Hypertrophic olivary degeneration (HOD) is an atypical form of trans-synaptic degeneration representing the final result of an alteration in the neuronal connection of the dentatorubral–olivary pathway. This anatomical pathway was described by Guillain and Mollaret1 in 1931. Clinical manifestations of HOD include involuntary movements by muscles dependent on cranial nerve nuclei in the brainstem, such as palatal myoclonus and ocular myoclonus.2
Its anatomical pathology is characterised by vacuolar degeneration of the cytoplasm associated with an increase in the number of astrocytes in the olivary body, which also entails an increase in that structure's volume.
The purpose of this article is to present the MRI findings and pathophysiological profile of the lesions in 2 cases of HOD; 1 case was ipsilateral and the other was contralateral to high brainstem lesions.
Case 1. Patient aged 43 with no relevant medical history who abruptly presented with dizziness, instability, and right hemifacial paraesthesia. Physical examination revealed tactile hypoaesthesia and hypalgesia on the right side of the face and the upper right limb, dysarthria, and left clumsy hand. Brain CT showed no abnormalities. Patient was diagnosed with vertebrobasilar stroke and scheduled for an MRI and neurological check-up.
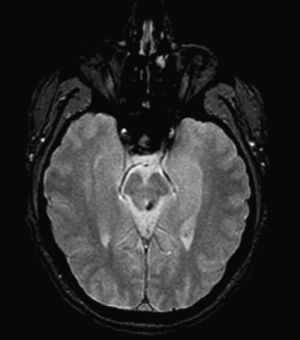
The MRI taken 6 months later revealed a linear millimetric hypointense focus in the left dorsolateral region of the midbrain (upper cerebellar peduncle), corresponding to traces of haemosiderin secondary to a haemorrhagic infarct or cavernous angioma (Fig. 1).
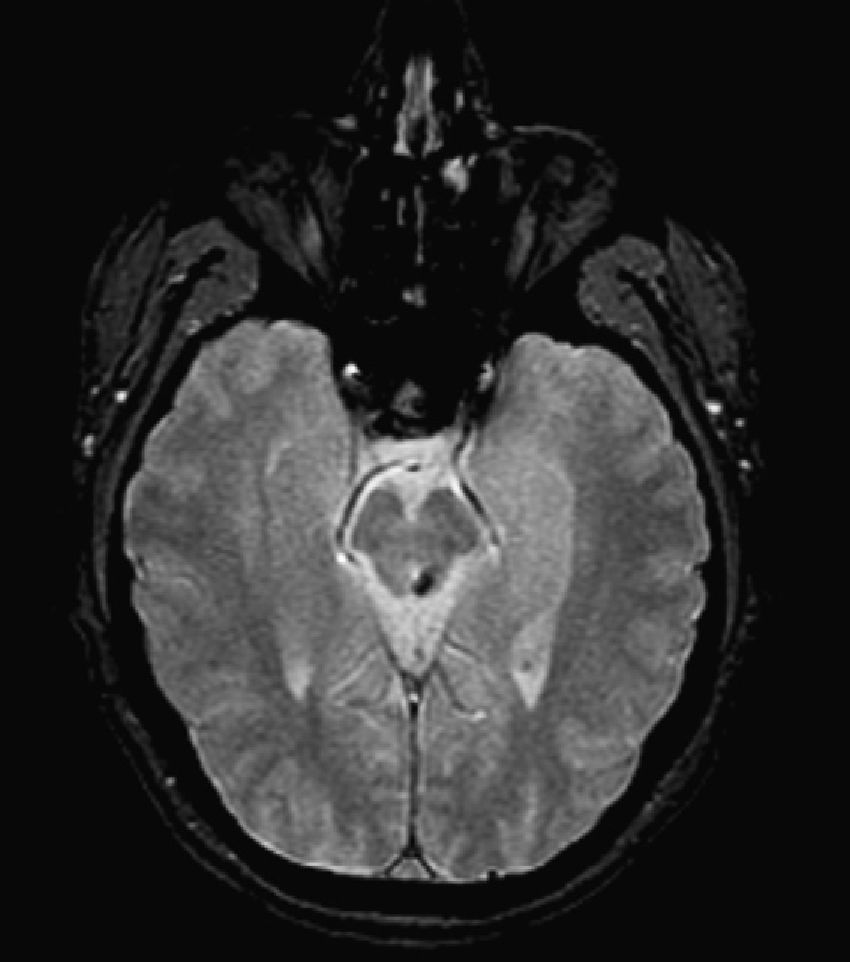
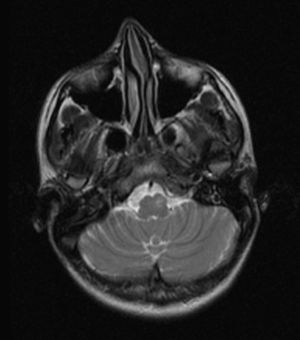
T2-weighted images showed a hyperintense lesion with an increase in volume in the right anterolateral region of the medulla oblongata corresponding to the location of the inferior olivary nucleus (Fig. 2).
Case 2. Patient aged 22 years with no relevant history who visited the emergency department due to diplopia, instability, right-sided paraesthesia, and intense headache. The patient reported having experienced several episodes of spontaneously remitting right-sided paraesthesia a year before. Physical examination revealed conjugate gaze palsy and unstable gait with deviation to the left.
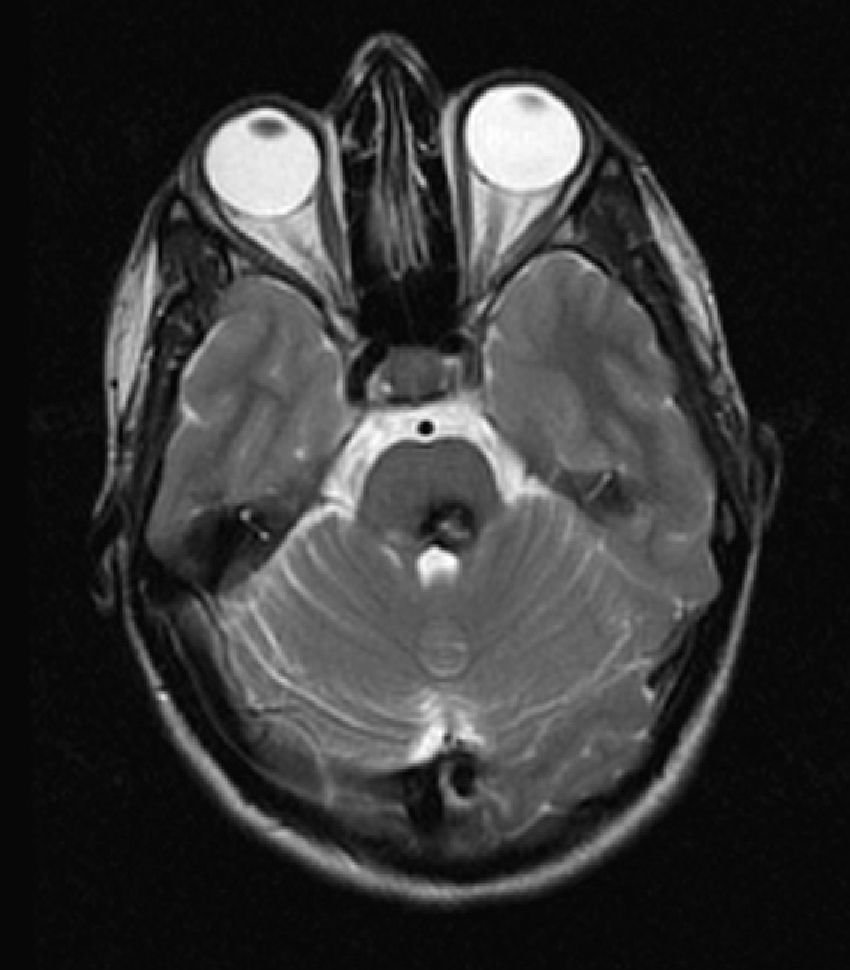
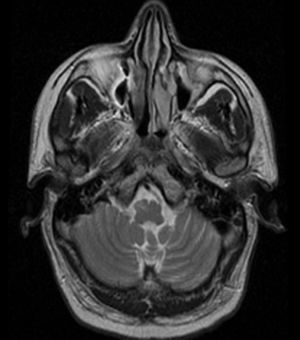
The MRI showed a lobulated lesion on the left dorsomedial pontine region with a hypointense peripheral halo in the T2-weighted sequence and a heterogeneous, predominantly hyperintense, central reticulated core (Fig. 3) suggestive of a cavernoma.
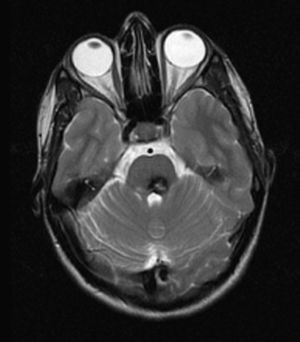
Inferior to this structure, we identified a hyperintense focus and asymmetry in the anterior rim of the medulla due to an increase in volume in the typical location of the left inferior olivary nucleus (Fig. 4).
In both cases, the array of findings is strongly suggestive of HOD secondary to the destruction of part of the dentatorubral–olivary pathway (Guillain–Mollaret or myoclonic triangle).
HOD is an atypical form of trans-synaptic degeneration caused by alterations in the neuronal connection in the dentatorubral–olivary pathway. In MR imaging studies, it manifests as increased size and hyperintensity of the inferior olivary nucleus in T2-weighted sequences; these changes are associated with midbrain–pontine lesion. Its anatomical pathology consists of an increase in volume due to cytoplasmic vacuolar degeneration and an increase in the number of astrocytes.3
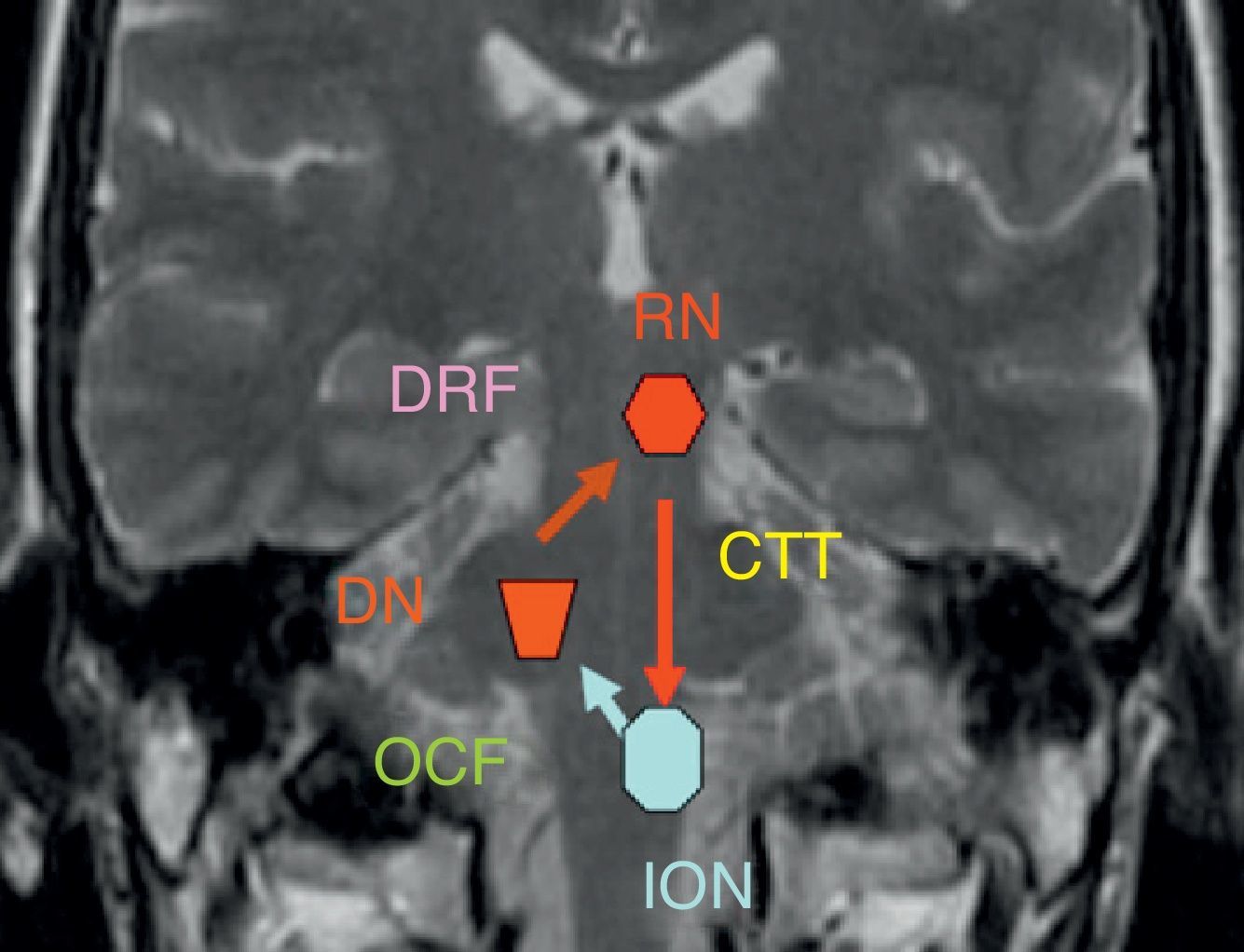
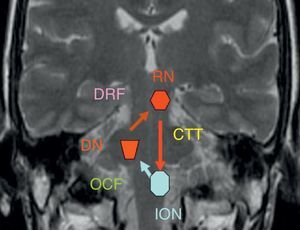
The dentatorubral–olivary pathway or myoclonic triangle connects the dentate nucleus to the contralateral red nucleus and inferior olivary nucleus (Fig. 5).
The dentate nucleus forms a synapse with the contralateral red nucleus through the superior cerebellar peduncle (dentatorubral pathway). Its fibres decussate in the inferior medial midbrain. The red nucleus and the ipsilateral inferior olivary nucleus are connected by the central tegmental tract, which is located in the dorsal paramedian pontine region immediately lateral to the medial longitudinal fasciculus and dorsal to the external portion of the medial lemniscus. The pathway is completed by the olivary–cerebellar network of connections leading from the inferior olivary nucleus to the contralateral dentate nucleus. This explains why there are 3 different patterns for olivary degeneration according to the location of the primary lesion: if the primary lesion is located in the dentate nucleus or the superior cerebellar peduncle (prior to the decussation of the dentatorubral tract), then olivary degeneration is contralateral, as in case 1. If the primary lesion is limited to the central tegmental tract (case 2), then olivary degeneration is ipsilateral; degeneration is bilateral in cases with both brainstem and cerebellar lesions.4–6
Typical clinical findings in HOD include palatal myoclonus, which may or may not be associated with other forms of myoclonus, such as ocular myoclonus, which depend on the cranial nerve nuclei in the brainstem. This occurs because damage to the central tegmental tract elicits lack of inhibitory control in those nuclei.7 However, our patients did not display abnormal movements and their symptoms related to lower cranial nerves stabilised and improved.
Hyperintensities in the olivary body generally appear 3 to 4 weeks after occurrence of the primary lesion. They may persist for years or even indefinitely. The increase in volume takes place during a period of 4 to 6 months and usually resolves within 3 years. The olivary body therefore appears normal-sized during the acute–subacute phase, hypertrophic by 4 to 6 months, and atrophic by 3 years.3,5,8,9
The most common causes of brainstem damage that may involve the dentatorubral–olivary pathway are haemorrhage secondary to hypertensive disease, head injury, vascular malformations, and infarction.4,6,10
The differential diagnosis for hyperintensities on the anterolateral medulla includes neoplasia (astrocytoma, metastasis, lymphoma); infections and inflammatory processes (tuberculosis, HIV, sarcoidosis, rhombencephalitis); multiple sclerosis; and ischaemia. In any case, the preliminary diagnosis is established by locating a lesion strictly limited to 1 or both inferior olivary nuclei in association with another distant lesion located in the contralateral dentate nucleus, along the contralateral superior cerebellar peduncle, the ipsilateral red nucleus, or the ipsilateral tegmental tract. A decrease in the size of the structure in subsequent examinations, no enhancement after administration of intravenous contrast, and atrophy of the dentate nucleus contralateral to the affected inferior olivary nucleus are other radiological signs that support this diagnosis.
A thorough foundation in the anatomy and physiology of the dentatorubral–olivary pathway (Guillain–Mollaret triangle) will help us to understand the different patterns of HOD. The condition may be diagnosed from an MR image based on the presence of a distant lesion at a specific location. This enables us to rule out other pathologies that cause hyperintensities in the anterolateral medulla.
Please cite this article as: Sánchez Hernández J, et al. Degeneración hipertrófica de la oliva por lesión del triángulo de Guillain-Mollaret. Presentación de 2 casos. Neurología. 2013;28:59–61.