Embolism is a well-established cause of stroke. From a histopathological standpoint, the composition of cerebral emboli varies considerably.1 In most cases, these emboli consist of accumulations of platelets, fibrin, and cholesterol, with traces of calcium. Descriptions of calcific emboli are infrequent. These emboli typically affect the anterior circulation, although there are cases in the literature of such emboli affecting the posterior cerebral artery.2 The treatment approach in these patients is controversial. There have been several isolated cases of treatment using intravenous fibrinolysis, but outcomes have varied considerably.2–4 We present the case of a patient who did not respond to intravenous thrombolysis and intra-arterial rescue treatment for an ischaemic cerebral infarct secondary to calcific embolus. We would like to stress the role that different types of fibrinolytic treatment may play in such situations.
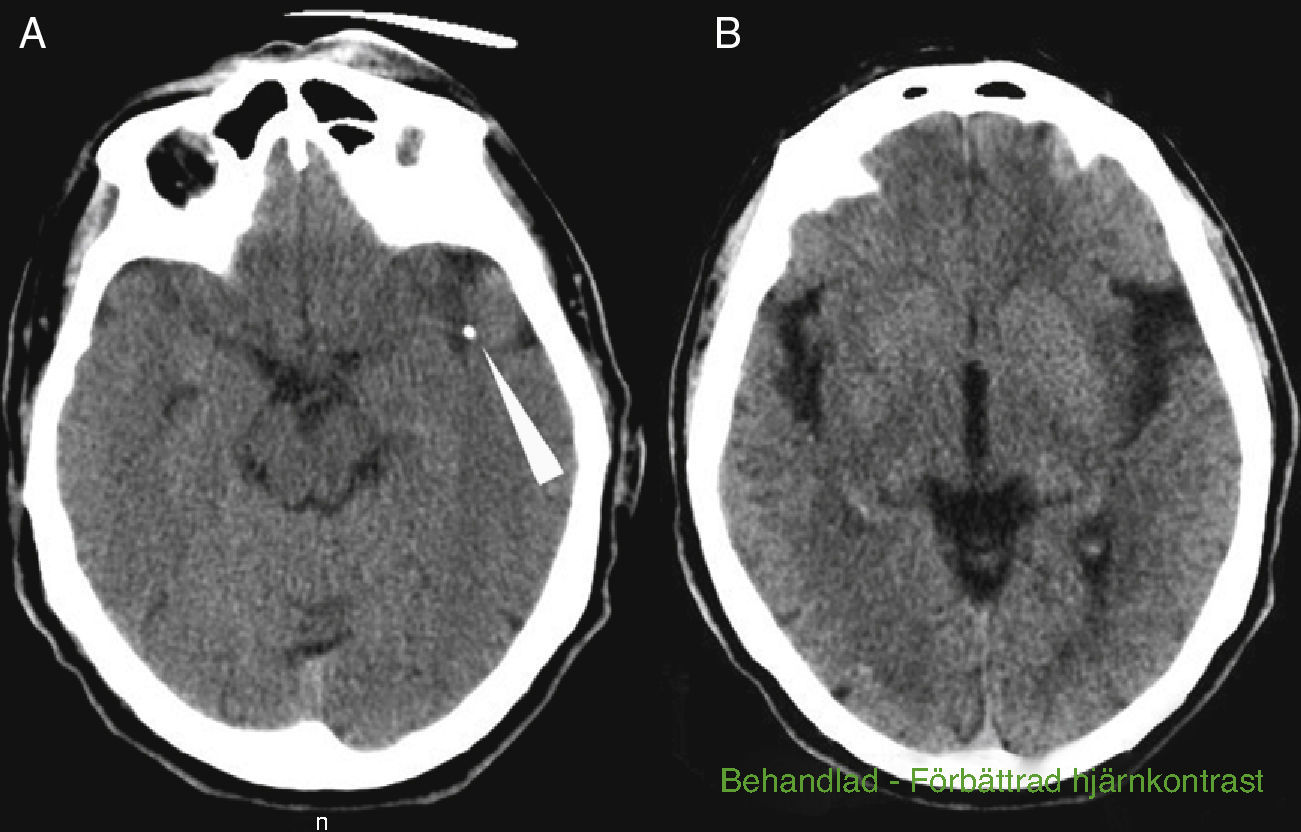
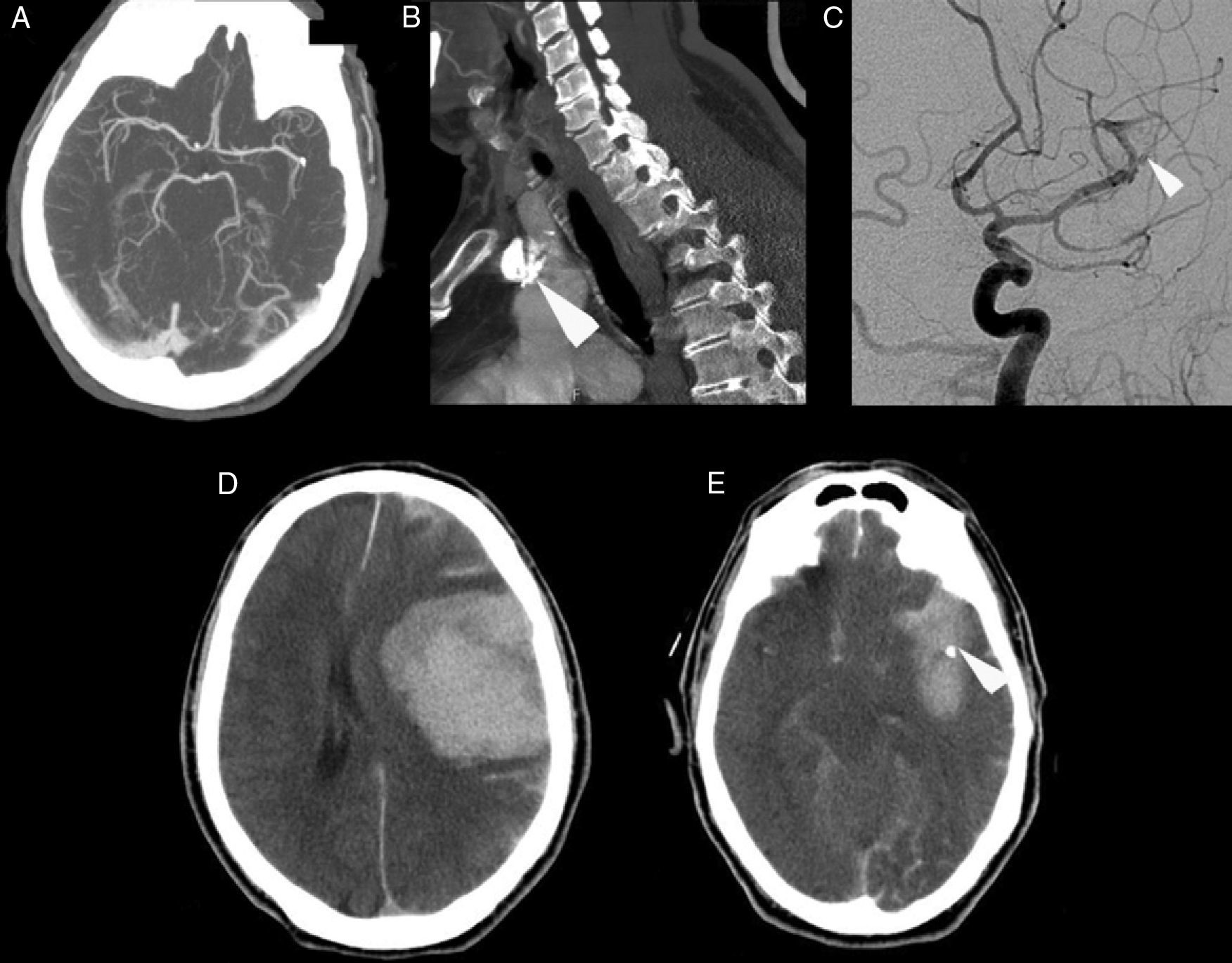
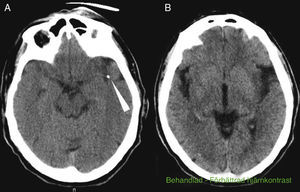
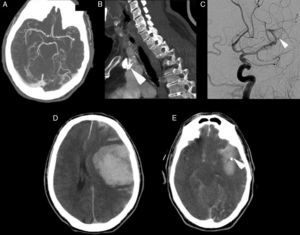
The patient was a 69-year-old male with a history of arterial hypertension and dyslipidaemia, both treated. He was independent for all activities of daily living (mRankin 0, Barthel index 100). The patient came to the emergency department due to left-hemisphere focal neurological deficits with unknown time of onset. He was last seen to be asymptomatic at 9.30. Family members found him in his home at 13.45 with decreased mobility in the right half of the body and speech impairment. Neurologists were called in for an ED consult at 15.40. Neurological examination revealed a total absence of expressive language with abolished comprehension and right-sided paralysis; NIHSS score was 21. A Doppler study indicated lack of flow in the left middle cerebral artery at a depth of 55mm, which was compatible with occlusion of the distal M1 segment. Head CT at admission showed a hyperdense image in the distal M1 segment of the left MCA and a slight ischaemic hypodensity in the left temporal and insular region; Alberta Stroke Programme Early CT score (ASPECTS) was 8 (Fig. 1A and B). The CT perfusion study revealed 60% discordance. CT angiography confirmed the occlusion of the proximal M2 segment and the presence of a significant calcified plaque in the aortic arch (Fig. 2A and B). Doctors began intravenous tPA treatment dosed at 0.9mg/kg at 16.05; there were no clinical or neurosonology changes in the following 60minutes. At 40minutes after beginning perfusion, doctors alerted interventional neuroradiologists and anaesthetists. Rescue intravascular treatment was started at 17.15 (some 70minutes after onset of intravenous fibrinolytic treatment. Initially, and in 2 separate attempts, doctors tried to extract the thrombus using a Bonnet device. When they could not mobilise the obstructive material, thought to be plaque calcified in situ, doctors decided to perform balloon angioplasty. The procedure was finished at 19.30 but the distal arterial bed had not been fully recanalised. Contrast extravasation was detected after one angioplasty balloon inflation (Fig. 2C). After the patient was moved to the recovery room, we were alerted at 20.00 that he displayed low level of consciousness, left-sided anisocoria, and decerebrate response to pain. An emergency CT revealed a large left temporoparietal intraparenchymal haematoma measuring some 100cm3 with ventricular and subarachnoid spillover and midline shift (Fig. 2D and E). The patient died at 20.30.
(A) Cranial computed tomography angiography showing occlusion in the proximal M2 segment. (B) Computed tomography angiography showing a calcified plaque in the aortic arch (arrow). (C) Conventional cranial angiography revealing partial recanalisation of the artery. The Bonnet device and angioplasty failed to open the distal artery bed and contrast extravasation occurred (arrow). (D) CT revealing an extensive left temporoparietal intraparenchymal haematoma measuring some 100cm3 with ventricular and subarachnoid spillover and midline shift. (E) Cranial computed tomography showing persistence of the possible calcium spot in the distal M1/proximal M2 segment (arrow).
The composition of the atheromatous plaque affects its embolic potential. Numerous studies have shown that there is less risk associated with calcified plaques than with heterogeneous plaques whose calcium content is lower.5 Calcific emboli tend to be iatrogenic in origin, especially in cases of carotid manipulation, cardiac catheterisation, or heart surgery. The most frequent cause of spontaneous calcific emboli is calcification of the mitral annulus and aortic valve,6,7 although emboli may also stem from calcific plaques located in the ipsilateral internal carotid artery,2,8,9 the aortic arch,3 or the brachiocephalic trunk.10 We feel that calcific embolism from the aortic arch is the most plausible mechanism in our case, based on results from complementary tests and the fact that the rest of the vascular tree was normal. In the head CT, a calcific embolism appears as one or more punctiform calcium-density images in a vessel's interior. There is no conclusive evidence about the best acute-phase treatment option in these situations, given that few cases have been published; in addition, outcomes, whether favourable2,3 or unfavourable,4 have varied considerably. This being the case, we must stress that the presence of highly calcified plaques may limit possibilities of pharmacological recanalisation or rescue by mechanical devices and increase the risk of vascular damage or mechanical rupture of the vascular wall. The latter mechanism most likely explains the cerebral haemorrhage in our patient. Nevertheless, despite increased complications and the difficulties that may arise in the process of extracting the thrombus, we should not conclude that these emboli are an absolute contraindication for all types of fibrinolytic treatment.11 On the other hand, we must emphasise that the composition of the embolism plays an important role. Increasingly widespread use of mechanical embolectomy devices allows us to obtain samples of fresh thrombi and complete anatomical pathology studies.12 Gaining a better understanding of the nature of the embolus causing the ischaemic event allows us to stratify risks and evaluate the efficacy and safety of different recanalisation treatments. This information helps us make final decisions.
Please cite this article as: Mayor Gómez S, Muñoz Arroniz R, Olier Arenas J, Gállego Cullere J. Infarto cerebral por embolismo cálcico y tratamiento trombolítico ineficaz. Neurología. 2014;29:123–125.