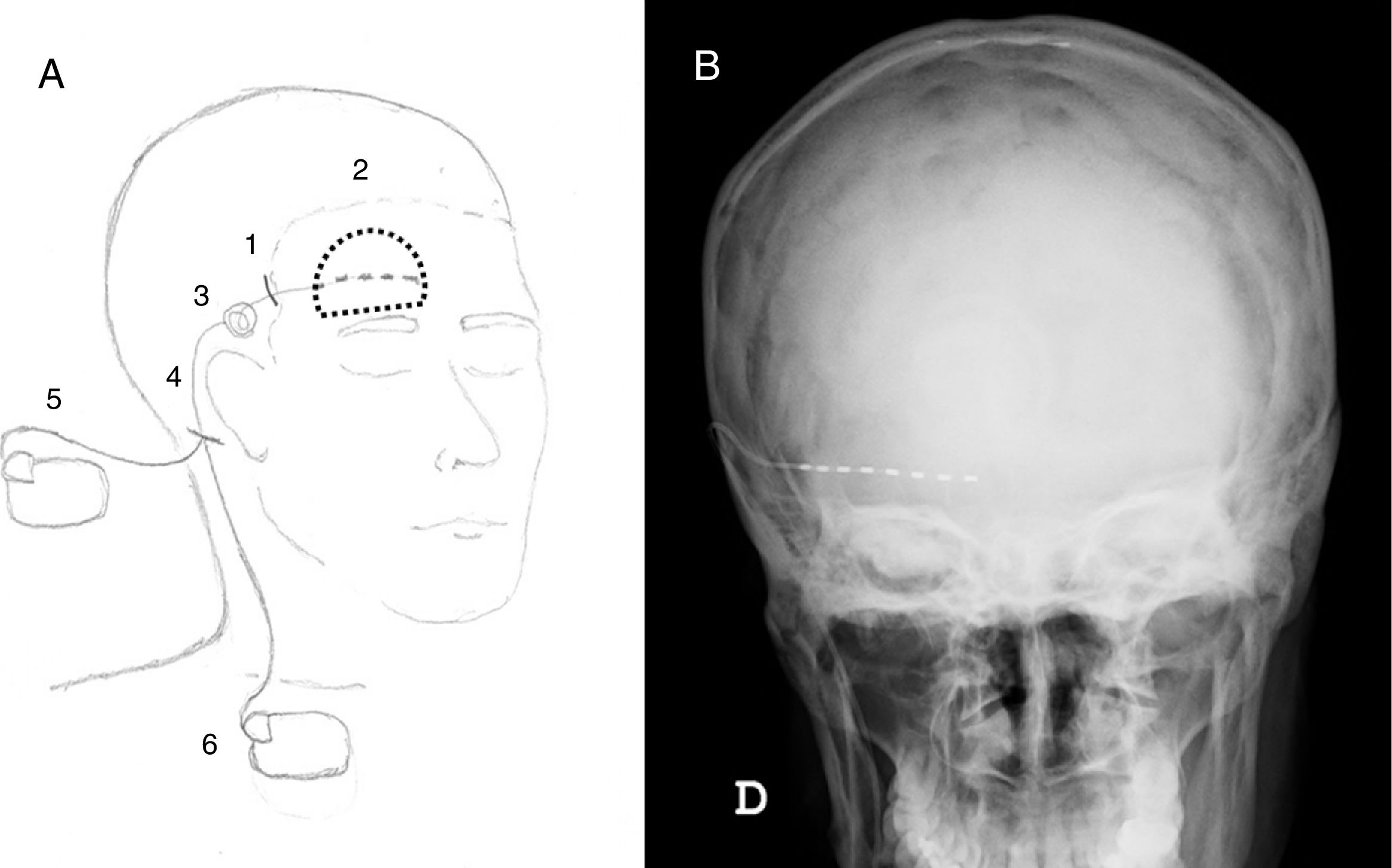
(A) Diagram illustrating the procedure by which the nerve stimulation system is implanted: (1) an incision is made in the scalp; (2) the distal end of the electrode is implanted below the painful area (dotted line); (3) the device is anchored to the temporal fascia and a discharge circuit is created; (4) a retroauricular incision is made; (5) in the trial phase, the terminal is external; (6) subsequently, the generator is implanted subcutaneously in the subclavicular region. (B) Skull X-ray, antero-posterior view showing the final position of the electrode.
Peripheral nerve field stimulation (PNFS) has for decades been a therapeutic alternative meriting consideration for use in patients with various types of refractory pain. This technique is more easily reversed and less invasive than other surgical options,1 and has been used successfully in patients with neuropathic pain (postherpetic neuralgia, trigeminal neuralgia, occipital neuralgia, traumatic neuralgia), complex regional pain syndrome, axial back pain, headache, and even musculoskeletal pain, fibromyalgia, and visceral pain.2 Clinical use of the technique, based on the gate control theory of pain, was first described in 1967 by Patrick D. Wall and William H. Sweet,3 who had confirmed its effect by testing it on themselves.4 However, it should be noted that most evidence on the subject is from retrospective case series and case studies; few randomised controlled trials have been performed.1,2
The success of the technique relies on careful patient selection. Patients with anaesthesia in the area to be stimulated are ineligible for treatment, as are patients with diffuse allodynia, which may be exacerbated by the technique. Similarly, patients with active psychiatric disorders, cognitive impairment, or who are involved in legal proceedings should not be treated with PNFS. It is also essential to consider history of bleeding diathesis, as well as active infections or immunosuppression. Response to percutaneous treatment is not predictive of the success of PNFS. A 2- to 14-day trial period is recommended; a decrease of at least 50% in PDI and VAS scores is considered a significant improvement.2 This trial period also enables a potential placebo effect to be evaluated. Possible complications include lead migration (15.6%), mechanical failure (11%), infection (4.4%), skin erosion (2.2%), and loss of effectiveness of the treatment over time (4.7%-17%).2
Please cite this article as: Carrasco-Moro R. Estimulación de nervio periférico: una alternativa terapéutica eficaz para el dolor refractario. Neurología. 2019;34:345–346.