The Global HPP Registry, an observational, prospective, multinational registry of patients with hypophosphatasia (HPP) (NCT02306720; EUPAS13514), was initiated in 2015. The aim of this study is to assess the symptomatology and impact of the disease in patients included in the national registry.
MethodsBaseline characteristics of patients included in the Registry by national centres were analysed (5/11/2015–4/03/2024).
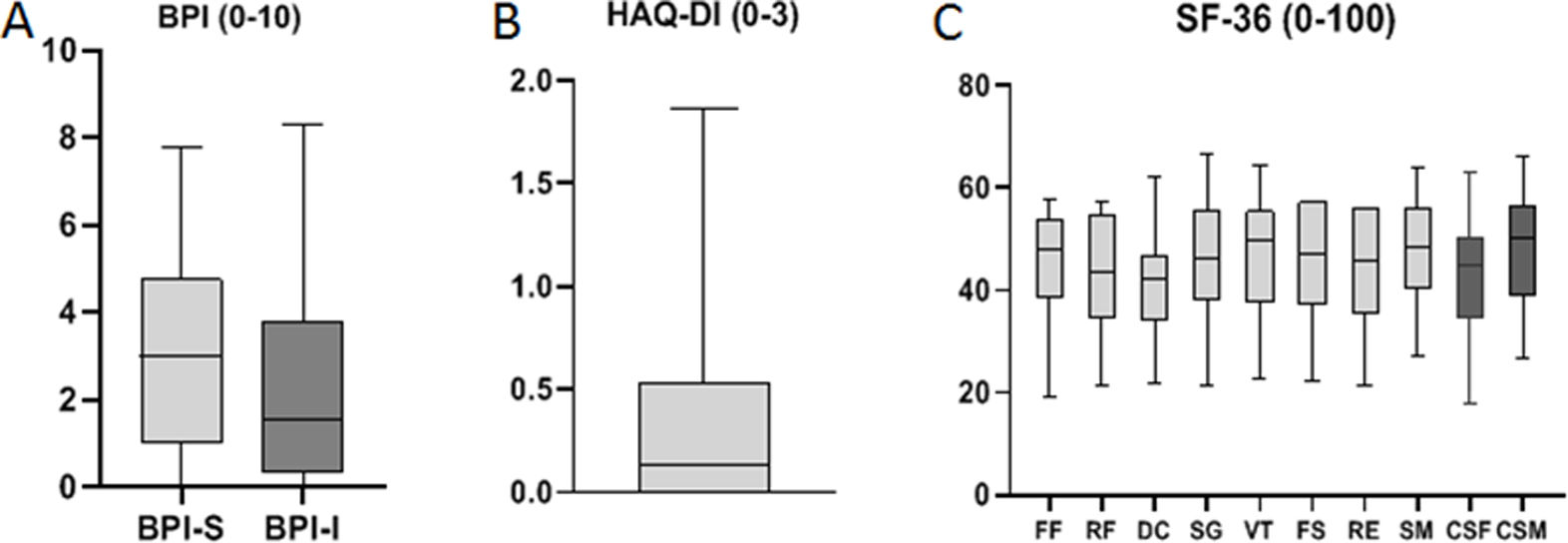
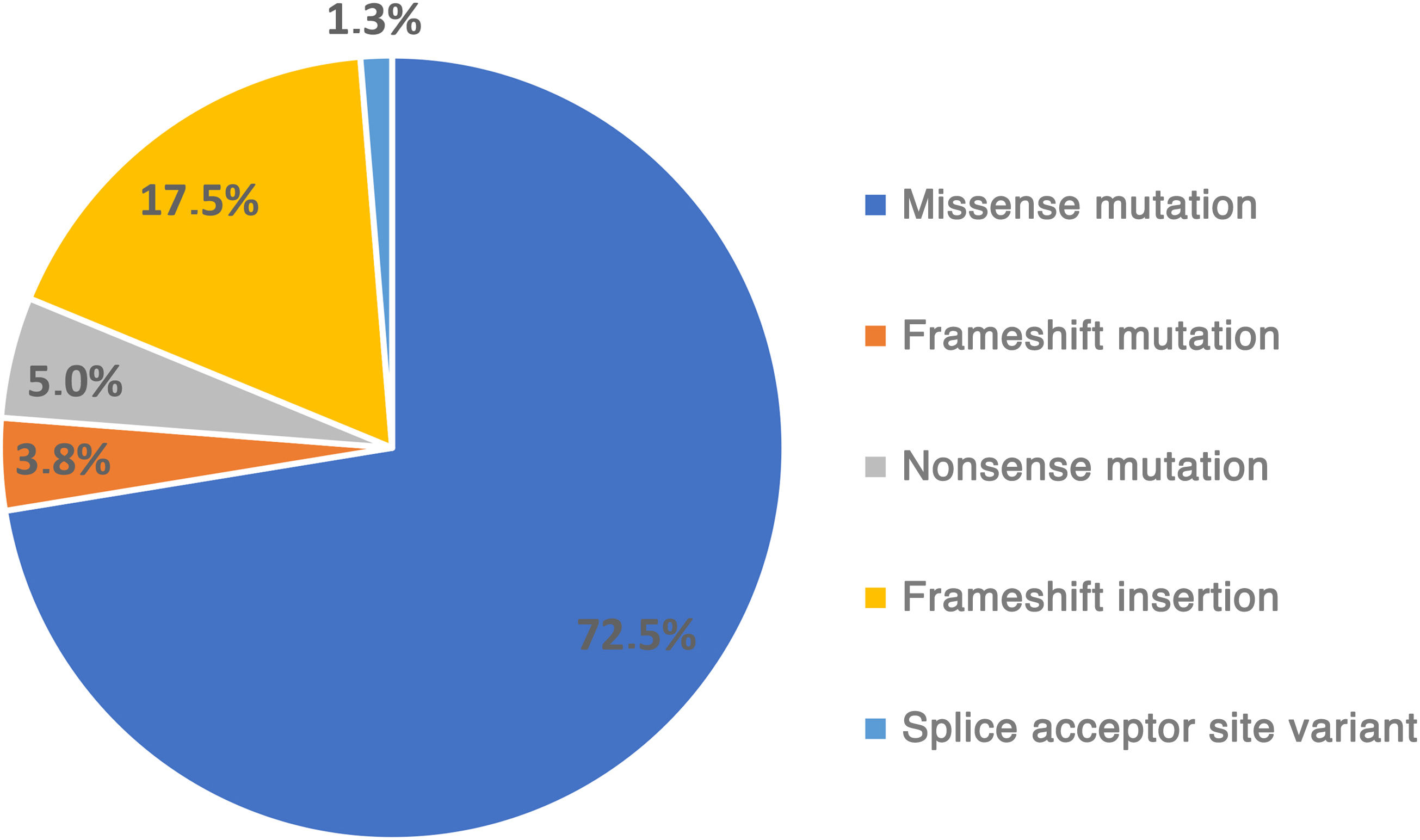
ResultsOf the 83 patients, 16 (19.3%) were under 18 years of age and 67 (80.7%) were adults. Median time to diagnosis was 4.8 (minimum: 0; maximum: 49.9) years. In paediatric patients, median age at symptom onset was 4 (minimum: 1.2; maximum: 11.8) years and the most prevalent symptoms were dental (37.5%), neurological (25%), and skeletal (18.8%). 23.7% of adults started with HPP-related symptoms before the age of 18 years and the most frequent symptoms were pain (64.2%), dental (50.7%), and skeletal (32.8%). Heterozygous ALPL variants were found in 96.3% of patients and the most frequent were c.343_348dup, c.334 G > C, and c.407 G > A. In adult patients, patient-reported outcomes (median [Q1; Q3]) on the quality of life questionnaire (SF-36v2) (scales 0–100) were 44.8 (34.5;50.2) for the physical component and 50.4 (38.9;56.5) for the mental component.
ConclusionsThere is a significant delay in the diagnosis of HPP. The most frequent manifestations were dental in paediatric patients and pain in adults, with a relevant impact on quality of life.
En 2015 se inició el Global HPP Registry, un registro observacional, prospectivo y multinacional de pacientes con hipofosfatasia (HPP) (NCT02306720; EUPAS13514). El objetivo de este estudio es evaluar la sintomatología e impacto de la enfermedad en los pacientes incluidos en el registro nacional.
MétodosSe analizaron las características basales de los pacientes incluidos en el Registro por los centros nacionales (5/11/2015-4/03/2024).
ResultadosDe los 83 pacientes, 16 (19,3%) eran menores de 18 años y 67 (80,7%) adultos. El tiempo (mediana) hasta el diagnóstico fue 4,8 (mínimo: 0; máximo: 49,9) años. En los pacientes pediátricos, la edad (mediana) al inicio de síntomas fue 4 (mínimo: 1,2; máximo: 11,8) años y los más prevalentes fueron dentales (37,5%), neurológicos (25%) y esqueléticos (18,8%). El 23,7% de adultos comenzó con síntomas relacionados con HPP antes de los 18 años y los más frecuentes fueron el dolor (64,2%), los dentales (50,7%) y esqueléticos (32,8%). El 96,3% de pacientes mostraron variantes enALPL en heterocigosis y las más frecuentes fueron c.343_348dup, c.334 G > C y c.407 G > A. En pacientes adultos, los resultados informados por el paciente (mediana [Q1; Q3]) en el cuestionario para la valoración de la calidad de vida (SF-36v2) (escalas de 0–100) fueron 44,8 (34,5;50,2) para el componente físico y 50,4 (38,9;56,5) para el mental.
ConclusionesExiste un retraso significativo en el diagnóstico de la HPP. Las manifestaciones más frecuentes fueron dentales en pacientes pediátricos y el dolor en adultos, con un impacto relevante en la calidad de vida.