Envenomation by black widow spiders manifests clinically with signs of neurotoxicity in paediatric patients.
ObjectiveIdentify typical neurological signs and symptoms in paediatric patients of different ages, and describe treatment and outcomes in a paediatric hospital in northwest Mexico.
Material and methodsWe reviewed 70 clinical records of patients hospitalised due to black widow spider bite between 1978 and 2014. We divided the total into 2 groups: Group 1, infants and preschool children; and Group 2, school-age children and adolescents. The demographic variables were age, sex, birthplace, place where envenomation occurred, body part(s) affected, degree of envenomation according to signs and symptoms, treatment, clinical outcome, and statistical differences.
ResultsBoys accounted for 61.4% of all cases, and infants younger than one year made up 14.2%. Most patients (70%) were bitten by the spider at home; the anatomical areas most frequently affected were the legs, neck, thorax, and abdomen. The neurological signs and symptoms displayed by Group 1 were irritability, constant crying, sialorrhoea, nausea, tachycardia, arrhythmias, fatigue when walking, agitation, muscle spasms paraesthesia, tetany, seizures, and nystagmus. Signs in Group 2 included localised pain, headache, sialorrhoea, paraesthesia, profuse sweating, anxiety, muscle weakness, muscle spasms, and fine tremor. The predominant autonomic sign in Group 1 was sialorrhoea (P<.0001) and in Group 2, paraesthesia (P<.0001). Patients who received Fab antivenom treatment displayed better outcomes and shorter hospital stays than those who did not. No deaths were reported.
ConclusionsThe neurological signs and symptoms caused by black widow spider bite are predominantly autonomic, and identifying them permits early diagnosis and more effective treatment.
El envenenamiento por mordedura de araña «viuda negra» (Latrodectus mactans) en niños se expresa clínicamente con neurotoxicidad.
ObjetivoIdentificar los signos y síntomas neurológicos característicos, en las diferentes edades pediátricas, la evolución y el tratamiento en pacientes atendidos por esta mordedura en un hospital pediátrico de noroeste México.
Material y métodosSe revisaron 70 expedientes de niños hospitalizados entre 1978-2014; estableciéndose 2 grupos: grupo 1, de 33 lactantes y preescolares, y grupo 2, con 37 escolares y adolescentes. Las variables consideradas fueron: edad, género, lugar de procedencia, sitio del accidente, área corporal afectada, grado de envenenamiento, tratamiento, evolución clínica, diferencias estadísticas.
ResultadosPredominó el género masculino, 61,4%; los lactantes menores de un año fueron un 14,2%. El 70% de los pacientes tuvieron el contacto con el arácnido dentro del domicilio; las áreas anatómicas más afectadas fueron miembros inferiores, cuello, tronco y abdomen; los signos y síntomas neurológicos más notables en el grupo 1 fueron: irritabilidad, llanto constante, náuseas, sialorrea, agitación, taquicardia, arritmias, incapacidad para caminar, espasmos musculares, parestesias, tetania, convulsiones, nistagmo. En el grupo 2 fueron: dolor local, cefalea, sialorrea, parestesias, sudoración profusa, ansiedad, debilidad muscular, espasmos musculares y temblor fino. La manifestación clínica autonómica predominante en el grupo 1 fue sialorrea, p<0,0001, y en el grupo 2, parestesias, p<0,0001. El uso de faboterápicos en el tratamiento permitió mejor evolución y menor tiempo de hospitalización. No hubo mortalidad.
ConclusionesLos signos y los síntomas de la mordedura por araña «viuda negra» son predominantemente autonómicos; identificarlos permite el diagnóstico oportuno y tratamiento eficaz.
The black widow spider (Latrodectus mactans) is an arthropod. It belongs to the order of the Araneae which, together with the order of the Scorpionidae, includes over 35000 species worldwide.1,2 The precise number of envenomation cases is unknown.
The venom of the black widow is more potent than that of rattlesnakes; it contains several toxins, including alpha-latrodectin, alpha-latrotoxin, latrocrustotoxin, latroinsectotoxin, and neurexin, all of which are proteins with an affinity for nerve terminals.
Neuronal latrophilins (LPHN) are a family of receptors that has not been studied in much detail. According to recent articles,3–5 the latrophilin family consists of 3 isoforms, LPHN1-3, and belongs to a unique branch of G protein-coupled receptors known as adhesion GPCR (aGPCR), the most important receptors of alpha-latrotoxin, which on binding stimulates massive neurotransmitter release. Experimental studies have shown that alpha-latrotoxin may be active on all types of vertebrate synapses, regardless of the neurotransmitter involved: acetylcholine, noradrenaline, dopamine, glutamate, and enkephalins. The action of this toxin can be classified as either Ca2+-dependent or Ca2+-independent. It should be noted, however, that these 2 action modes of alpha-latrotoxin only co-occur in interneuron synapses since only one mode, Ca2+-dependent, is present in the neuromuscular junction. The role of alpha-latrotoxin in neurotransmitter release suggests that the effect of this toxin on neuronal membranes may be a result of its interaction with specific receptors.3–5 Neurexins (NRXN) are involved in cell recognition within the nervous system.
Latrotoxin and latrodectin selectively interact with latrophilin and neurexin receptors in the lipid bilayer membrane of presynaptic terminals, resulting in the formation of cation channels allowing an influx of Ca2+ into the cell. Toxin activity causes the release of a large amount of such neurotransmitters as noradrenaline and acetylcholine, as well as calcium-independent gamma-aminobutyric acid, and has an important effect on the neuromuscular junction.4–7
Symptoms depend on the number of bites and the patient's age and physical condition, and tend to be more severe in young children and older adults.5–11
In Mexico, 11% of the total number of envenomations by poisonous animals are caused by spider bites. Every year, 3000 to 5000 people are bitten by black widow and brown recluse spiders (Loxosceles reclusa), especially the former.12–16 Interpreting clinical manifestations correctly and treating patients with suitable antivenoms prevents severe complications and reduces hospitalisation times.13–16 The present article describes our experience in a paediatric hospital in northwestern Mexico.
ObjectiveTo analyse the most common neurological signs and symptoms in a series of paediatric patients of different ages who visited our hospital due to Latrodectus mactans envenomation, and to describe our patients’ treatment and progression.
Working hypothesisClinical symptoms of neurotoxicity due to Latrodectus mactans bites may vary depending on the patient's age and must therefore be identified and described.
Material and methodsWe retrospectively reviewed the medical records of 70 patients who were admitted due to black widow spider bite to our paediatric hospital, Hospital Infantil del Estado de Sonora, between January 1978 and December 2014. We included the following variables: place of residence, age, sex, place where the incident took place, affected body area, treatment before admission, and time elapsed from bite to medical attention.
To analyse the different clinical manifestations, patients were classified into 2 groups: group 1 (infants and preschool children) and group 2 (school-age children and adolescents). We registered signs and symptoms, degree of envenomation, laboratory test results, treatment, hospitalisation time, complications, and progression. We used descriptive non-parametric statistics (Fisher exact test, Pearson's chi-square test, likelihood ratio test, correlation test) to express the results.
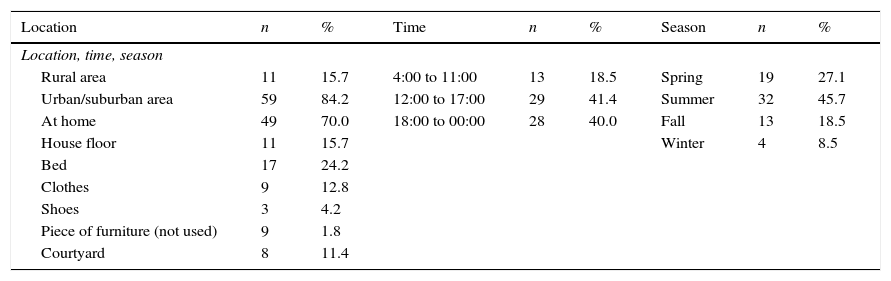
ResultsOur patient sample included 70 patients; of these, 43 (61.4%) were boys and 27 (38.5%) were girls. Patients’ ages ranged from 4 months to 18 years. Fifty-nine patients (84.2%) came from urban and suburban areas and the remaining 11 (15.7%) were from rural areas. Spider bites were more frequent during the summer months (32 patients, 45.7%) and occurred mostly at home between 12:00am and 12:00pm (Table 1).
Demographic and epidemiological data (n=70).
| Location | n | % | Time | n | % | Season | n | % |
|---|---|---|---|---|---|---|---|---|
| Location, time, season | ||||||||
| Rural area | 11 | 15.7 | 4:00 to 11:00 | 13 | 18.5 | Spring | 19 | 27.1 |
| Urban/suburban area | 59 | 84.2 | 12:00 to 17:00 | 29 | 41.4 | Summer | 32 | 45.7 |
| At home | 49 | 70.0 | 18:00 to 00:00 | 28 | 40.0 | Fall | 13 | 18.5 |
| House floor | 11 | 15.7 | Winter | 4 | 8.5 | |||
| Bed | 17 | 24.2 | ||||||
| Clothes | 9 | 12.8 | ||||||
| Shoes | 3 | 4.2 | ||||||
| Piece of furniture (not used) | 9 | 1.8 | ||||||
| Courtyard | 8 | 11.4 | ||||||
| Ages | Months/years | n | % | Affected area | n | % |
|---|---|---|---|---|---|---|
| Ages, location of bite | ||||||
| 4 to 11 | Months | 10 | 14.2a | Neck, trunk, and abdomen | 17 | 24.2 |
| 1 to 2 | Years | 6 | 8.5 | Arms | 8 | 11.4 |
| 1 to 5 | Years | 17 | 24.2 | Hands and fingers | 10 | 14.2 |
| 6 to 10 | Years | 18 | 25.7 | Legs | 19 | 27.1 |
| 11 to 18 | Years | 19 | 27.1 | Buttocks | 3 | 4.2 |
Spider bites were reported on the legs (19 patients, 27.3%); neck, chest, or abdomen (17 patients, 24.2%); feet (10 patients, 14.2%); arms (8 patients, 11.4%); hands and fingers (10 patients, 14.2%); thighs (3 patients); and buttocks (2 patients). For one of the patients, the affected body area was not specified.
Two patients were administered garlic and milk by their relatives, one patient received aspirin, and another one was given diphenhydramine. Regarding the time elapsed from bite to medical attention, 38 patients (54.2%) were attended within an hour, 22 patients (31.4%) between 2 and 5hours, 3 patients (4.3%) between 6 and 12hours, one patient was attended at 24hours, and 3 at 48hours.
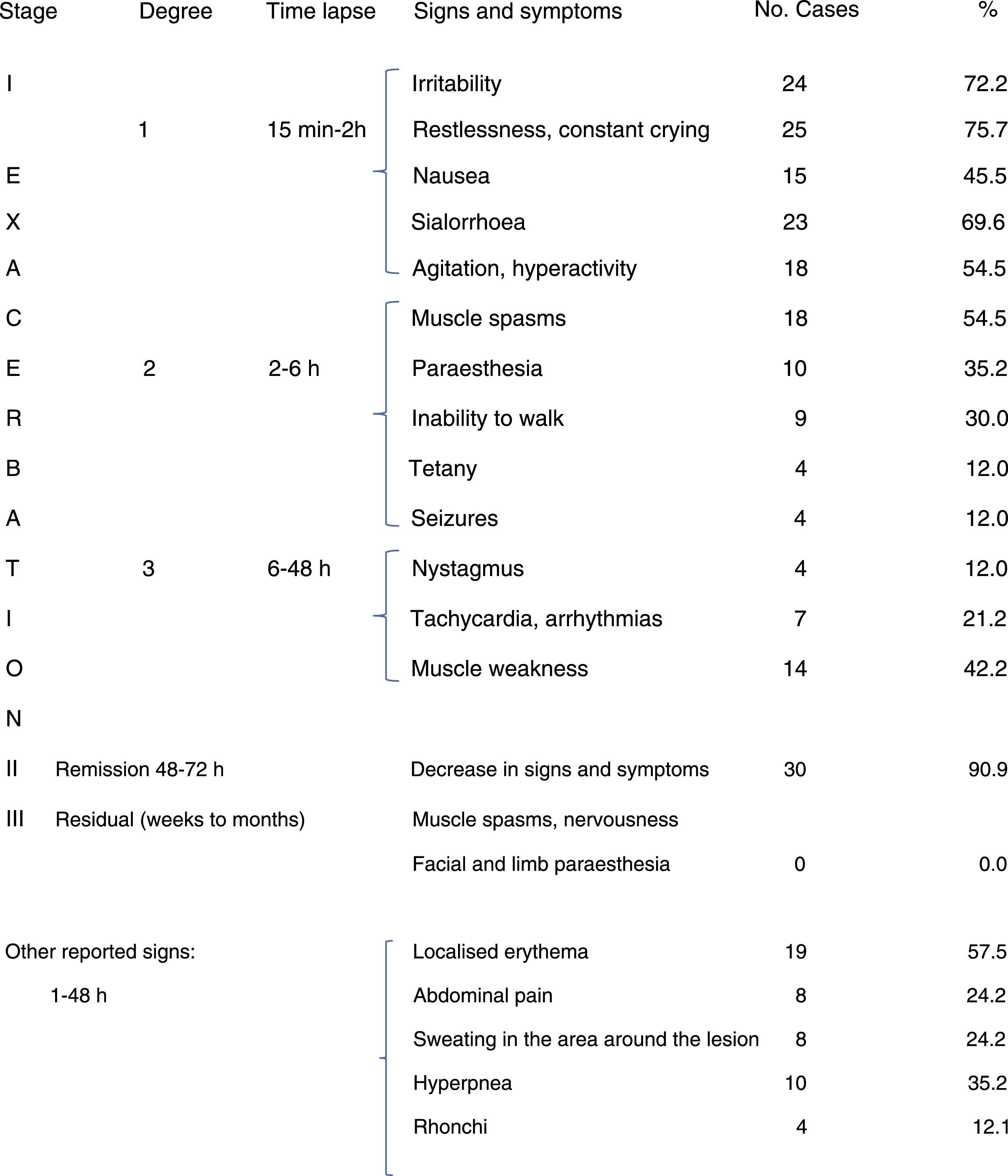
Group 1 included 33 patients. In this group, the main neurological manifestations across all stages and degrees of envenomation were: irritability, constant crying, nausea, sialorrhoea, agitation, hyperactivity, muscle spasms, paraesthesia, inability to walk, tetany, seizures, nystagmus, arrhythmias, and tachycardia. Other signs and symptoms were: localised erythema, sweating in the area around the lesion, polypnea, weakness, rales, and rhonchi. After 2 to 3 days, when the lesion began to disappear, signs and symptoms improved in 90% of patients; residual manifestations did not last long in infants and preschool children (Table 2).
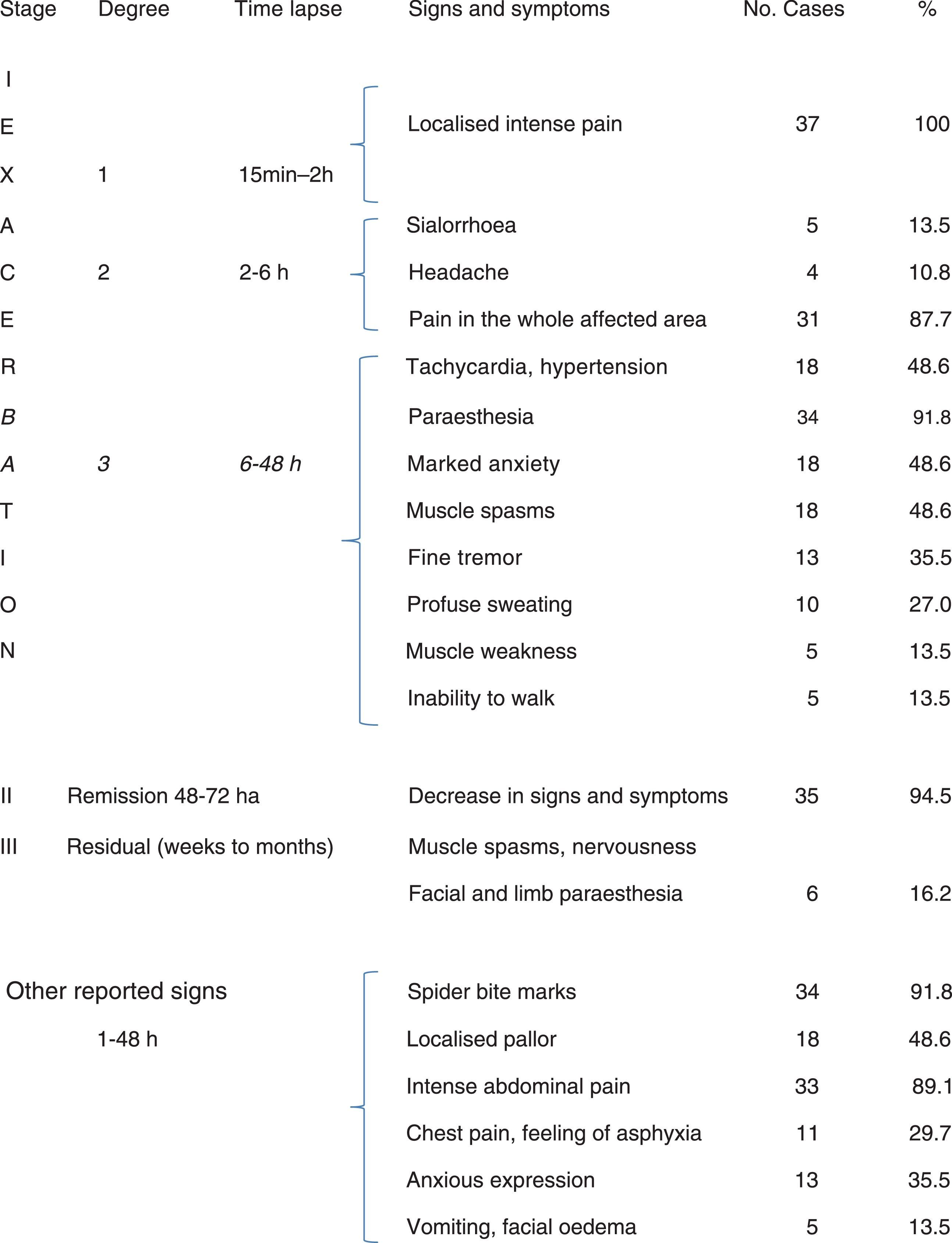
Group 2 comprised 37 patients. The initial symptom was intense pain. Over the following hours, the most common symptoms were: sialorrhoea, headache, pain in the affected area, paraesthesia, anxiety, tachycardia, hypertension, muscle spasms, fine tremor, profuse sweating, muscle weakness, and gait alterations. In 35 patients (94.5%), manifestations decreased in the remission stage (48 to 72hours), but in 6 patients residual manifestations lasted longer. Other signs and symptoms, including late autonomic manifestations appearing one or more hours after the bite were: bite marks, localised pallor and erythema, generalised limb pain, intense abdominal pain, chest pain with a feeling of asphyxia, marked anxiety, painful muscle spasms, and fine tremor (Table 3).
Regarding laboratory test results, 18 patients (25.7%) displayed leukocytosis, 7 (10%) had haemoglobin levels below 10g/dL, 2 patients showed hypocalcaemia (< 8g/dL), and one patient had glycaemia >120mg/dL. Patients were treated with physiologic saline solution (65 patients, 92.8%), hydrocortisone (32, 45.7%), antihistamines (30, 42.8%), calcium gluconate (21, 30%), methocarbamol (11, 15.7%), neostigmine (6, 8%), diazepam (5), phenobarbital (2), and nalbuphine (1). Approval of spider Fab antivenom treatment occurred in 1999; from that year to 2014, antivenoms were used in 47 patients (67.1%). Hospitalisation time was less than 12hours in 14 patients (20%), 6 of whom were infants; 13 to 24hours in 42 patients (60%), 24 of whom were preschool children; 48 to 72hours in 10 patients (14.2%); and 4 to 7 days in 5% of the patients. All patients improved; no deaths were reported.
DiscussionAccidents caused by black widow spider bites occur on a daily basis during the summer, both inside and outside the home. Most of the patients in our series came from urban areas. Accidents usually occurred during the spring and summer months and at home. The site of the lesion was correlated with the area where the arthropod was in contact with the patient.1,13–16
As previously mentioned, the venom of the black widow spider contains several protein toxins. These have great affinity for nerve terminals and interact with synaptic vesicle proteins, forming a complex that leads to a massive release of neurotransmitters. Latrophilins and neurexins are involved in cell recognition in the nervous system for latrotoxin and latrodectin proteins, resulting in depolarisation at the neuromuscular junction, autonomic hyperreactivity, and exhaustion of acetylcholine, which may explain the clinical symptoms associated with black widow spider envenomation.1–11
Diagnosis is based on the following findings: pain appears in the affected area from 15minutes to an hour after the accident, the affected area shows 2 marks (caused by the chelicerae of the spider) and moderate pallor, erythema appears in the affected area, and the skin displays lower temperature, pruritis, diaphoresis, and piloerection. If no specific treatment is administered, other neurological signs and symptoms appear as envenomation progresses11,13–18; these may vary depending on the patient's age, as shown in Tables 2 and 3.
A number of clinical signs and symptoms involving the sympathetic and parasympathetic nervous system can be identified. They are associated with increased neurotransmitter interaction and may affect several body systems and mechanisms, including the neuromuscular junction, eyes, salivary glands, heart, lungs, digestive system, bladder, reproductive organs, and skin.4,5,19 In our sample, infants and preschool children showed irritability, constant crying, nausea, sialorrhoea, agitation, hyperactivity, muscle spasms, paraesthesia, inability to walk, tetany, seizures, nystagmus, tachycardia, and arrhythmias.
Clinical symptoms in young infants may be misdiagnosed for scorpion envenomation since irritability and constant crying are common symptoms of scorpion stings. However, children who can talk may report other symptoms (intense muscle pain, chest and abdominal pain, muscle spasms, etc.), which may be useful for differentiating between black widow and scorpion envenomation. If envenomation progresses and no treatment is administered, nystagmus and muscle spasms worsen, distal fine tremor increases, and other symptoms appear: tachycardia, arrhythmias, hypertension, seizures, pulmonary oedema, hypotension, vascular collapse, and shock.18–22
Preschool and school-age children who can express themselves clearly make it easier to record clinical manifestations: headache of varying intensity, nausea and even vomiting, generalised paraesthesia which the patient may describe as ‘tingling’, muscle pain, inability to walk, muscle weakness manifesting as fatigue, and even painful priapism in preschool children and at other ages.
In adolescents, autonomic symptoms are more intense as time passes. These patients may experience sialorrhoea, headache, pain in the affected area, paraesthesia, anxiety, tachycardia, hypertension, muscle spasms, fine tremor, profuse sweating, muscle weakness, gait alterations, intense muscle pain which increases during seizures, and intense abdominal pain that may be mistaken for acute abdomen. This latter symptom has been reported in 33 out of 37 patients,16–22 as can be seen in Table 3. In adolescents and adults, chest pain may be more intense and be associated with a feeling of asphyxia. People with high anxiety levels may describe this feeling as ‘imminent death’, which may be mistaken for myocardial infarction. Patients usually present an anxious expression on their faces and excessive facial sweating.16,20 These 2 symptoms were reported in 11 (chest pain) and 13 (anxious expression) patients in group 2. In school-age children and adolescents, these symptoms may be more intense than in infants and preschool children due to psychological factors, since older patients tend to show greater awareness of the incident. It is therefore necessary to correctly establish the stage and degree of envenomation and start appropriate treatment.14–17
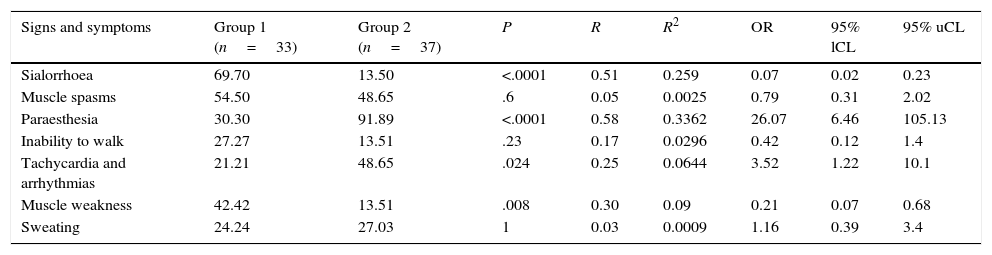
We compared 7 autonomic neurological symptoms in the 2 groups and found that sialorrhoea was statistically significant in group 1 (P<.0001) whereas paraesthesia was significant in group 2 (P<.0001) (Table 4).
Comparison of 7 neurological symptoms between group 1 (infants and preschool children) and group 2 (school-aged children and adolescents).
| Signs and symptoms | Group 1 (n=33) | Group 2 (n=37) | P | R | R2 | OR | 95% lCL | 95% uCL |
|---|---|---|---|---|---|---|---|---|
| Sialorrhoea | 69.70 | 13.50 | <.0001 | 0.51 | 0.259 | 0.07 | 0.02 | 0.23 |
| Muscle spasms | 54.50 | 48.65 | .6 | 0.05 | 0.0025 | 0.79 | 0.31 | 2.02 |
| Paraesthesia | 30.30 | 91.89 | <.0001 | 0.58 | 0.3362 | 26.07 | 6.46 | 105.13 |
| Inability to walk | 27.27 | 13.51 | .23 | 0.17 | 0.0296 | 0.42 | 0.12 | 1.4 |
| Tachycardia and arrhythmias | 21.21 | 48.65 | .024 | 0.25 | 0.0644 | 3.52 | 1.22 | 10.1 |
| Muscle weakness | 42.42 | 13.51 | .008 | 0.30 | 0.09 | 0.21 | 0.07 | 0.68 |
| Sweating | 24.24 | 27.03 | 1 | 0.03 | 0.0009 | 1.16 | 0.39 | 3.4 |
95% lCL: 95% lower confidence limit; 95% uCL: 95% upper confidence limit; P: statistical significance; R and R2: correlation; OR: odds ratio.
The differential diagnosis must include appendicitis, abdominal aortic aneurysm, cholecystitis, intestinal ischaemia, hypocalcaemia, tetanus, and organophosphate poisoning.2,14–20
There are no specific laboratory tests for diagnosing black widow spider envenomation. Some patients may display leukocytosis and elevated creatine kinase levels, while some difficult cases may show anaemia, haemolysis, alterations in liver enzyme levels, or azotemia.16,20–22
A spider Fab antivenom treatment has been available for over 15 years now; this antivenom is albumin-free and modified by enzymatic digestion. This treatment is estimated to neutralise the venom produced by 180 venom glands (LD50 6000). It is prescribed according to the severity of envenomation of each case. It can be administered intravenously, diluted in 20mL of physiological saline, for 2minutes; this process is repeated every hour. Patients are assessed every 4hours until symptoms disappear. The antivenom can also be administered intramuscularly, diluted in 5mL of physiological saline.13,16,22 Symptoms usually disappear after the first dose, limiting the use of such other drugs as sedatives and relaxants. In cases of late diagnosis, patients should be administered as many doses as necessary until symptoms of neurotoxicity disappear. Likewise, whenever necessary, they should receive electrolyte solutions and drugs to treat irritability, pain, and muscle spasms, preferably sedatives, benzodiazepines, and opiates. Due to their high effectiveness, antivenoms minimise the risk of hypertension, seizures, pulmonary oedema, and shocks.13,16,22–27
In our series, patients’ outcomes were good. Although no specific antivenom treatment was available during the first 20 years of the study period, no deaths were reported. In addition, between 1999 (when Fab antivenom treatment was introduced) and 2014, the mean hospitalisation time dropped to 12hours with a maximum hospitalisation time of 24hours, limiting the use of other drugs.
Our study has a number of limitations due to its retrospective design. However, we hope it provides some useful information for other researchers interested in this topic.
FundingThe authors have received no financial support.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Sotelo-Cruz N, Gómez-Rivera N. Manifestaciones de neurotoxicidad en el envenenamiento por mordedura de araña «viuda negra» en edades pediátricas. Neurología. 2016;31:215–222.