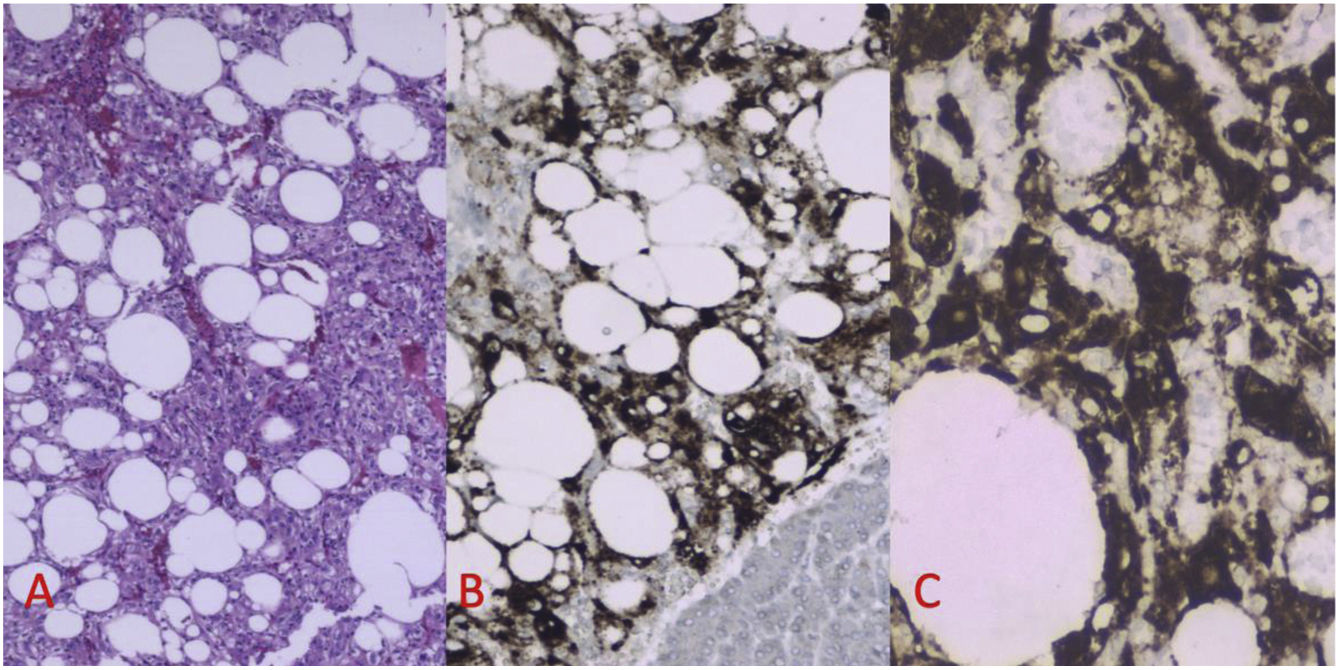
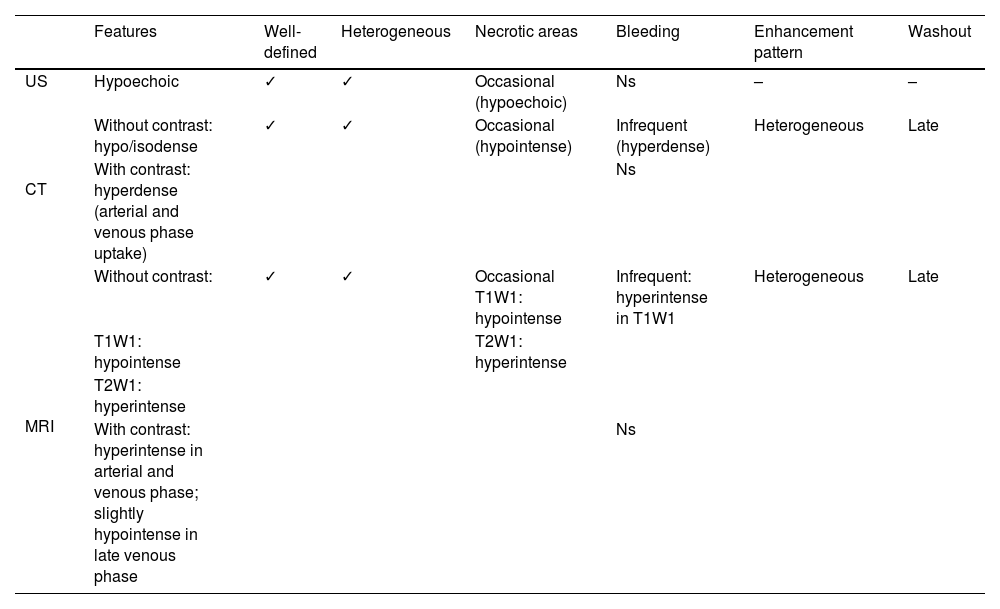
Pancreatic PEComas are extremely rare neoplasms with malignant potential, which mostly affect middle-aged women and are characterized by presenting melanocytic and myogenic markers in immunohistochemical analysis. There are no symptoms or pathognomonic imaging tests, so the diagnosis is established with the analysis of the surgical specimen or the FNA obtained with preoperative endoscopic ultrasound. The mean treatment consists on radical excision, adapting the intervention to the location of the tumor. To date, 34 cases have been described; however, more than 80% of them have been reported in the last decade, which suggests that it is a more frequent pathology than expected. A new case of pancreatic PEComa is reported and a systematic review of the literature is carried out according to the PRISMA guidelines with the aim of divulge this pathology, deepening its knowledge and updating its management.
Los PEComas pancreáticos son neoplasias con potencial maligno extremadamente raras, que afectan mayoritariamente a mujeres de mediana edad y que se caracterizan por presentar marcadores melanocíticos y miogénicos en el análisis inmunohistoquímico. No existen síntomas ni pruebas de imagen patognomónicas. El diagnóstico se establece con el análisis de la pieza quirúrgica o de la PAAF obtenida con ecoendoscopia preoperatoria. El tratamiento más habitual consiste en la exéresis radical, adecuando la intervención a la localización del tumor. Hasta la fecha se han descrito 34 casos, no obstante, más del 80% de los mismos han sido reportados en la última década, lo que sugiere que es una patología más frecuente de lo esperado. Se reporta un nuevo caso de PEComa pancreático y se realiza una revisión sistemática de la literatura de acuerdo con las guías PRISMA con el objetivo de difundir su existencia, profundizar en su conocimiento y actualizar su manejo.