To determine the relationship between TNF alpha and the etiology, localization, extension, intima media thickness, carotid atherosclerotic plaque, and outcome after an acute stroke.
MethodsWe randomly selected 75 patients with acute strokes from a total of 253 patients that were admitted prospectively from May, 2008 to December, 2010. We analyzed TNF alpha levels and compared it with demographic data, clinical outcome upon hospital discharge, and at 3 months post discharge with neuroimaging studies. We used the Chi-square test, the U-Mann–Whiney test and the Cox logistic regression adjusted for age, gender and stroke extension.
ResultsWe included 47 men and 28 women. The most common etiologies were atherosclerotic (39%) and small vessel disease (27%). TNF alpha levels did not differ between atherosclerotic and cardioembolic stroke etiologies, except for the lacunar infarction, which had the lowest levels (p=0. 048), and did not correlate with a worse functional outcome upon hospital discharge (p=0. 852) or at 3 months following discharge (p=0.194). Additionally, we found a positive relation between intima media thickness >1mm and TNF alpha (p=0. 004). TNF alpha was not associated with the extension of the stroke by an ASPECTS score with CT or MRI (p=0.323) or with the arterial territory involved (p=0. 289).
ConclusionsTNF alpha was not globally associated with functional outcome after acute stroke, just in the lacunar infarction, which has the lowest levels. We also found a positive relation between TNF alpha and intima-media thickness.
Strokes are the most common cause of functional disability in adults1 and the sixth leading cause of death in our country.2 It is considered to be the second most common cause of death in the world, and its prevalence is increasing in developing countries due to increased life expectancy and changes in eating habits and lifestyle.1
A stroke may have different etiologies. Among them the most common are known to be large artery atherosclerosis, small vessel disease or lacunar type, and caused due to cardiac embolism.3 Other less common causes are pro-thrombotic states of systemic diseases, primary or secondary cerebral vasculitis, and arterial dissection secondary to medications or harmful substances.3 When a patient has two probable causes or there is a failure to clarify the etiology, it is defined as undetermined until the moment that its origin is detected. Up to 15–25% of strokes remain undetermined.1
Atherosclerosis is a more common cause, especially in adults older than 50 years.3 Unfortunately, there are no reliable statistics on the prevalence of atherosclerotic carotid artery stenosis in strokes in our country. However, in other developed countries, it has been established that 35–50% of cases are caused by atherosclerosis of large vessels, such as the carotid artery, the thoracic aorta and the proximal intracranial vessels.3
The second most common cause of a stroke is a disease of the penetrating vessels by degeneration of the vascular wall or lipohyalinosis, with the consequent microangiopathy of the small cerebral vessels and infarcts of a more limited volume, usually less than 1.5 or 2.0cm.1 This disease is typical of patients with hypertension and/or chronic diabetes. The volume of injuries from a lacunar infarct is small; therefore, a lesser number of neurons are affected. However, its clinical consequences depend on the location of the lesion.
Thirdly, there is the cardio-embolic etiology. This may be due to an arrhythmic phenomenon, or an injury to the heart wall that conditions a focal myocardial dyskinesia and thus the formation of an intracavitary thrombus, as occurs after a heart attack.3 Although more common in younger subjects, its diverse etiology makes it likely in the elderly as well. Strokes, as a consequence of an embolus from the heart, can hide a distal artery in the brain, and generally hide a vessel of great size, leading to a wide-reaching stroke.3
The etiology may vary according to the study population, which is what makes it important to emphasize the etiological definition of a stroke in all prospective cohorts of patients.
After a stroke, inflammatory phenomena as a result of the damage and its extent occur,4 as well as others, which seek to limit the consequences of the lack of irrigation in the peripheral area of a stroke, known as the ischemic penumbra area.5,6
Among the molecules that have been defined to be associated with strokes, their extension and prognosis, are cytokines, such as IL-1, IL-6, and TNF alpha.7 Some molecules increase in blood immediately and remain in the blood at high levels for several days. Their quantification may be a reflection of the degree of damage or the extent of the stroke, and may even help to etiologically differentiate the type of stroke.5,6,8–10
Tumor Necrosis Factor (TNF) alpha and Cerebral Vascular Disease (CVD)TNF alpha is produced by brain cells, both neurons and microglia, after suffering ischemia.11 It is involved in all stages of brain injury by stroke. Its pro-inflammatory effect appears to initially maintain cerebral flow in the periphery of the stroke and is proportional to stroke size.5–9
Many studies that have associated etiological subtype and functional prognosis following a stroke, but few who have studied TNF alpha as a pro-inflammatory cytokine cerebro-spinal fluid and serum that can be associated with the size, clinical severity and etiology of a stroke.4,6,8–10,12–14
In a study of 123 stroke patients and 123 healthy controls, patients with cardioembolic CVD showed higher levels of TNF alpha plasma in plasma when compared with the other subtypes.13 In a study of 36 patients with acute ischemic stroke at admission,it was found that concentrations of TNF alpha did not vary significantly from controls, but the amount of soluble TNF receptor type 1 in the first week correlated strongly with stroke size by CT at 5–7 days, as well as the mRS (modified Rankin Scale) at 3 and 12 months.11
There have only been a few studies on the behavior of TNF alpha in strokes, especially TNF alpha in acute strokes that emphasize the stroke etiological subtypes and clinical features.
Materials and methodsPatientsThis is an analytical cross-sectional study nested in a prospective cohort of patients diagnosed with a stroke who were admitted to the Neurology Department at a tertiary level teaching hospital in Northeastern Mexico (Intrahospital Registry of NeuroVascular Disease – iRENE-) in the period of May, 2008 to December, 2010. The protocol was approved by the Ethics Committee; patients and/or family members signed a letter of consent.
During the aforementioned period, 253 patients with a diagnosis of cerebrovascular disease were hospitalized. 75 patients were chosen at random from a selection of 133 patients with a confirmed stroke diagnosis, with complete imaging studies, laboratory and clinical follow-up at hospital discharge and at 3 months.
Demographic information, etiology, stroke location and laboratory and imaging studies were obtained from the form used by the Neurovascular Care Room since 2008. The extent of the stroke was determined by the ASPECTS (Alberta Stroke Program Early CT Scan) scale, by CT or by MRI. All patients’ functional status was measured with modified Rankin and NIHSS (National Institutes of Health Stroke Scales) scales upon admission, hospital discharge and during a follow-up at 3 months, either by outpatient or phone call using the validated Rankin telephone tool. Among the comorbidity background, we included a sedentary lifestyle (lack of routine exercise for at least 30min a week in the last 6 months) and cardiovascular disease (history of angina pectoris, myocardial infarction, heart failure or valve disease).
Serum samples were taken by venipuncture between day 0 and day 16 of hospitalization. 56% of the samples were taken in the first 48h. Samples were stored at −80°C in our Department of Biochemistry of the School of Medicine.
Functionality scales in CVDThe use of standardized functional scales is essential to compare populations of different origins. The mRS is a comprehensive clinical evaluation of disability. It consists of degrees from 0 to 6, with 0 being an asymptomatic patient, grade 5 being a patient that requires full support 24h a day, unable to perform the most basic activities, and 6 describes a deceased patient. A simplified mRS questionnaire has demonstrated excellent reproducibility and reliability when applied by telephone.16,17
The NIHSS are widely used and validated to assess severity. In the acute phase of a heart attack, it is a powerful predictor of the final clinical outcome.18
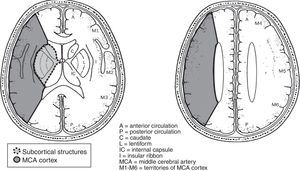
The ASPECTS scale determines changes in ischemia on CT. The value is obtained from 2 axial cuts. It divides the territory of the middle cerebral artery into 10 regions, and one point for each affected area is subtracted. It is related inversely with the severity of the stroke. A lower value of 7 is associated with increased reliance and the risk of death at 3 months. In a study of the ability to detect early ischemic changes, ASPECTS was similar to CT and MRI in diffusion sequences.17
Analysis of TNF alphaAfter obtaining the blood samples, they were centrifuged and the plasma was separated. They were taken to the Department of Biochemistry in our School of Medicine, where they were stored frozen. The samples were thawed and measurement of TNF alpha was performed with the conventional technique using commercial Milliplex Map kits. Measurements were performed at the laboratory at a tertiary level teaching hospital in Northeastern Mexico.
Statistical analysisA descriptive analysis of the population's demographic and clinical variables was made. The dependent variable for functional status at discharge and at 3 months was obtained through a dichotomized modified Rankin scale with a good prognosis (0–3) and a poor prognosis (4–6). Comparisons were made with Pearson's Chi-square tests (or Fisher's exact) for contingency tables, or the Mann–Whitney U test for comparison against quantitative or qualitative non-parametric variables, as appropriate.
TNF alpha values were analyzed with the Kolmogorov-Smirnov normality test to learn their distribution, and further analyzed in continuous values for Spearman correlation tests. 75 percentile values were taken to contrast with the patients’ clinical characteristics, stroke location and extent, and the presence of atherosclerosis, as well as their etiology and prognosis.
Finally, the variables that were significant in their association with the stroke prognosis underwent a multivariate logistic regression model along with the TNF alpha values. p<0.05 was used as statistical significance, and we used the SPSSv.15 statistical program.
ResultsSeventy-five patients were randomly selected for this study, from 133 patients with the complete information required upon admittance and at the clinical follow-up 3 months after discharge.
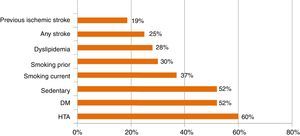
The mean age was 12.9±65 years (29–91 years old); 63% male and 37% female. The prevalence of comorbidities is shown in Graph 1. Of these patients, 96% had functional independence prior to their cerebrovascular event.
Time of arrival after the start of symptoms was <3h in 10.6%, 3–6h in 17.3%, 6–24h in 45.3%, 1–7 days in 25.3% and >7 days in 1.3%.
Regarding etiology, 39% were atherothrombotic, 27% were due to small vessel disease (lacunar infarcts), 15% to cardioembolism and the etiology could not be determined, or more than one possible etiology was found, in 20%.
The thickness of the carotid intima-media in the common carotid artery was determined for all patients via Doppler ultrasound, finding an average value of 0.930±0.303mm (0328–2672). 68% of patients had carotid atherosclerotic plaques (greater than 30% stenosis), and we found an association between the presence of plaque and age (p=0.036) as well as with a history of diabetes mellitus (p=0.028).
Regarding vascular infarct location, the middle cerebral artery region was the most frequent (62%), followed by the vertebrobasilar region (20%), the anterior cerebral artery (6.6%), and the carotid artery (6.6%).
Upon arrival at the emergency room, the patients’ score on the NIHSS were determined. An NIHSS score of 22.6% presented between 0 to 4 points (mild stroke), 54.6% presented an NIHSS score between 5 to 12 points, and 22.6% presented an NIHSS score higher than13 points. Their score was re-evaluated upon entering the Neurology Department, where 24% scored 0–4, 58.6% scored 5–12 and 17.3% scored greater than 13.
Upon hospital discharge, after an average stay of 8.5 days (range 2 to 32 days), an mRS showed 62.6% of patients between 0 and 3, 32% between 4 to 5 points and death (mRS=6) in 5.3%. Upon hospital discharge, their NIHSS score was between 0 and 4 points on 48% (mild sequelae), between 5 and 12 points on 36% (moderate scale) and was higher than 13 in 16% of patients (disabling sequelae). At the 3-month follow-up, only the modified Rankin test was performed, which showed a score of 0–3 (functional independence) in 50.6%, 4–5 in 36% (functional dependence) and death (mRS=6) in 13.3%.
Regarding the extent of their strokes, we found an average value of 8.2 points, with a range of 6–9 points, on the ASPECTS scale (Fig. 1).
There were differences in the results between associations of neurological recovery and the etiological type of stroke depending on the functional scale evaluated, with the small vessel disease or lacunar infarct having lower scores on the NIHSS scale, and therefore less neurological damage (p=0.044). On the other hand, with the modified Rankin scale, the etiology with the poorest prognosis at discharge was atherothrombosis (p=0.041) and the small vessel disease had the best functional prognosis again, i.e.lower mRS values (p=0.003).
At 3 months, the etiology with the poorest prognosis was cardioembolic (p=0.001), and the etiology with the best prognosis was small vessel disease (p<0.0001).
Tumor Necrosis Factor alphaTNF alpha values averaged 6pg/ml with a range of 0.79–17.38pg/dl. The values differed according to the etiology of the stroke. The average values of TNF alpha are shown in Table 1. Lacunar infarcts were associated with lower levels of TNF alpha (p=0.048). Glucose levels on admission were not related to TNF alpha (p=0.545).
Tumor Necrosis Factor (TNF) alpha values by etiological stroke group.
Regarding atherosclerosis, a significant association between a thickening of the intima media (greater than 1mm) and the levels of TNF alpha (p=0.004) was found. However, the presence of an atherosclerotic plaque greater than 30% was not related to TNF alpha (p=0.625).
TNF alpha levels were not found to be associated with the extent of the strokes, even eliminating the lacunar infarctions (p=0.323), which suggests an association with TNF alpha regardless of infarct size. Similarly, there was no relationship between infarct location and TNF alpha levels (p=0.289).
Low levels of TNF alpha were not associated with a better prognosis at hospital discharge, or with the modified Rankin Scale (mRS 0–3: p=0.852), and only marginally with the group of patients with a lower NIHSS score (from 0 to 4; p=0.059).
As for their prognosis at three months, measured by the telephone mRS, no association was found with TNF alpha levels at admission (p=0.194).
DiscussionTNF alpha is produced by brain cells after suffering ischemia19 and some authors have associated it with a worse functional prognosis.6,11,12,20,21
In our study, TNF alpha levels were not related to functional outcome at hospital discharge, and very discretely at 3 months with the NIHSS scale. This is possibly due to the small number of cases in the study, and also may be because the prognosis of the target population for this study was generally good, with a functional recovery of 84%, according to the Rankin scale.
Stroke by small vessel or lacunar disease is the second leading cause of strokes in our hospital (19%), and its etiology is defined until we can make a correlation between cardiovascular risk factors, clinical manifestations and findings in brain imaging studies, such as MRI.
Patients with a stroke due to small vessel disease had lower levels of TNF alpha, and better outcomes at hospital discharge and at 3 months. This could be assumed to be due to having strokes of smaller size. However, upon analyzing the ASPECT scale for stroke size and levels of TNF alpha, no association was found. Small vessel or lacunar disease generates the least cytotoxic edema, besides being the stroke with fewer affected cells. This could be useful, since the determination of TNF alpha levels in pre-hospital care would help predict the lacunar subtype of stroke and hence lower ischemic penumbra, with no need for an endovascular approach, and a better prognosis. In addition, lacunar infarction is not accompanied by a demonstrable arterial occlusion in imaging studies such as CT angiography or a magnetic resonance angiography, which predicts a good response to an intravenous thrombolytic drug.
Other authors have found that TNF levels correlate with stroke volume. In our population, the ASPECTS scale scores were very similar, possibly caused by not having the dispersion of the values necessary to find a significant difference.
Interestingly, we discovered an association between TNF alpha levels and the presence of thickening of the carotid intima-media greater than 1mm, and not with the occurrence of atherosclerotic plaques in the carotid arteries, which suggests that the intima media thickness is a factor independent to plaque formation, similar to what others have suggested.22
Fifty-two percent of the samples were taken, centrifuged and frozen within 48h of admission, and 91% in the first five days, a time when others have seen the largest increase in concentrations of TNF alpha or its receptor occurs.
In our study population, high glucose levels at admission had no influence on the patients’ prognosis; even the highest levels, at 180mg/dl, were associated with a good prognosis at the follow-up 3 months after the acute event.
Hospital mortality was 5.3%, and mortality was 13.3% at 3 months. This is less than the 16% mortality rate in the first month reported in the literature.23 But this may be related to the fact that our chosen group had fewer patients with very extended strokes.
Almost half of our patients came for medical care within 6 to 24h after symptom onset and only 10.6% arrived within 3h, so the chances of thrombolytic treatment were very few. Of the 8 patient candidates, only 3 received thrombolytic treatment.
Cardiac ischemia studies have consistently found a significant difference between TNF alpha levels and cardiovascular prognosis.7 On the other hand, results in strokes have been mixed. There are reports in the literature of a significant association between stroke prognosis and TNF alpha levels,5,9 while others have found no association,7 or just a trend,12 and there are even those who report an opposite effect and a protector of pro-inflammatory cytokines.10
This may be because the stroke, unlike the other vascular pathologies, such as cardiac, has diverse etiologies. That is why it is important to define its causes via cohort studies, including the study of prognostic biomarkers.
Welsh et al. demonstrated an association between TNF alpha levels and the risk of recurrent heart attacks, and emphasized that it only occurs in those with very high concentrations of TNF alpha (> 2.89pg/ml). In our population, the percentage of patients whose levels of TNF alpha were found to be above these values was very high (66%), with a maximum value of 17.38p/ml and an average value of 6pg/ml. Despite this, the overall prognosis for our group at 3 months was good. This could be due to the fact that we had a significant percentage of lacunar infarcts (27%), with a good prognosis and lower TNF alpha concentrations.
Like other studies,13 we found that patients with a cardioembolic etiology have worse outcomes, and that patients with CVD by small vessel disease are those with a better prognosis, being no relationship observed with the first to TNF alpha levels, since those values were indistinguishable from those by atherothrombotic causes or unknown causes.
This relates to the findings of Licata et al.,13 where it was observed that patients with cardioembolisms show a greater inflammatory response associated with an increase in TNF alpha, hence worsening the evolution.
In this group of patients, their strokes showed an average of 8 on the ASPECTS scale, with the largest showing values of 6. However, there were few cases of strokes with values less than 7, a cutoff value related to prognosis in other studies.17 It is likely that the lack of association found between TNF alpha levels and stroke size is because bigger strokes were not presented.
Our study had several limitations. (1) The reduced number of patients selected for this sample could interfere with the lack of association between TNF alpha levels and prognostic variables in the functional scales. However, the association between. TNF alpha values and lacunar infarct subtypes is similar to other authors, and the population was randomly selected in order to reduce the sample size and thus the cost of laboratory studies. (2) Follow-up at 3 months was made by phone. This could have interfered with our results. However, the survey was made via a validated telephone Rankin scale which is used routinely by our Service. (3) Strokes with an undetermined etiology were included. Strokes with an undetermined etiology probably arise due to the fact that sometimes not all necessary diagnostic studies are performed, either due to a lack of funds or a lack of adherence to outpatient follow-up. However, since the creation of the database on stroke and systematic registration in the Neurovascular Care Unit in the Department of Neurology, we have reduced this percentage significantly.
ConclusionsSmall vessel disease had significantly lower levels of TNF alpha, while other etiologies were unrelated. However, they were not associated with better functional prognosis at hospital discharge and at follow-up at 3 months. An association between higher levels of TNF alpha and carotid intima-media thickness of more than 1mm was found. More prospective studies are needed with larger numbers of patients to validate these results. Its routine use in pre-hospital evaluation could be useful in deciding whether or not the patient should move to a neurovascular unit with or without endovascular therapy intervention.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
FundingNo financial support was provided.
Conflict of interestThe authors have no conflicts of interest to declare.
Margarito Raúl Hinojosa-García, Meztli Artemisa Espinosa-Ortega and Helda Estrella Sánchez-Terán. Students Against Neurologic Diseases (GECEN by its Spanish acronym) of GESTIMED, School of Medicine at the Autonomous University of Nuevo Leon, Monterrey, Mexico.