To compare clinical, laboratory, treatment and live birth rate data between women with aPL-related obstetric complications (OMAPS) not fulfilling the Sydney criteria and women fulfilling them (OAPS).
Materials and methodsRetrospective and prospective multicentre study. Data comparison between groups from The European Registry on Antiphospholipid Syndrome included within the framework of the European Forum on Antiphospholipid Antibody projects.
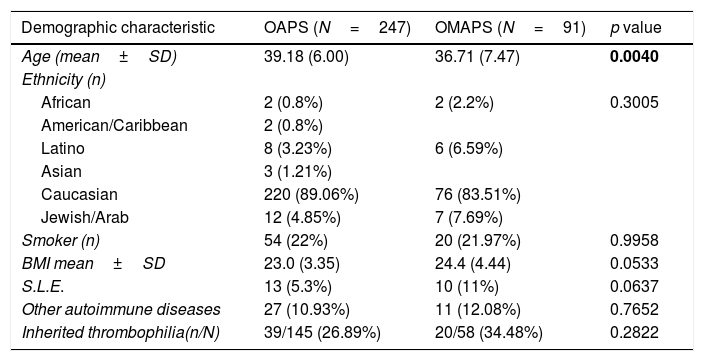
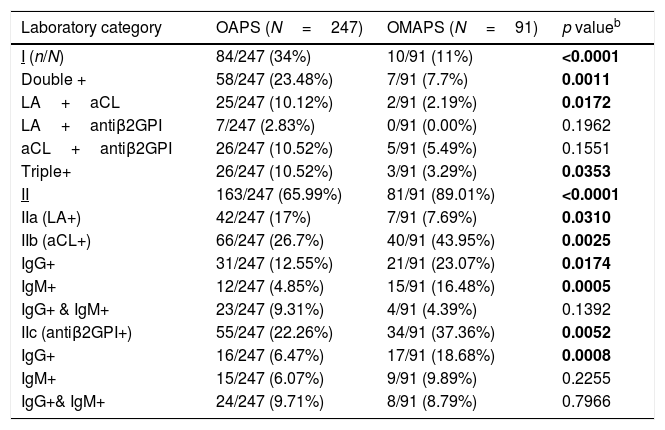
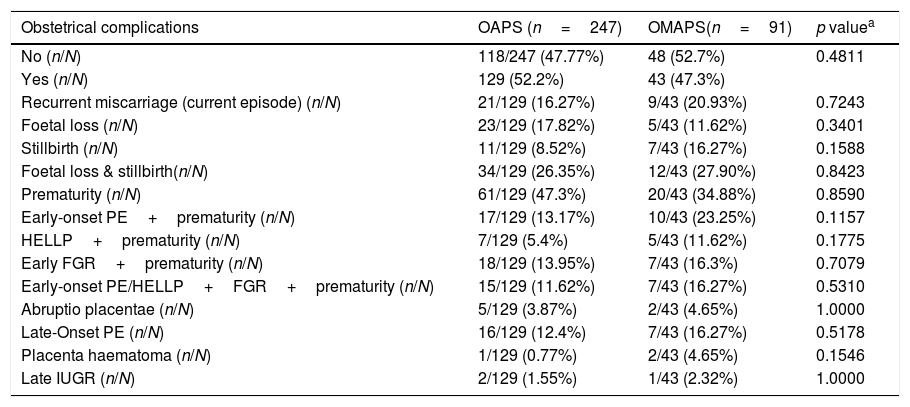
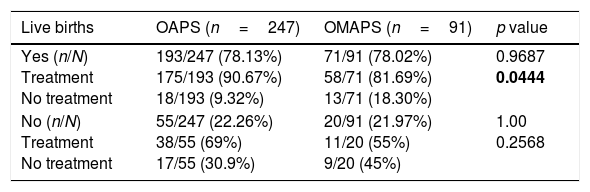
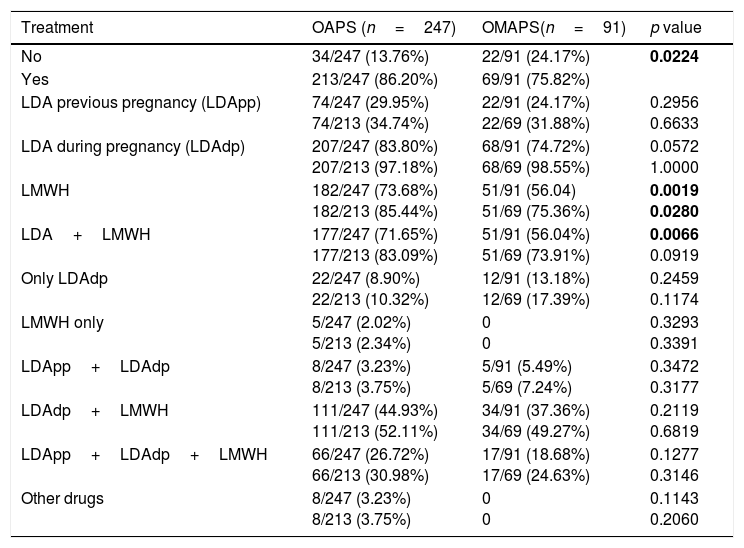
Results338 women were analysed: 247 fulfilled the Sydney criteria (OAPS group) and 91 did not (OMAPS group). In the OMAPS group, 24/91 (26.37%) fulfilled laboratory Sydney criteria (subgroup A) and 67/91 (74.63%) had a low titre and/or non-persistent aPL-positivity (subgroup B). Overall, aPL laboratory categories in OAPS vs. OMAPS showed significant differences: 34% vs. 11% (p<0.0001) for category I, 66% vs. 89% (p<0.0001) for category II. No differences were observed when current obstetric complications were compared (p=0.481). 86.20% of OAPS women were treated vs. 75.82% of OMAPS (p=0.0224), particularly regarding the LDA+LMWH schedule (p=0.006). No differences between groups were observed in live births, gestational, puerperal arterial and/or venous thrombosis.
ConclusionsSignificant differences were found among aPL categories between groups. Treatment rates were higher in OAPS. Both OAPS and OMAPS groups had similarly good foetal-maternal outcomes when treated. The proposal to modify OAPS classification criteria, mostly laboratory requirements, is reinforced by these results.
Comparar características clínicas, analíticas, tratamiento y tasa de hijos vivos entre gestantes con Síndrome Antifosfolípido Obstèc)trico (SAFO) y gestantes con morbilidad obstèc)trica relacionada con el síndrome que no cumplen los criterios de clasificación actuales.
Material Y mèc)todosEstudio observacional retrospectivo y prospectivo multicèc)ntrico: datos de once hospitales terciarios europeos recogidos en el European Registry on Antiphospholipid Syndrome.
ResultadosSe analizaron 338 mujeres: 247 cumplían criterios de Sydney para SAFO (grupo OAPS), y 91 no (grupo OMAPS). En el grupo OMAPS, 24/91(26.37%) cumplían criterios analíticos, pero no clínicos para SAFO (subgrupo A) y 67/91(74.63%) presentaban títulos medio-bajos o títulos positivos no persistentes de anticuerpos antifosfolípido, con o sin cumplir criterios clínicos (subgrupo B). Se observaron diferencias significativas entre los 2 grupos en cuanto a las categorías analíticas: 34% vs. 11% (p<0.0001) para la categoría I y 66% vs. 89% (p<0.0001) para la categoría II, OAPS vs OMAPS, respectivamente. No se observaron diferencias significativas en cuanto a las complicaciones obstèc)tricas (p=0.481). El 86.20% del grupo OAPS recibió tratamiento vs.el 75.82% del grupo OMAPS (p=0.0224). No se observaron diferencias en la tasa de hijos vivos, ni en la tasa de trombosis arterial y/o venosa gestacional y/o puerperal.
ConclusionesAmbos grupos fueron muy homogèc)neos, excepto en cuanto a la distribución de las categorías analíticas y en la tasa de tratamiento. Ambos grupos mostraron buenos resultados al ser tratados. Los resultados respaldan la opinión de muchos expertos de tener que revisar los criterios de clasificación actuales del Síndrome Antifosfolípido Obstèc)trico.